



Chromatography Methods & Everyday Applications
We all know that a mixture is a combination of different elements or compounds. These elements are mixed uniformly and cannot be separated normally. Then how can we separate the elements of a mixture? Chromatography is a physical method of separation of different components of a mixture.
In this article, we are going to understand what is chromatography, types of chromatography, how can chromatography be used to separate mixtures, and how two dimensional chromatography is used for separating mixtures.
What is Chromatography?
Chromatography is a physical method of separation of different components of a mixture. In chromatography, the components to be separated are distributed between two phases, one of which is stationary while the other moves in a definite direction.
Types of Chromatography
On the basis of phases of separation of elements of a mixture, chromatography is divided into the following categories:
Column Chromatography - In column chromatography, mobile phase flows through the packed column.
Thin Layer Chromatography - In thin layer chromatography, the mobile part moves by capillary action.
Partition Chromatography - In partition chromatography, the stationary part is thin film of liquid adsorbable on an essentially inert support.
Paper Chromatography - Paper chromatography is an example of partition chromatography within which liquid present within the pores of paper is stationary phase and a few other liquid is movable part.
Absorption Chromatography - In adsorption chromatography, the stationary part could be a finely divided solid adsorbent and the mobile part is typically a liquid.
How Can Chromatography Be Used to Separate Mixtures?
The most widely used methodology of chromatography is paper chromatography. Chromatography separates components in a mixture according to their solubility within the mobile part and their absorbance to the stationary part. The mobile phase is the part of the chromatography set up that moves; the stationary part is the part of the set up that does not move.
Components that are more soluble within the mobile phase, i.e. adsorb less well to the stationary part, will move through the chromatography set up quicker as a result of there are fewer interactions with the stationary section to slow the component down. These components are going to be observed initially at the end point. Components that adsorb more powerfully to the stationary part have more interactions with the stationary part to block the movement of the component. These parts will be observed last at the end point.
Two Dimensional Chromatography
Two dimensional chromatography is used for separating mixtures. The combination of various separation techniques into one experiment (multi-dimensional chromatography; additionally known as 2D chromatography, orthogonal chromatography and cross-fractionation) permits for tackling the core of restricted chromatographic resolution by vastly improving the peak capability. This is specifically vital for the analysis of advanced polymeric materials.
In 2-dimensional chromatography, the sample is fractionated in a first dimension by one structural parameter and the fractions are collected. These fractions are then afterwards separated in a second chromatographic experiment separating according to a second structural feature.
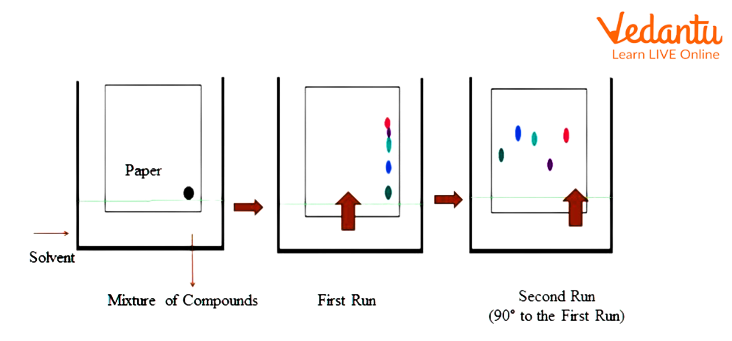
2D Paper Chromatography
Two dimensional chromatography could be an advanced setup that is employed to separate complex mixtures.
The solvent is placed at the bottom of the tank and also the filter paper saturated with the stationary part is then kept within the tank. The development occurs upwards but very slowly because it's against gravity, additionally, as the compound could be a complex one.
After a few hours, the filter paper is turned 90 degree clockwise and also the tank is filled with a different kind of solvent. If there's no pronounced separation then development proceeds to the “c” stage.
Again the filter paper is turned ninety degrees clockwise and another solvent is used. This results in a satisfactory separation.
Although this may take some time, this allows a high degree of separation.
Interesting Facts
Chromatography is helpful in determining the antibodies that fight numerous diseases. Scientists used chromatography in the fight against the Ebola virus, responsible for over 11,000 deaths. The process was used to resolve which antibodies are the most effective at neutralising the deadly virus.
Chromatography is additionally used to facilitate catching criminals. In line with programs like CSI, gas chromatography (GC) is employed to research blood and fabric samples, helping to identify criminals and bring them to justice.
Conclusion
Chromatography is a physical method of separation of different components of a mixture. In chromatography, the components to be separated are distributed between two phases, one of which is stationary while the other moves in a definite direction. On the basis of phases of separation of elements of a mixture, chromatography is divided into the five different categories. Two dimensional chromatography is used for separating mixtures.
FAQs on Chromatography Explained: Key Concepts for Students
1. What is chromatography in simple terms?
Chromatography is a laboratory technique used to separate the different components of a mixture. Think of it as a race where different substances in the mixture travel at different speeds through a material (the stationary phase) while being carried along by a fluid (the mobile phase), causing them to separate out based on their unique properties.
2. What is the fundamental principle behind chromatography?
The basic principle of chromatography is differential distribution. This means the components of a mixture distribute themselves differently between two phases: a stationary phase (which is fixed in place, like paper or a solid in a column) and a mobile phase (a liquid or gas that moves over the stationary phase). Separation occurs because each component has a different affinity for the two phases, causing them to move at different rates.
3. What are the main types of chromatography based on their separation mechanism?
Based on the principle of separation, chromatography is broadly classified into two main types as per the NCERT syllabus:
- Adsorption Chromatography: In this type, separation is based on the differential adsorption of components onto the surface of a solid stationary phase like silica gel. Components that adsorb more strongly travel slower.
- Partition Chromatography: Here, separation is based on the continuous differential partitioning or distribution of components between a liquid stationary phase and a liquid or gaseous mobile phase. Paper chromatography is a common example.
4. How is the Retention Factor (Rf value) calculated in paper chromatography and what is its significance?
The Retention Factor (Rf) is a crucial measurement in paper and thin-layer chromatography. It is calculated using the formula:
Rf = (Distance travelled by the component) / (Distance travelled by the solvent front)
Its primary significance is in identifying substances. Under a constant set of conditions (the same stationary phase, mobile phase, and temperature), a specific compound will always have a characteristic Rf value, allowing for its identification by comparing it to known standards.
5. What are some key real-world applications of chromatography?
Chromatography has numerous important applications across various fields:
- Pharmaceutical Industry: To ensure the purity of drugs and to separate chiral compounds.
- Forensic Science: To analyse blood and urine samples for drugs, or to test crime scene evidence like ink or fibres.
- Food Industry: To detect spoilage, measure nutritional quality, and identify unauthorised additives.
- Environmental Testing: To monitor air and water pollution by identifying trace amounts of contaminants.
6. How do the mobile phase and stationary phase work together to separate components in a mixture?
The mobile phase and stationary phase work together through a balance of competing interactions. The mobile phase attempts to carry all components of the mixture forward. Simultaneously, the stationary phase tries to hold onto the components through physical or chemical forces like adsorption or solubility. A component that is more soluble in the mobile phase and has a weaker attraction to the stationary phase will travel faster. Conversely, a component with a strong affinity for the stationary phase will be held back and travel more slowly, leading to effective separation.
7. What is the key difference between adsorption chromatography and partition chromatography?
The key difference lies in the nature of the stationary phase and the resulting separation mechanism. In adsorption chromatography, the stationary phase is a solid adsorbent, and separation occurs based on how strongly different components stick (adsorb) to its surface. In partition chromatography, the stationary phase is a liquid coated onto a solid support, and separation is based on the different solubilities (or partitioning) of components between the stationary liquid and the mobile phase. Essentially, it is a difference between surface adsorption versus bulk distribution/solubility.
8. Why would a scientist choose to use two-dimensional (2D) chromatography instead of a single separation?
A scientist would opt for 2D chromatography when analysing extremely complex mixtures where a single separation is insufficient to resolve all components. After an initial separation with one solvent, the paper or plate is rotated 90 degrees and developed with a different solvent system. This second separation resolves components that were not separated in the first run, vastly increasing the overall resolving power and allowing for a much cleaner separation of individual substances from a complex sample.
9. In High-Performance Liquid Chromatography (HPLC), how does applying high pressure improve the separation?
Applying high pressure in HPLC is crucial because it allows the use of a column packed with very fine stationary phase particles. These tiny particles provide a much larger surface area for interactions, leading to significantly better and more efficient separation. The high pressure forces the mobile phase through this tightly packed column at a fast and constant rate, which reduces the analysis time and minimises the diffusion of separated components, resulting in sharper, more well-defined peaks and superior resolution compared to standard column chromatography.
10. The name 'chromatography' means 'colour writing'. Why was it named this, and is the technique limited to separating only coloured substances?
The technique was named by the botanist Mikhail Tsvet, who first used it to separate plant pigments like chlorophylls. These pigments separated into distinct coloured bands on his column, inspiring the name 'chromatography' (from Greek *chroma* for 'colour' and *graphein* for 'to write'). However, the technique is not limited to coloured substances. Modern chromatography is widely used to separate colourless compounds, which are then identified using various detectors that can sense them as they exit the column.
























