



How Does Oxidative Phosphorylation Work for NEET Exam Preparation?
Oxidative phosphorylation is a crucial biological process by which cells generate most of their energy in the form of ATP. Understanding oxidative phosphorylation is essential for NEET aspirants, as it forms a key part of cellular respiration and energy metabolism – topics that are frequently tested. This concept helps students build a strong foundation in biology and improves problem-solving for related NEET questions.
What is Oxidative Phosphorylation?
Oxidative phosphorylation is the final phase of cellular respiration, taking place in the mitochondria of eukaryotic cells. It refers to the process where ATP (adenosine triphosphate) is synthesized using energy released by the transfer of electrons from NADH and FADH2 to oxygen, through a series of protein complexes called the electron transport chain. This process is termed "oxidative" because it involves oxidation-reduction reactions, and "phosphorylation" because a phosphate group is added to ADP to form ATP.
Core Principles of Oxidative Phosphorylation
To understand oxidative phosphorylation in depth, it is important to break down the key ideas and events that make up this process.
1. Electron Transport Chain (ETC)
The electron transport chain is a series of protein complexes (Complex I, II, III, IV) embedded in the inner mitochondrial membrane. Electrons from NADH and FADH2 are transferred through these complexes, which release energy used to pump protons (H+) into the intermembrane space, creating a proton gradient.
2. Chemiosmosis and Proton Gradient
The movement of electrons down the chain creates a high concentration of protons in the intermembrane space compared to the mitochondrial matrix. This difference in proton concentration leads to an electrochemical gradient, also called the proton motive force.
3. ATP Synthase and ATP Generation
Protons flow back into the mitochondrial matrix through a protein channel called ATP synthase. As protons move down their concentration gradient, ATP synthase uses this energy to convert ADP into ATP. This coupling of proton movement with ATP synthesis is called chemiosmotic coupling.
4. Oxygen as the Final Electron Acceptor
Oxygen acts as the final electron acceptor in the chain, combining with electrons and protons to form water. Without oxygen, this chain stops, and ATP generation by oxidative phosphorylation ceases.
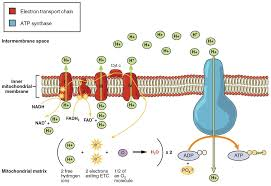
Key Sub-concepts Related to Oxidative Phosphorylation
Several important biological ideas are closely connected to oxidative phosphorylation. Understanding these helps clarify the main process.
Mitochondria - The Powerhouse of the Cell
Oxidative phosphorylation occurs specifically on the inner membrane of mitochondria. The structure of mitochondria, with its folds (cristae), increases the surface area for the electron transport chain and ATP synthase complexes, making energy production efficient.
NADH and FADH2 – Electron Donors
NADH and FADH2, generated during glycolysis, pyruvate oxidation, and the Krebs cycle, act as electron carriers. They transfer high-energy electrons to the electron transport chain, driving ATP synthesis.
Redox Reactions in the ETC
The protein complexes in the ETC undergo oxidation (loss of electrons) and reduction (gain of electrons) as electrons are passed along the chain. These redox reactions release energy needed to pump protons.
ATP Yield in Cellular Respiration
Most ATP generated by cells comes from oxidative phosphorylation. This process is much more efficient compared to glycolysis or fermentation. For every glucose molecule, oxidative phosphorylation is responsible for synthesizing the majority of the approximately 30-32 ATP molecules produced.
Formulas, Mechanisms, and Relationships in Oxidative Phosphorylation
While oxidative phosphorylation is largely a conceptual process, there are key relationships and summarized reactions that NEET students should know.
- Overall Reaction: 2NADH + 2H+ + O2 → 2NAD+ + 2H2O
- ATP Yield: Each NADH yields about 2.5 ATP; each FADH2 yields about 1.5 ATP (values are approximate).
- Chemiosmotic Theory: Proposed by Peter Mitchell, explains how the proton gradient drives ATP synthesis.
Features and Importance of Oxidative Phosphorylation
Oxidative phosphorylation is the most efficient way for aerobic cells to generate ATP. Here are the reasons for its biological significance:
- Provides the bulk of cellular energy in aerobic organisms.
- Couples oxidation of nutrients to ATP synthesis.
- Requires intact mitochondria and oxygen for efficiency.
- Disruption leads to various diseases and metabolic issues.
Why is Oxidative Phosphorylation Important for NEET?
Questions related to oxidative phosphorylation frequently appear in NEET, either directly or as part of broader cellular respiration topics. Understanding this concept supports:
- Problem-solving in questions on energy metabolism and respiration.
- Linking concepts like mitochondria structure and function, electron carriers, and ATP production.
- Grasping the mechanisms behind “energy currency” of the cell.
- Understanding connections to disorders and inhibitors of the electron transport chain in applied questions.
How to Study Oxidative Phosphorylation Effectively for NEET
To master oxidative phosphorylation for NEET, focus on both conceptual clarity and application. Here are some tips:
- Draw and label diagrams of mitochondria and the electron transport chain for better visualization.
- Understand each step of electron flow, proton gradient formation, and ATP synthesis.
- Memorize the sequence of complexes (I to IV) and the role of ATP synthase.
- Practice NEET MCQs on oxidative phosphorylation, mitochondrial function, inhibitors, and ATP yield.
- Revise the chemiosmotic theory and its significance regularly.
- Write summary notes and flowcharts for quick last-minute review.
- Watch animations or use reference images (like the one above) to reinforce learning.
Common Mistakes Students Make in Oxidative Phosphorylation
- Confusing the steps of the electron transport chain with those of the Krebs cycle or glycolysis.
- Misunderstanding the flow of electrons and protons - electrons pass through the ETC, protons are pumped into the intermembrane space.
- Incorrectly identifying ATP yield per NADH and FADH2.
- Forgetting the role of oxygen as the final electron acceptor.
- Overlooking the function of ATP synthase or the importance of the proton gradient.
Quick Revision Points: Oxidative Phosphorylation
- Takes place on the inner mitochondrial membrane.
- Involves electron transport chain and ATP synthase.
- Electron donors: NADH and FADH2.
- Proton gradient drives ATP synthesis.
- Oxygen is the final electron acceptor; forms water.
- Majority of cellular ATP is produced here.
- Disruption leads to energy deficiency in cells.
- Frequently asked in NEET under cell biology and energy metabolism.
FAQs on Oxidative Phosphorylation in NEET Biology
1. What is oxidative phosphorylation in biology?
Oxidative phosphorylation is the process by which cells produce ATP in the mitochondria using energy released from the transfer of electrons through the electron transport chain.
Main features:
- Takes place in the inner mitochondrial membrane
- Uses NADH and FADH2 as electron donors
- Involves the electron transport chain and ATP synthase
- Oxygen acts as the final electron acceptor
2. What is the role of electron transport chain in oxidative phosphorylation?
The electron transport chain (ETC) is responsible for transferring electrons from NADH and FADH2 to oxygen, producing water and creating a proton gradient needed for ATP synthesis.
Key points:
- Located in the inner mitochondrial membrane
- Consists of protein complexes I, II, III, IV
- Facilitates proton pumping across the membrane
- Essential for NEET biology, as it is central to energy conversion
3. Where does oxidative phosphorylation take place in the cell?
Oxidative phosphorylation occurs in the inner membrane of mitochondria in eukaryotic cells.
Location details:
- Electron transport chain is embedded in the inner mitochondrial membrane
- ATP synthase operates here to generate ATP
- Relevant for NEET exam questions on cell structure and function
4. How is ATP produced during oxidative phosphorylation?
ATP is produced during oxidative phosphorylation by the enzyme ATP synthase using the energy from a proton gradient created by the electron transport chain.
Steps involved:
- Electrons pass through ETC releasing energy
- Energy pumps protons into the intermembrane space
- Proton gradient drives protons back via ATP synthase
- ATP synthase converts ADP + Pi to ATP
5. Why is oxygen important in oxidative phosphorylation?
Oxygen acts as the final electron acceptor in the electron transport chain, making oxidative phosphorylation possible.
Main roles:
- Accepts electrons and combines with protons to form water
- Maintains electron flow through the chain
- Absence of oxygen stops ATP production and energy generation
6. What are the main steps of oxidative phosphorylation?
Oxidative phosphorylation involves two main stages: electron transport and chemiosmosis.
Main steps:
- Electron transport chain passes electrons from NADH/FADH2 to O2
- Protons are pumped into the intermembrane space
- A proton gradient forms across the inner membrane
- ATP synthase utilizes this gradient to synthesize ATP from ADP
7. What is the difference between oxidative phosphorylation and substrate-level phosphorylation?
The main difference is in the source of phosphate and energy used to form ATP.
Comparison:
- Oxidative phosphorylation: ATP formed using energy from electron transport and a proton gradient
- Substrate-level phosphorylation: ATP formed directly in metabolic reactions by transfer of phosphate group from a substrate to ADP
- Oxidative phosphorylation occurs in mitochondria, while substrate-level occurs in glycolysis and Krebs cycle
8. How many ATP are produced during oxidative phosphorylation?
Oxidative phosphorylation typically produces about 26 to 28 ATP molecules per molecule of glucose.
ATP production summary:
- From NADH: 2.5 ATP per molecule
- From FADH2: 1.5 ATP per molecule
- Total ATP from oxidative phosphorylation: 26–28
9. What enzymes are involved in oxidative phosphorylation?
Major enzymes in oxidative phosphorylation include the protein complexes of the electron transport chain and ATP synthase.
Key enzymes:
- NADH dehydrogenase (Complex I)
- Succinate dehydrogenase (Complex II)
- Cytochrome bc1 complex (Complex III)
- Cytochrome c oxidase (Complex IV)
- ATP synthase
10. What happens if oxidative phosphorylation is inhibited?
If oxidative phosphorylation is inhibited, cells cannot produce enough ATP, leading to energy failure and potential cell death.
Consequences:
- Build-up of NADH and FADH2
- Lack of ATP for vital cell functions
- Possible accumulation of lactic acid due to anaerobic pathways
- Relevant for understanding metabolic disorders in NEET
11. What are the products of oxidative phosphorylation?
The main products of oxidative phosphorylation are ATP, water (H2O), and reoxidized carriers (NAD+ and FAD).
Summary:
- ATP for cellular energy
- Water formed as oxygen accepts electrons
- Oxidized forms of electron carriers (NAD+, FAD)























