



What Are Alkali Metals? Definition, Properties, and Periodic Table Group
Have you ever considered which group of the modern periodic tables contains alkali metals? If not, then don't worry. Group 1 of the periodic table consists of alkali metals along with Hydrogen. There are six elements in the group. These are named Alkali Metals because they form strong bases on reactions with water. This article contains information about alkali metals, important properties of alkali metals and the uses of alkali metals in a simple and easy language for a better understanding of the concepts.
Definition of Alkali Metals
Alkali metals form a part of the leftmost group, i.e. group 1 of the modern periodic table. It contains six elements, which are collaboratively called the Lithium family. These metals are highly reactive metals of the periodic table due to the presence of one electron in their outermost shell, called the valence electron.
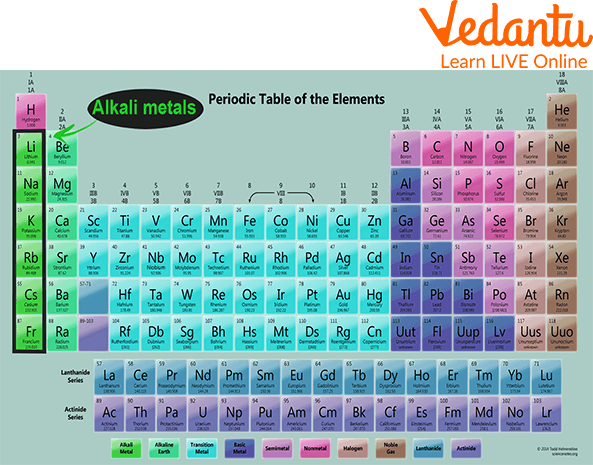
Periodic Table showing Alkali Metals
Properties
Physical and Chemical
The physical and chemical properties of the Alkali Metals can be promptly clarified by their having an ns1 valence Electron setup, which results in frail Metallic holding. Henceforth, all the Alkali Metals are soft and have low densities, melting and bubbling points, and heats of sublimation, vaporization, and dissociation. They all solidify in the body-centered cubic precious stone structure and have particular fire hues on the grounds that their external Electron is all-around effectively excited. The ns1 setup likewise results in the Alkali Metals having huge nuclear and ionic radii, just as high heat and electrical conductivity. Their Chemistry is overwhelmed by the loss of their solitary valence Electron in the peripheral s-orbital to obtain the +1 oxidation state, because of the simplicity of ionizing this Electron and the high second ionization energy. Most of the Chemistry has been observed just for the first five individuals from the group. The Chemistry of francium isn't settled because of its very high level of radioactivity; accordingly, the introduction of its properties here is constrained. What little that is thought about francium demonstrates that it is exceptionally close in conduct to cesium, not surprisingly? The physical properties of francium are much sketchier in light of the fact that the mass component has never been observed; subsequently, any information that might be found in the writing is surely theoretical extrapolations.
Lithium
The Chemistry of Lithium demonstrates a few contrasts from that of whatever is left of the group. Lithium and magnesium have a diagonal relationship due to their comparable nuclear radii, with the goal that they demonstrate a few similitudes. For instance, Lithium frames a steady nitride, a property normal among all the soluble earth Metals (magnesium's group) however exceptional among the Alkali Metals. Further, among their particular groups, just Lithium and magnesium structure organoMetallic compounds with critical covalent character (for example LiMe and MgMe2). Lithium fluoride is the first Alkali Metal halide that is inadequately dissolvable in water, and Lithium hydroxide is the first Alkali Metal hydroxide that isn't deliquescent. Conversely, Lithium perchlorate and other Lithium Alkalis with vast anions that can't be enraptured are considerably more steady than comparable to compounds of the other Alkali Metals, presumably in light of the fact that Li+ has high solvation energy. This impact likewise implies that most straightforward Lithium Alkalis are usually experienced in hydrated structure, on the grounds that the anhydrous structures are amazingly hygroscopic: this permits Alkalis like Lithium chloride and Lithium bromide to be utilized in dehumidifiers and forced air systems.
Francium
Francium is likewise anticipated to demonstrate a few contrasts because of its high nuclear weight, making its Electrons travel at significant parts of the speed of light and hence making relativistic impacts increasingly conspicuous. As opposed to the pattern of diminishing Electronegativities and Ionization energies of the Alkali Metals, francium's Electronegativity and Ionization energy are anticipated to be higher than cesium because of the relativistic adjustment of the 7s Electrons; likewise, its nuclear span is relied upon to be strangely low. Hence, in spite of the expectation, cesium is the most receptive of the Alkali Metals, not Francium.
Nuclear
All the Alkali Metals have odd nuclear numbers; thus, their isotopes must be either odd– odd (both proton and neutron numbers are odd) or odd-even (proton number is odd, however, neutron number is even). Odd– odd cores have even mass numbers, while odd-even cores have odd mass numbers. Odd– odd primordial nuclides are uncommon in light of the fact that most odd– odd cores are exceedingly unstable with beta decay, on the grounds that the decay items are even-even, and are hence all the more emphatically bound, because of atomic blending impacts.
Periodic Trends
The Alkali Metals are more like each other than the components in any of the other groups are to each other. For example, while moving down the table, all the discovered and recognized Alkali Metals show expanding nuclear radius, diminishing Electronegativity, expanding reactivity, and diminishing, dissolving, and bubbling points just as heats of fusion and vaporization. By and large, their densities rise while moving down the table, with the exemption that potassium is less thick than sodium.
Atomic and Ionic Radii
The nuclear radii of the Alkali Metals rise, going down the group. Because of the protecting impact, when a particle has more than one Electron shell, every Electron feels electric repugnance from alternate Electrons just as electric attraction from the nucleus. In the Alkali Metals, the peripheral Electron just feels a net charge of +1, as a portion of the atomic charge (which is equivalent to the atomic number) is dropped by the internal Electrons; the quantity of inward Electrons of an Alkali Metal is constantly one less than the atomic charge. In this manner, the first factor which influences the nuclear range of Alkali Metals is the number of Electron shells. Since this number rises down the group, the nuclear range should likewise move down the group.
First Ionization Energy
The first ionization energy of a component or particle is the energy required to move the most loosely held Electron from one mole of vaporous atoms of the component or atoms to shape one mole of vaporous particles with electric charge +1. The factors influencing the primary ionization energy are the atomic charge, the measure of protection by the internal Electrons, and the separation from the most loosely held Electron from the core, which is dependably an external Electron in fundamental group components. The initial two components change the viable atomic charge the most loosely held Electron feels. Since the peripheral Electron of Alkali Metals dependably feels the equivalent compelling atomic charge (+1), the first factor which influences the principal ionization energy is the separation from the furthest Electron to the core. Since this separation rises down the group, the furthest Electron feels less attracted to the core and along these lines the primary ionization energy diminishes.
Reactivity
The reactivity of the Alkali Metals rises going down the group. This is the consequence of a blend of two factors: the first ionization energy and the atomization energy of the Alkali Metals. Since the first ionization energy of the Alkali Metals diminishes down the group, it is less demanding for the peripheral Electron to be expelled from the atom and participate in chemical reactions, hence increasing reactivity down the group.
Electronegativity
Electronegativity is a chemical property that portrays the tendency of an atom to pull in Electrons (or Electron thickness) towards itself. If the bond among sodium and chlorine in sodium chloride were covalent, the pair of shared Electrons would be pulled into the chlorine on the grounds that the viable atomic charge on the external Electrons is +7 in chlorine yet is just +1 in sodium.
Melting and Boiling Points
The melting point of a substance is where it changes its state from solid to liquid while the boiling point of a substance (in the fluid state) is where the vapor pressure of the fluid equals the atmospheric pressure encompassing the liquid and all the fluid changes state to gas. As the Metal is heated to its melting point, the Metallic bonds keeping the atoms set up debilitate with the goal that the molecules can move around, and the Metallic bonds in the long run break totally at the Metal's boiling point. Therefore, the falling, melting, and boiling points of the Alkali Metals demonstrate that the quality of the Metallic obligations of the Alkali Metals diminishes down the group. This is on the grounds that Metal particles are held together by the electromagnetic force from the positive particles to the delocalized Electrons.
Density
The Alkali Metals all have a similar structure (body-centered cubic) and along these lines, the density is the mass of the total number of atoms that can fit into a specific volume. The principal factor relies upon the volume of the molecule and hence the nuclear range, which rises going down the group; along these lines, the volume of an Alkali Metal atom rises going down the group. The mass of an Alkali Metal atom likewise rises going down the group. In this manner, the pattern for the densities of the Alkali Metals relies upon their nuclear loads and nuclear radii; if figures for these two factors are known, the proportions between the densities of Alkali Metals would then be able to be determined.
Alkali Metal, any of the six compound parts that make up Group 1 (Ia) of the unpredictable table—to be express, Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs), and Francium (Fr). The salt Metals are demonstrated considering the way that they respond with water structures Alkalies (i.e., solid bases fit for killing acids). Sodium and potassium are the 6th and seventh generally plentiful of the parts, including, autonomously, 2.6 and 2.4 percent of Earth's outside. The other dissolvable base Metals are in a general sense seriously astonishing, with rubidium, Lithium, and cesium, freely, illustrating 0.03, 0.007, and 0.0007 percent of Earth's outer layer. Francium, a brand name radioactive isotope, is especially remarkable and was not found until 1939.
The Alkali Metals are responsive in that they are generally found in nature and get along with different parts. Fundamental minerals, like halite (sodium chloride, NaCl), sylvite (potassium chloride, KCl), and carnallite (a potassium-magnesium chloride, KCl · MgCl2· 6H2O), are dissolvable in water and along these lines are reasonably confined and filtered. Truly dumbfounding, water-insoluble minerals are, regardless, absolutely more adequate in Earth's casing. A phenomenally cripple gas of nuclear sodium (around 1,000 particles for each cubic cm [about 16,000 particles for each cubic inch]) is passed on in Earth's mesosphere (tallness around 90 km [60 miles]) by clearing of meteors. Coming about the response of sodium with ozone and nuclear Oxygen produces strengthened sodium particles that send the light we consider the "tail" of a meteor likewise as the more diffuse climatic nightglow. More unpretentious extents of Lithium and potassium are in like ways present.
The Alkali Metals have the silver-like brilliance, high flexibility, and superb conductivity of power and hotness, everything considered related with Metals. Lithium is the lightest Metallic part. The salt Metals have low progressing focuses, going from a high of 179 °C (354 °F) for Lithium to a low of 28.5 °C (83.3 °F) for cesium. Composites of Alkali Metals exist that gather as low as −78 °C (−109 °F).
The salt Metals respond rapidly with barometrical Oxygen and water fumes. (Lithium likewise responds with nitrogen.) They respond eagerly, and regularly violently, with water to pass on hydrogen and develop solid burning strategies. Most common nonMetallic substances like brilliant light, halogen acids, sulfur, and phosphorus respond with the Alkali Metals. The salt Metals themselves respond with different normal mixes, especially those containing a halogen or a promptly replaceable hydrogen molecule.
Sodium is by a wide margin the truly salt Metal to the degree present day use. Metal is utilized in the decreasing of ordinary mixes and in the situation with different business compounds. As a free Metal, it is utilized as a hotness move liquid in some atomic reactors. Boundless gigantic stacks of business fortifies that contain sodium are utilized yearly, including run of the mill salt (NaCl), baking pop (NaHCO3), sodium carbonate (Na2CO3), and acidic pop (NaOH). Potassium has through and through less use than sodium as a free Metal. Potassium salts, notwithstanding, are eaten up in incredible burdens in the improvement of excrements. Lithium Metal is utilized in express light-Metal composites and as a reactant in normal blends. A basic utilization of Lithium is in the improvement of lightweight batteries. Principal Lithium batteries (not battery-controlled) are generally utilized in different contraptions like cameras, cell phones, and pacemakers. Battery-controlled Lithium gathering batteries that could be fitting for vehicle main thrust or energy putting away are the subject of concentrated examination. Rubidium and cesium and their mixes have restricted use, yet cesium Metal smoke is utilized in nuclear watches, which are unquestionably precise that they are utilized as time principles.
Stomach settling specialist Metal salts were known to people of old. The Old Testament recommends a salt called neter (sodium carbonate), which was detached from the rubbish of vegetable matter. Saltpeter (potassium nitrate) was hazardous, which was made in China about the tenth century AD and had been brought into Europe by the thirteenth century.
History of Alkali Metal
In October 1807 the English logical master Sir Humphry Davy isolated potassium and subsequently sodium. The name sodium is derived from Italian pop, a term applied in the Middle Ages to all Alkalies; potassium comes from the French potasse, a name used for the development left in the dissemination of liquid courses of action from wood ashes.
Lithium was found by the Swedish logical master Johan August Arfwedson in 1817 while inspecting the mineral petalite. The name Lithium is derived from lithos, the Greek word for "stony." The part was not disconnected in pure construction until Davy conveyed a brief sum by the electrolysis of Lithium chloride.
While the German logical specialists Robert Bunsen and Gustav Kirchhoff were investigating the mineral waters in the Palatinate in 1860, they got a filtrate that was depicted by two lines in the blue region of its reach (the light released when the model was implanted into a fire). They suggested the presence of another dissolvable base part and called it cesium, gotten from the Latin caesius, used to relegate the blue of the sky. Comparative examiners, on eliminating the Alkalies from the mineral lepidolite, detached another game plan, which yielded two shocking lines of red tone. They proposed the name rubidium for the part in this course of action from the Latin rubidus, which was used for the darkest red tone. Francium was not found until 1939 by Marguerite Perey of the Radium Institute in Paris.
In the nineteenth century the principle use for the stomach settling agent Metals was crafted by sodium as a reagent in the collecting of aluminum. Exactly when the electrolytic association for aluminum disinfecting was set up, it made the feeling that immense extension usage of sodium would stop. Following updates in the electrolytic formation of sodium, in any case, decreased the cost of this part such a lot of that it might be used monetarily to manufacture fuel added substances, reagents for compound industry, herbicides, bug harms, nylon, medications, and reagents for Metal refining. The incessant electrolysis of sodium hydroxide, a technique called the Castner cycle, was replaced in 1926 by the Downs cell process. This cycle, wherein a fluid sodium Chloride–Calcium Chloride mix (to decrease the melting point) is electrolyzed, produces both Sodium Metal and Chlorine.
Important Common Properties of Alkali Metals
These metals are highly reactive and, thus, are only found in combined form in nature.
Alkali metals are lustrous, like silver.
These are extremely ductile and malleable.
Alkali metals are good conductors of heat and electricity.
Among all the six alkali metals, lithium is the lightest metallic element.
These have a lower melting point, and caesium has the least melting point.
Uses of Alkali Metals
The uses of alkali metals are given below:
Sodium is the only alkali metal with the most probable use in industries.
Lithium is highly used in batteries but can also be used in lubricating greases and manufacturing aluminium.
Potassium is widely used in producing soaps and fertilisers.
Sodium is generally used in nuclear reactions as a heat transfer fluid and other reactions as a free metal.
Caesium has limited use due to its nature. It is used in the generation of photoelectric cells.
What Do the Alkali Metals All Have in Common?
Many characteristics that all alkali metals have in common include:
These react vigorously with water and oxygen to form compounds.
Each alkali metal contains an electron in its valence shell.
These are highly electropositive and least electronegative metallic elements.
The general electronic configuration of alkali metals is given by [noble gas] ns1.
3 Common Differences Between Lithium and Other Alkali Metals
3 common differences between lithium and other alkali metals are given below:
Lithium has a higher melting point than all other alkali metals.
It is the only alkali metal that shows a reaction with nitrogen.
Lithium has the least density among all the alkali metals.
Lithium is highly electronegative, while all other alkali metals are electropositive.
Summary
In this article, we have covered everything about alkali metals, i.e. the definition of alkali metals, alkali metals elements, the important common properties of alkali metals, and the uses of alkali metals. Images are used to make learning interesting and easy for the students. The knowledge given in this article would be useful for the students in their higher classes.
FAQs on Alkali Metals: Characteristics, Trends & Examples
1. What are alkali metals and where are they located in the periodic table?
Alkali metals are the chemical elements that constitute Group 1 of the periodic table. These elements are Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Cesium (Cs), and Francium (Fr). They are characterised by having a single electron in their outermost shell, which makes them highly reactive.
2. Why are the elements of Group 1 specifically called 'alkali metals'?
The name 'alkali metals' comes from their characteristic reaction with water. When these metals react with water, they form strong hydroxides (like NaOH or KOH), which are basic in nature. These water-soluble bases are known as alkalies, hence the name for the entire group.
3. What are the key physical properties that define alkali metals?
Alkali metals share several distinct physical properties. They are:
- Appearance: Silvery-white, soft, and shiny metals that tarnish quickly upon exposure to air.
- Density: They have very low densities; Lithium, Sodium, and Potassium are even less dense than water.
- Hardness: They are extremely soft and can be easily cut with a knife.
- Melting and Boiling Points: They have low melting and boiling points compared to other metals due to weak metallic bonding.
4. How does the chemical reactivity of alkali metals change as you move down the group?
The chemical reactivity of alkali metals increases significantly as you move down the group from Lithium to Cesium. This trend is because the atomic size increases and the ionisation enthalpy decreases. A lower ionisation enthalpy means the outermost electron is held less tightly by the nucleus and can be lost more easily, leading to a more vigorous chemical reaction.
5. Why are alkali metals some of the most reactive elements?
The high reactivity of alkali metals is primarily due to two factors based on their atomic structure:
- Low Ionisation Enthalpy: They require very little energy to lose their single valence electron.
- Large Atomic Size: The outermost electron is far from the nucleus, reducing the electrostatic attraction and making it easy to remove.
By losing one electron, they readily form a stable cation with a noble gas electronic configuration.
6. Why do alkali metals give a characteristic colour when heated in a flame?
Alkali metals impart a characteristic colour to a flame because their valence electrons have very low ionisation energies. When heated, the flame's energy excites this electron to a higher energy level. As the electron returns to its original ground state, it emits the absorbed energy as light. The wavelength of this emitted light falls within the visible spectrum, resulting in a distinct colour for each element (e.g., Sodium gives a golden yellow flame).
7. What is the fundamental difference between alkali metals and alkaline earth metals?
The primary difference lies in their group number and valence electrons. Alkali metals are in Group 1 and have one valence electron, which they lose to form a +1 ion (M⁺). Alkaline earth metals are in Group 2 and have two valence electrons, which they lose to form a +2 ion (M²⁺). Consequently, alkali metals are generally more reactive and have lower melting points than their alkaline earth metal counterparts in the same period.
8. Why are alkali metals soft and have low melting points compared to most other metals?
Alkali metals are soft and have low melting points due to their weak metallic bonding. Each atom contributes only one electron to the 'sea' of delocalised electrons that holds the metallic lattice together. This, combined with their large atomic size, results in weaker forces between the atoms, making the metal soft and easy to melt.
9. How do alkali metals react with halogens?
Alkali metals react vigorously with halogens (Group 17 elements like chlorine and bromine) to form crystalline ionic salts called metal halides. The general equation for this reaction is 2M + X₂ → 2MX, where M is an alkali metal and X is a halogen. These reactions are highly exothermic, and their reactivity increases down the group.
10. What are some important applications of sodium and potassium in real life?
Sodium and potassium have crucial applications.
- Sodium (Na): Liquid sodium is used as a coolant in nuclear reactors. Sodium vapour is used in streetlights to produce yellow light. Sodium chloride (table salt) is essential for life and used as a preservative.
- Potassium (K): Potassium compounds are vital components of fertilisers to support plant growth. Potassium is also a critical electrolyte in the human body, necessary for nerve function and muscle contraction.




































