



Proven Strategies to Memorize Elements & Ace Your Chemistry Exams
We know that when it comes to the periodic table, it is considered to be one of the most challenging topics in Chemistry. Students spend hours memorising it and yet fail to recall it when required. The periodic table is an arrangement of chemical elements within the form of a table based on their atomic numbers and electronic configurations. The elements are unit arranged by their periods horizontally, whereas the same groups are unit placed vertically below one another. The elements in similar groups have similar properties. Here, we present a unique and easy approach of nailing down the periodic table, and you may for sure retain it without too much effort!
Memorise the Periodic Table
How you memorise the table depends on what works best for you and your learning style. However, here are some recommendations that will help:
Break Down the Table into Sections: You may memorise element groups (different colour groups), go one row at a time, or learn in sets of twenty elements. It's going to be useful to view an ordered list of the elements.
Use Colour to Learn Element Groups: If you wish to learn the element groups additionally to element symbols and names, follow writing the elements using completely different coloured pencils or markers for every element group.
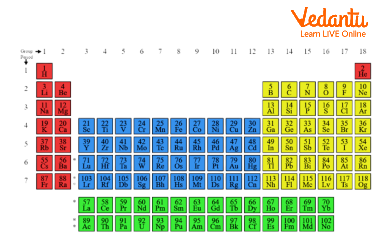
The Periodic Table
Print multiple copies of the blank periodic table to practise filling within the symbols or names of the elements. It's best to learn the element symbols that go along with the names, write in the symbols, and then add the names.
Start little, with one or 2 rows or columns at a time. Whenever you get a chance, write out what you remember, so add to it. If you get bored learning the elements sequentially, you can skip round the table, however it's tougher to recollect that info weeks or years down the road. If you memorise the table, it's worth committing to your memory, therefore learn it over time (days or weeks) and apply writing it out.
Trick to Learn the Periodic Table
The Modern Periodic Table has eighteen vertical columns called ‘groups’ and seven horizontal rows called ‘periods’. Let us see what decides the placing of a component in a bound group and period.
K Shell – 2 × (1)2 = 2, therefore, the first period has two elements.
L Shell – 2 × (2)2 = eight, therefore, the second period has eight components.
M Shell – 2 × (3)2 = eighteen, however the outermost shell will have only eight electrons, therefore the third period conjointly has only eight elements.
Periodic Table Learning Trick
It is helpful when you put efforts in finding patterns and origin when learning the periodic table. Elements are organised on the table according to their atomic number, usually found near the top of the square. The atomic number refers to the number of protons an atom of that element has. For example, H has 1 proton, therefore its atomic number is one.
Dmitri Ivanovich Mendeleev was born in Tobolsk in Western Siberia, Russia on 8 February 1834. The arrangement of elements he proposed is termed Mendeléev’s periodic table.
The periodic table proved to be the unifying principle in chemistry. It was the motivation for the invention of some new elements.
Periodic Table consists of names, symbols, atomic numbers, and atomic weight of the component.
The table is organised in 2 manners, elements within each vertical column are similar or have one thing in common known as groups whereas elements in one period have a similar number of electron shells forms.
Metals reside on the left side of the table, whereas non-metals reside on the right.
There are 118 confirmed elements within the periodic table. Among those, ninety elements are often found in nature, others are artificial .
Mind Tricks to Help
Create Relations – Try and remember the element with the help of a background story. Try to establish relations between elements and things that might help you memorise them.
Mnemonic Devices – Use words that you’ll be able to associate with the elements. You'll be able to create something that rhymes or reads like a string.
Make Use of Images – Pictures are often used to associate elements. how does one recognise that A stands for Apple? It’s simply your brain associating the alphabet with the word.
Make Use of Songs – You can place the name of elements into songs and then sing them to memorise them.
Make a Phrase – You can remember using the primary letters or symbols of the elements. For instance, for the primary 9 elements, you may use:
Happy Hector Likes Beer But Couldn't Obtain Food.
H - Hydrogen
He - Helium
Li - Lithium
Be - Beryllium
B - boron
C - Carbon
N - Nitrogen
O - oxygen
F - Fluorine
Summary
Memorising the periodic table is vital because it's organised and provides loads of information concerning elements. Also, it makes students perceive how elements relate to one another. It's used to predict the properties of every element. Groups and periods provide info concerning the elements sharing similar characteristics. The table provides important info which may be utilised in the equations.
FAQs on Master the Periodic Table: Essential Guide for Students
1. What is the easiest way to memorise the periodic table?
You could use the acronym HHeLiBeBCNOF (pronounced 'Beelzebub kernoff') to remember the first 9 chemical components. It is a nonsense word; however, it condenses 9 names into one mental prompt or cue. Or the acrostic “Here He Lies below Bed clothes, Nothing On, Feeling Nervous” would equate to H He Li Be B C N O F Ne. Otherwise you can learn a section of the table, go off and do something else, write out what you learned in that 1st section, and try to learn a new section.
2. Do all chemists need to memorise the periodic table?
Memorising the periodic table is very important because it's organised and provides tons of information regarding components. Also, it makes students perceive how components relate to one another. It's used to predict the properties of each element. Students do not need to memorise the periodic table. By putting the elements together in numerous combinations, as we do when we spell words, we are able to build our dictionary, containing all the substances within the universe.
3. What are the first twenty elements of the periodic table?
The periodic table of elements puts all the best-known components into groups with similar properties. This makes it a very important tool for chemists, nanotechnologists and other scientists. If you get to understand the periodic table, and learn to use it, you will be able to predict how chemicals will behave. Lithium, Beryllium, Sodium, Magnesium, Aluminium, Potassium, and Ca are metals within the first twenty components. Hydrogen, Helium, Carbon, Nitrogen, Oxygen, Fluorine, Neon, Phosphorus, Sulphur, Chlorine, and Ar are the non-metals within the first twenty components.





































