What is the Urea Cycle Steps Enzymes and Function
The principal metabolic mechanism involved in the elimination of nitrogenous wastes generated by the breakdown of protein and other nitrogen-containing molecules is called the urea cycle. In the mitochondria of liver cells, the urea cycle converts excess ammonia to urea. Urea is formed, enters the bloodstream, is filtered by the kidneys, and is eventually excreted in the urine.
The biochemical part of excretion is urea synthesis using the urea cycle, also known as the Ornithine Cycle. It is also known as the Kreb-Henseleit cycle and occurs in the liver. In this article, we will see the regulation and significance of this cycle.
What is Urea Cycle?
The urea cycle is a series of biochemical reactions that results in the formation of urea CO(NH2)2 from ammonia (NH3).
Ureotelic animals are amphibians and mammals that use this cycle.
The urea cycle converts highly toxic ammonia to urea, which is then excreted. This cycle was discovered five years before the TCA cycle by Hans Krebs and Kurt Henseleit (Hans Krebs and Kurt Henseleit, 1932). Ratner and Cohen went into greater detail about this cycle later on.
The urea cycle is primarily carried out in the liver and, to a lesser extent, in the kidneys.
The urea cycle is irreversible and requires 4 ATP to complete.
Only the liver has all of the enzymes needed to generate urea from ammonia, and this route is only located in periportal hepatocytes.
The urea cycle is made up of four different enzyme processes, one mitochondrial and three cytosolic. Carbamoyl phosphate synthetase (CPS), ornithine carbamoyltransferase (OCT), argininosuccinate synthetase, argininosuccinate lyase, and arginase are all implicated.
The urea cycle enzymes ornithine carbamoyltransferase and arginase are also found in the mitochondria, whereas the cytoplasm contains argininosuccinate synthetase and argininosuccinate lyase.
Urea Cycle Biochemistry and Steps Involved
Carbamoyl Phosphate Synthesis
The mitochondrial carbamoyl phosphate synthetase I (CPS I) catalyses the condensation of NH4+ ions with CO2 to generate carbamoyl phosphate. This process is irreversible and rate-limiting, consuming 2 ATP. CPS I requires N-acetyl glutamate to function. Another enzyme involved in pyrimidine production, carbamoyl phosphate synthetase II (CPS-II), is found in the cytosol. It accepts the amino group from glutamine and does not need N-acetyl glutamate to function.
Citrulline Formation
Citrulline is produced by ornithine transcarbamoylase from carbamoyl phosphate and ornithine. Ornithine is recycled and utilised in the urea cycle.
Argininosuccinate Synthesis
Argininosuccinate synthase combines with citrulline and aspartate to generate argininosuccinate. This process incorporates the second amino group of urea. This process requires the breakdown of ATP into AMP and pyrophosphate (PPi). The PPi is instantly degraded to inorganic phosphate (Pi).
Argininosuccinate Cleavage
Arginosuccinase cleaves argininosuccinate to produce arginine and fumarate. Urea is a direct precursor of arginine.
Urea Formation
Arginase is the last enzyme that cleaves arginine to produce urea and ornithine. The regenerated ornithine enters the mitochondria for reuse in the urea cycle. Both CO2 and Mn2+ are activated by arginase.
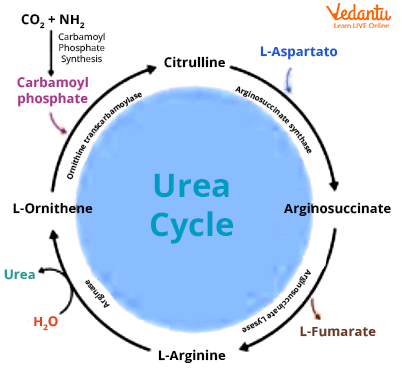
Urea Cycle
Regulation of Urea Cycle
The first reaction, catalysed by Carbamoyl Phosphate Synthetase I (CPS I), is a rate-limiting or essential step in the production of urea. N-acetyl glutamate (NAG) activates CPS I allosterically. The rate of urea production in the liver is linked to N-acetyl-glutamate concentration. NAG levels rise when arginine levels rise. A protein-rich meal raises the amount of NAG in the liver, resulting in increased urea production.
The mitochondria include carbamoyl phosphate synthetase I and glutamate dehydrogenase. They collaborate in the creation of NH3 and its usage in the synthesis of carbamoyl phosphate. The remaining four urea cycle enzymes are primarily regulated by the concentration of their respective substrates.
Overall Reaction and Energetics
The urea cycle is irreversible and requires 4 ATP to complete. The production of carbamoyl phosphate requires two ATPs. One ATP is converted to AMP and PPi to yield argininosuccinate, which is equivalent to two ATP. As a result, 4 ATP is actually consumed.
NH4+ + CO2 + Aspartate + 3ATP → Urea+ Fumarate + 2ADP + 2 Pi + AMP + PPi
Significance of Urea Cycle
The urea cycle, also known as the “Ammonia Detox Cycle”, is the process by which ammonia is removed from the body.
Toxic ammonia is transformed into harmless urea. It eliminates two waste products: ammonia and CO2.
It produces arginine, a semi-essential amino acid. It helps to regulate blood pH, which is determined by the dissolved CO2 ratio, e.g. H2CO3 to HCO3.
Ornithine is a precursor of polyamines such as spermidine and spermine.
Ammonia is a byproduct of protein metabolism that is hazardous to the human body, particularly the central nervous system. As a result, ammonia is transformed into urea, a harmless water-soluble molecule that is excreted through urine.
Importance of Urea Cycle
NH4+ is a byproduct of amino acid degradation.
Cells require NH4+ for the production of nitrogen-containing molecules.
Excess NH4+ is extremely harmful to our body. So, the excess NH4+ is transformed into urea and eliminated via the urea cycle. The urea cycle accounts for 80% of the nitrogen excreted.
TCA cycle intermediates are recycled.
Amino acids and keto acids are recycled.
Urea Cycle Disorder
The table describes metabolic disorders related to each of the five urea cycle enzymes. All of the disorders always result in an increase in blood ammonia (hyperammonemia), which leads to toxicity. Another urea cycle byproducts accumulate as well, depending on the individual enzyme deficiency. Clinical signs of urea cycle enzyme deficiencies include vomiting, drowsiness, irritability, stiffness, and mental retardation.
Conclusion
Urea cycle is a series of metabolic reactions that occur in the liver to convert ammonia to urea. The urea cycle is made up of six enzymes that rid the body of nitrogen produced during amino acid metabolism. They break it down into urea, which is excreted in the urine. This article talks about the importance and regulation of the Urea Cycle. There are different disorders also related to the improper functioning of the enzymes of the urea cycle.


FAQs on Urea Cycle in Human Physiology
1. What is the urea cycle?
The urea cycle is a metabolic pathway in the liver that converts toxic ammonia into urea for safe excretion in urine. It is also called the ornithine cycle and is a key process in nitrogen metabolism. It:
- Occurs mainly in hepatocytes (liver cells)
- Converts ammonia (NH₃) into urea
- Prevents toxic buildup of nitrogenous waste in the blood
2. Where does the urea cycle occur in the cell?
The urea cycle occurs partly in the mitochondria and partly in the cytoplasm of liver cells. Specifically:
- The first two steps occur in the mitochondrial matrix
- The remaining steps occur in the cytosol
3. What are the steps of the urea cycle?
The urea cycle steps convert ammonia and carbon dioxide into urea through five enzyme-catalyzed reactions. The steps are:
- Formation of carbamoyl phosphate from ammonia and CO₂ by carbamoyl phosphate synthetase I
- Formation of citrulline from carbamoyl phosphate and ornithine
- Formation of argininosuccinate from citrulline and aspartate
- Cleavage into arginine and fumarate
- Conversion of arginine into urea and regeneration of ornithine
4. What is the function of the urea cycle?
The main function of the urea cycle is to detoxify ammonia by converting it into urea for excretion. Ammonia is produced during amino acid metabolism and is highly toxic, especially to the brain. The cycle:
- Prevents hyperammonemia
- Maintains nitrogen balance
- Allows safe elimination of nitrogen in urine
5. Why is ammonia toxic to the body?
Ammonia is toxic because it disrupts brain function and alters cellular metabolism. High levels of ammonia (NH₃) can:
- Interfere with the central nervous system
- Alter pH balance in cells
- Lead to confusion, coma, or brain swelling
6. What are the enzymes involved in the urea cycle?
The urea cycle enzymes are five key enzymes that catalyze each step of the pathway. They include:
- Carbamoyl phosphate synthetase I (CPS I)
- Ornithine transcarbamylase (OTC)
- Argininosuccinate synthetase
- Argininosuccinate lyase
- Arginase
7. What is the difference between the urea cycle and the Krebs cycle?
The urea cycle removes excess nitrogen, while the Krebs cycle (citric acid cycle) produces energy through oxidation of acetyl-CoA. Key differences include:
- Urea cycle occurs mainly in the liver; Krebs cycle occurs in all aerobic cells
- Urea cycle detoxifies ammonia; Krebs cycle generates ATP
- Urea cycle produces urea; Krebs cycle produces CO₂ and reducing equivalents
8. What are urea cycle disorders?
Urea cycle disorders are genetic conditions caused by deficiency of one of the urea cycle enzymes, leading to ammonia accumulation. These inherited metabolic disorders can result in:
- Hyperammonemia
- Vomiting and lethargy in infants
- Neurological damage if untreated
9. How is the urea cycle regulated?
The urea cycle is regulated mainly by substrate availability and activation of carbamoyl phosphate synthetase I. Regulation occurs through:
- Allosteric activation of CPS I by N-acetylglutamate (NAG)
- Increased enzyme expression during high-protein diets or fasting
- Availability of ammonia and aspartate
10. What is the overall reaction of the urea cycle?
The overall reaction of the urea cycle converts ammonia, carbon dioxide, and aspartate into urea using ATP. In summary:
- 2 nitrogen atoms enter as ammonia and aspartate
- 1 carbon atom enters as CO₂
- Energy is supplied by ATP
- Final product is urea, excreted in urine










