Principle Procedure And Rf Value In Paper Chromatography Of Plant Pigments
A compound that absorbs light is called a pigment. Chlorophylls a and b are primary photosynthetic pigments that absorb light for photosynthesis. The accessory pigments carotenoids and xanthophyll absorb light and pass it to chlorophyll a. Even though chlorophyll is the primary pigment, the other pigments are essential to the plant's ability to produce colour and engage in photosynthesis because they absorb each light differently and effectively across the electromagnetic spectrum.
Paper Chromatography
Chromatography, which means "colour writing," is a Greek term that is formed from the words "chromo" and "graph". Chromatography enables the separation of the constituent parts of a given mixture, enabling scientists to observe and produce findings and theories.
Paper chromatography is a method for classifying dissolved substances according to how soluble they are in a given solvent, such as chlorophyll, carotene, and xanthophyll. Paper chromatography can be used to separate the colours in plant cells. The stationary element in chromatography paper permits the reaction between the solute and solvent to take place and produce results.
Leaf Chromatography
The separation of leaf colours using chromatography is known as leaf chromatography. Leaf chromatography is an experiment that is conducted to determine the colour of the photosynthetic pigments.
The experiment is conducted to learn about the pigments in the leaf, and it is mostly done by using paper and thin-layer chromatography. Let’s discuss some brief points of leaf chromatography.
There is a procedure by which this experiment is conducted in labs.
Sample leaves should be crushed into small pieces and put in a mortar for pestle grinding. Add solvent and keep using the pestle to crush.
Then, carefully draw a pencil line 1 cm from the bottom of the chromatography paper, spot a little amount of leaf extract repeatedly onto the centre of the line, and let each spot dry.
Make sure the paper dips into the solvent but the spot of leaf extract doesn't by suspending it using a pin attached to a bung within a test tube with a 1 cm depth of solvent.
The solvent is allowed to run up the paper until it is close to the bung, at which point the paper is removed. The solvent's location is marked, and the paper is allowed to dry.
The final chromatography paper is known as a chromatogram, and it may be photographed to determine the exact position of each pigment. Next, determine the Rf value for each pigment spot on the chromatogram.
The retention factor is pronounced Rf. The retention factor is calculated by dividing the component's travel distance by the solvent's travel distance.
The colour dissolves as the alcohol goes through the filter paper. Some pigments in the leaf travel more quickly than others because of their properties.
The pigment's movement rate is measured by the Rf (retention factor) value. Rf value = distance transported by pigment from origin to centre of pigment spot/distance from the origin to the solvent front. By applying this formula, you can determine the Rf value.
Separation of Plant Pigments by Paper Chromatography
The pigments in the plant's leaf are separated by paper chromatography, i.e., separation chromatography. It is the same as a leaf chromatography experiment. The process of paper chromatography is also the same as the leaf chromatography experiment.
Separation of Chlorophyll Pigments by Paper Chromatography
The chlorophyll molecule is present in the leaf and can be separated by using paper chromatography. The paper chromatography separates the pigments in the leaf based on the distance travelled by pigment molecules on the paper in a nonpolar solvent.
Separation of Plant Pigments by Paper Chromatography Diagram
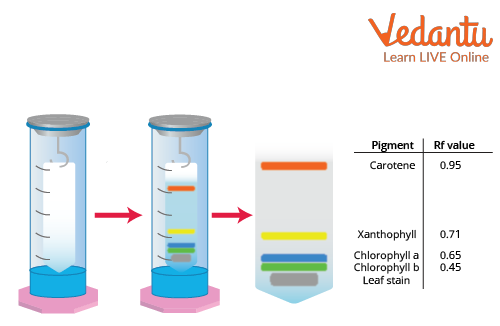
The Experimental Setup of Paper Chromatography
Chromatogram Report
The final chromatography paper is known as a chromatogram, and it may be photographed to determine the exact position of each pigment. The pattern of pigment spots on the chromatography paper at the conclusion of the experiment is called a chromatogram. Along with the alcohol, the pigments also migrate along the strips of paper.
Chromatography Conclusion
Carotene is identified as having the lowest molecular weight by its yellow to orange tint near the top of the paper. In the pigment separation of chlorophyll, chlorophyll may be distinguished by its blue or dark green hue. When chlorophyll pigments are separated, the colour yellow-light green identifies chlorophyll B. In the chromatography solvent, xanthophyll is more soluble since it has gone up the paper. This describes the conclusion of paper chromatography.
Conclusion
The pigments are light-absorbing molecules and are separated by using paper chromatography techniques in the lab. The pigments move on the paper based on their solubility in the solvent. Along with the alcohol, pigments also migrate along the strips of paper. Some pigments in the leaf travel more quickly than others because of their properties.


FAQs on Separation Of Plant Pigments Using Paper Chromatography Technique
1. What is separation of plant pigments through paper chromatography?
The separation of plant pigments through paper chromatography is a laboratory technique used to separate different pigments present in plant leaves based on their solubility and affinity to the paper. It works because:
- Plant pigments dissolve in a suitable solvent.
- Different pigments travel at different speeds on the chromatography paper.
- This difference causes them to separate into distinct colored bands.
It is commonly used to separate pigments such as chlorophyll a, chlorophyll b, carotene, and xanthophyll.
2. What are the main plant pigments separated by paper chromatography?
The main plant pigments separated by paper chromatography are chlorophyll a, chlorophyll b, carotene, and xanthophyll. These pigments differ in color and polarity:
- Chlorophyll a – blue-green pigment
- Chlorophyll b – yellow-green pigment
- Carotene – orange pigment
- Xanthophyll – yellow pigment
Each pigment plays a role in photosynthesis by absorbing different wavelengths of light.
3. How does paper chromatography separate plant pigments?
Paper chromatography separates plant pigments based on differences in solubility and adsorption between the solvent and the paper. The process occurs as follows:
- The paper acts as the stationary phase.
- The solvent acts as the mobile phase.
- Pigments that are more soluble in the solvent travel farther.
- Pigments that adhere more strongly to paper travel shorter distances.
This difference in movement results in distinct colored bands on the chromatogram.
4. What is the principle of paper chromatography in plant pigment separation?
The principle of paper chromatography is based on partition chromatography, where components separate due to different distribution between the stationary and mobile phases. In plant pigment separation:
- The stationary phase is water trapped in cellulose fibers of paper.
- The mobile phase is an organic solvent.
- Pigments distribute differently between these two phases.
This unequal distribution leads to separation of pigments according to polarity.
5. What is the Rf value in paper chromatography?
The Rf value (Retention factor) is the ratio of the distance traveled by a pigment to the distance traveled by the solvent front. It is calculated as:
- Rf = Distance moved by pigment / Distance moved by solvent front
Rf values help identify specific plant pigments because each pigment has a characteristic Rf value under fixed conditions.
6. Why do different plant pigments travel different distances on chromatography paper?
Different plant pigments travel different distances because they differ in polarity and solubility in the solvent. Specifically:
- Non-polar pigments like carotene dissolve better in non-polar solvents and move farther.
- More polar pigments like chlorophyll b interact more with the paper and move less.
This difference in chemical properties causes visible separation on the chromatogram.
7. What is the role of solvent in paper chromatography of plant pigments?
The solvent in paper chromatography acts as the mobile phase that carries plant pigments up the paper. Its functions include:
- Dissolving the extracted leaf pigments.
- Transporting pigments by capillary action.
- Allowing separation based on pigment solubility.
Common solvents include mixtures of petroleum ether, acetone, or alcohol.
8. What are the steps involved in separating plant pigments by paper chromatography?
The steps in separating plant pigments by paper chromatography involve extraction, spotting, and solvent development. The procedure includes:
- Crushing green leaves to extract pigments using a solvent.
- Applying the extract as a small spot on chromatography paper.
- Placing the paper in a solvent without submerging the spot.
- Allowing the solvent to rise and separate the pigments.
- Marking the solvent front and observing colored bands.
The final chromatogram shows distinct pigment layers.
9. What is the importance of separating plant pigments?
Separating plant pigments is important to understand the different components involved in photosynthesis. It helps to:
- Identify various photosynthetic pigments.
- Study pigment composition in different plants.
- Analyze plant health and physiological conditions.
This technique is widely used in biology practicals and plant physiology research.
10. What is the difference between chlorophyll a and chlorophyll b in chromatography?
The main difference between chlorophyll a and chlorophyll b in chromatography is their color and Rf value due to polarity differences. Specifically:
- Chlorophyll a is blue-green and usually travels farther.
- Chlorophyll b is yellow-green and travels a shorter distance.
- Chlorophyll b is slightly more polar than chlorophyll a.
These differences allow them to form separate bands on the chromatogram.










