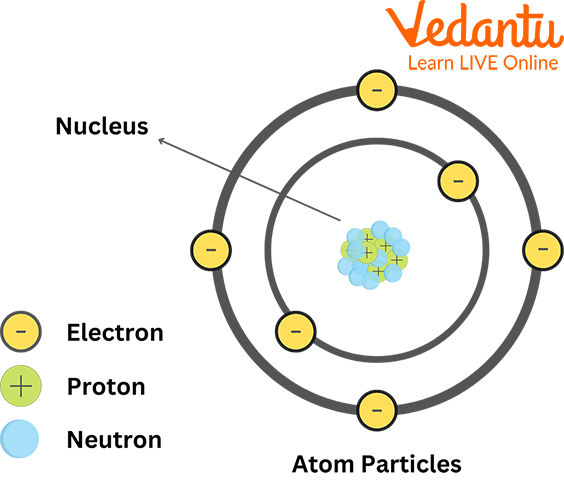
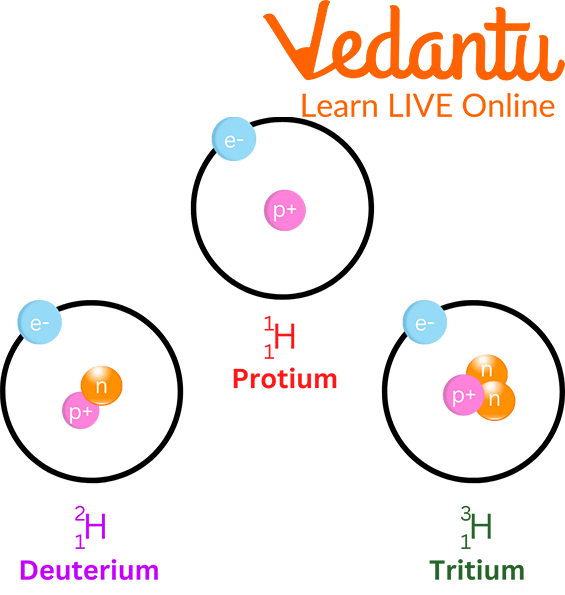
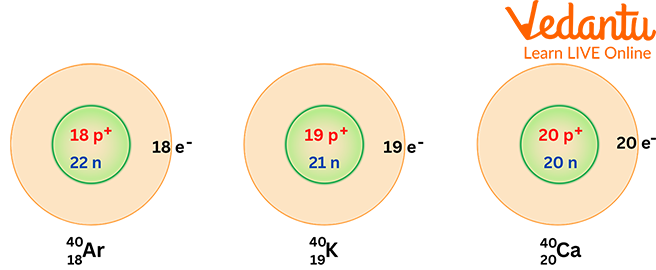
Overview of Chemistry Terms
Chemistry Formulas
FAQs on Basic Chemistry Terms
1. Why is it important to learn about basic Chemistry terms?
It is important to learn about basic Chemistry terms because learning proper chemistry words will help you fully understand the presence of different materials surrounding you and the risk levels of hazardous chemicals around your home.
2. Who is the father of Chemistry?
Antonie Laviosier is popularly known as the father of Chemistry
3. Who discovered the atom?
A chemist, John Dalton, is credited with the discovery of the atom.
4. What is Chemistry?
Chemistry is the study of matter, including its composition, characteristics, and processes of change.
It is the branch of science that examines the characteristics, make-up, and structure of substances (described as elements and compounds), as well as the changes they go through and the energy that is expended or consumed. Chemicals are present in everything and everywhere. Chemicals can be found in anything you touch, smell, or see. While most happen naturally, some are man-made.
For more information, refer to https://www.vedantu.com/chemistry
5. Why is it important to study chemistry?
Because it unites the fields of physics and mathematics, biology and medicine, and the earth and environmental sciences, chemistry is frequently referred to as the central science. Reading and comprehending product labels is made easier with a basic understanding of chemistry. Chemicals are present in everything you can touch, taste, or smell. So studying chemistry is essential if you want to comprehend the world around you.
For more information, refer to https://www.vedantu.com/chemistry
6. What is organic chemistry?
The area of chemistry known as "organic chemistry" is dedicated to the investigation of organic substances (compounds that contain covalently bonded carbon atoms). Another way to view organic chemistry would be to think of it as the study of molecules with the carbon-hydrogen (C-H) bond and their reactions because not all carbon reactions are organic.
7. What is inorganic chemistry?
Inorganic chemistry is the area of chemistry that deals with the study of substances that don't contain carbon-hydrogen atoms. Life cannot exist without inorganic substances like carbon dioxide, oxygen, minerals, and water. In a nutshell, organic chemistry is the reverse of inorganic chemistry. Numerous sectors of the chemical industry use inorganic chemistry, including catalysis, materials science, pigments, surfactants, coatings, pharmaceuticals, fuels, and agriculture.
8. What is physical chemistry?
The area of chemistry known as physical chemistry is focused on the investigation of how matter behaves at the atomic or molecular level. It attempts to quantify, correlate, and explain the quantitative features of reactions by focusing, in contrast to other branches, on the physics principles that underlie all chemical interactions (such as the gas laws). It serves as a stepping stone between basic forces and sophisticated life.