



Stepwise Claisen Rearrangement Mechanism with Practice Examples
The investigations on the stereochemistry elements of the Claisen reactions deserve special note. The Claisen rearrangement has generated a great deal of investigation due to its value in synthetic activities, especially in the production of natural compounds, and also its use as an instrument to explore the bond configurations of aromatic compounds and its inherent significance as a molecular rearrangement.
The Claisen rearrangement is a systematic, pericyclic, exothermic reaction that involves bond cleavage and rearrangement. Crossover studies rule out the likelihood of the rearrangement taking place through a mechanism involving an intermolecular interaction and are in line with an intramolecular action. It's crucial to remember that Claisen condensation and Claisen rearrangement are two very different phenomena. This article offers a broad outline of the main important subjects pertaining to the Claisen rearrangement.
What is Claisen Rearrangement?
Claisen rearrangement is known as an organic chemical process which provides a potent way to generate carbon-carbon bond formation. When heated or exposed to a Lewis acid, the initiator of this reaction, for example, allyl vinyl ether, transforms into an unsaturated carbonyl molecule.
The mechanism of Claisen rearrangement exhibits significant solvent influences, with polar solvents having a larger tendency to speed up the process. The maximum reaction rates were produced by hydrogen-bonding solvents. The process is more quickly accelerated by polar solvent influences.
Examples of Claisen Rearrangement
The reorganisation of allyl vinyl ether and allyl aryl ether, such as allyl phenyl ether, is accomplished through this Claisen rearrangement. The latter is employed to prepare allyl phenol. These are some of the Claisen rearrangement examples. A Lewis acid or heat can cause the initiator of this Claisen rearrangement mechanism, allyl vinyl ether, to change towards a gamma, delta-unsaturated carbonyl molecule.
Combining C-C π-Bonds
Formally, the Claisen rearrangement can be described as the intramolecular addition of an allylic ether, a sulphide, or an amine to a carbonyl enol (X = O), thiocarbonyl enol (X = S), or an enamine (X = N), accordingly, generating a carbon-carbon σ-bond. The mechanism, which is categorised as a 3, 3-sigmatropic shifting, includes concurrent π-bond movement. It is typically described as coordinated, even though in reality, a variety of processes may be in play. As a result of hetero- and polyhetero-Claisen rearrangements, heteroatoms may occupy additional places and the unsaturation level might well be greater than indicated.
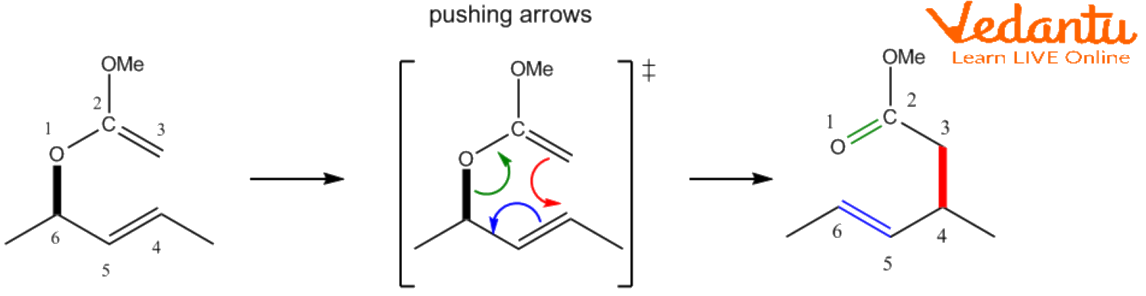
CC 𝝅-Bond
Mechanism of Claisen Rearrangement
A particular kind of sigma tropic rearrangement is the Claisen rearrangement. It is a mechanism in which every bond concurrently forms and breaks. Allyl phenyl ethers can also be used to carry out the Claisen rearrangement mechanism. Allyl phenyl ether is rearranged into its aromatic enol form in Claisen rearrangement. The meta-substitution in this rearrangement has an impact on the regioselectivity. An intermediate is produced via the [3, 3]-sigmatropic rearrangement of the allyl phenyl ether. This intermediate is then subjected to tautomerization, producing a phenol that has an ortho substitution. The allyl phenyl ether Claisen rearrangement is given in below diagram:
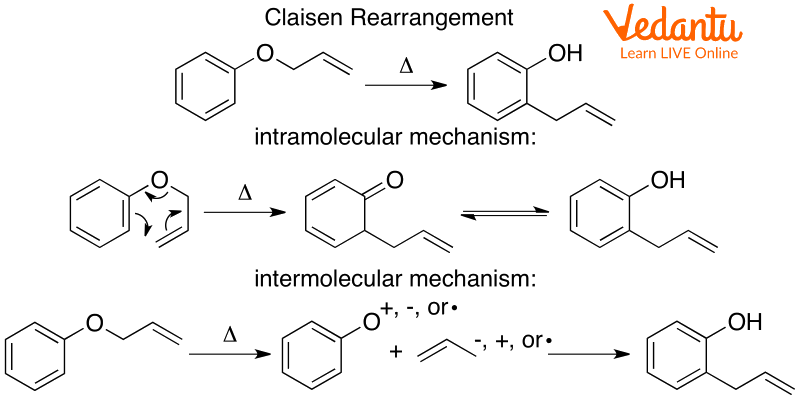
Allyl Phenyl Ether Claisen Rearrangement
Variations
Aside from the standard allyl vinyl ether rearrangement, a number of variants have been created that enhance the Claisen rearrangement's synthetic significance in terms of the formation of the parental compounds, the reactive environment, and stereoselectivity. The different types of variations are listed beneath:
Aromatic Claisen rearrangement
Bellus Claisen rearrangement
Eschenmoser–Claisen rearrangement
Ireland–Claisen rearrangement
Johnson–Claisen rearrangement
Kazmaier–Claisen rearrangement
Photo-Claisen rearrangement
Interesting Facts
The Claisen rearrangement mechanism is called after its inventor, German chemist Rainer Ludwig Claisen, who made the discovery in 1912.
It's significant to note that sigmatropic rearrangement was the first [3, 3]- sigmatropic rearrangement process to ever be documented.
In contrast to usual Claisen rearrangements, zwitterionic Claisen rearrangements take place at or beneath room temperature. In milder circumstances, the acyl ammonium ions are especially selective for Z-enolates.
Key Features to Remember
The organic reaction mechanism known as the Claisen rearrangement provides a potent way to generate carbon-carbon bonds.
Claisen rearrangement is an exothermic, pericyclic reaction. When energy is released, the arranging reaction changes and its intermediary transitional phase is cyclic.
If heated or exposed to a Lewis acid, the source of this synthesis, allyl vinyl ether, transforms into a gamma, delta-unsaturated carbonyl molecule.
The conversion occurs intramolecularly, follows a well-organised cyclic transitional phase, and possesses first-order kinetics.
FAQs on Claisen Rearrangement Explained: Mechanism & Applications
1. What is the Claisen rearrangement in organic chemistry?
The Claisen rearrangement is a powerful, heat-induced chemical reaction where an allyl aryl ether or an allyl vinyl ether rearranges itself. It is an intramolecular process, meaning it happens within a single molecule. Specifically, it's a [3,3]-sigmatropic rearrangement that results in the formation of a new carbon-carbon bond, typically yielding a γ,δ-unsaturated carbonyl compound or, in the case of an aryl ether, an ortho-allylphenol.
2. What is the mechanism of the Claisen rearrangement of allyl phenyl ether?
The mechanism is a concerted, single-step process that occurs through a cyclic six-membered transition state. The key steps are:
- [3,3]-Sigmatropic Shift: Upon heating, the allyl phenyl ether undergoes a pericyclic reaction where the C-O bond breaks and a new C-C bond forms between the allyl group's terminal carbon and the ortho carbon of the benzene ring.
- Intermediate Formation: This shift forms a non-aromatic intermediate, a substituted cyclohexa-2,5-dienone.
- Tautomerisation: The intermediate quickly tautomerises by losing a proton from the ortho position to regain its aromatic stability, resulting in the final product, o-allylphenol.
3. What are some important examples and real-world applications of the Claisen rearrangement?
A classic example is the thermal conversion of allyl phenyl ether to o-allylphenol, a fundamental reaction taught in CBSE Class 12 chemistry. A significant real-world application is found in biochemistry. The Claisen rearrangement is a crucial step in the shikimate pathway in plants and microorganisms, where chorismate is converted to prephenate. This prephenate is an essential precursor for synthesising vital amino acids like phenylalanine and tyrosine.
4. Why is the Claisen rearrangement classified as a [3,3]-sigmatropic shift?
It is classified as a [3,3]-sigmatropic shift because of how the bonds rearrange. If you number the atoms in the rearranging chain starting from the oxygen or vinyl group, the old sigma bond that breaks is between atoms numbered 1 and 1 (C-O bond). The new sigma bond that forms is between atoms numbered 3 and 3 (C-C bond). This simultaneous breaking of a bond at the [1,1] position and formation of a new bond at the [3,3] position is the defining characteristic of this type of pericyclic reaction.
5. Why is the Claisen rearrangement of allyl aryl ethers generally considered an irreversible reaction?
The rearrangement is largely irreversible due to a significant thermodynamic driving force. The starting material is an ether with a stable aromatic ring. The reaction proceeds through a non-aromatic intermediate, but the final product is a phenol. The formation of the highly stable phenolic hydroxyl group and the restoration of the aromatic ring makes the product much more stable than the reactant. This large release of energy upon aromatisation drives the equilibrium far to the product side, making the reverse reaction energetically unfavorable.
6. What happens during a Claisen rearrangement if both ortho positions on the benzene ring are already blocked by other groups?
If both ortho positions are blocked, the reaction does not simply fail. Instead, a para-rearrangement occurs. The allyl group first migrates to the ortho position to form the standard dienone intermediate. Since there is no hydrogen at this position to allow for tautomerisation, the molecule undergoes a second [3,3]-sigmatropic shift. This second shift moves the allyl group from the ortho to the para position. Finally, the resulting intermediate tautomerises to form the stable p-allylphenol as the final product.
7. How do different types of solvents influence the rate of the Claisen rearrangement?
The rate of a Claisen rearrangement is significantly influenced by the polarity of the solvent. While the reaction can occur without a solvent, using polar solvents like ethanol or water tends to accelerate the reaction. This is because the transition state of the rearrangement, although cyclic, is often more polar than the starting ether. Polar solvents stabilize this transition state, lowering the overall activation energy and thus increasing the reaction rate. Solvents capable of hydrogen bonding are particularly effective.
























