Types of Plastids and Their Functions in Plants
Imagine a bustling factory within every plant cell where specialised workers convert sunlight into energy and store vital nutrients. Plastids, the double-membrane organelles found in plant cells, serve as these essential factories. From photosynthesis in chloroplasts to pigment production in chromoplasts, plastids play diverse roles that keep plants thriving. In this engaging guide, we explore what are plastids, their structure, various types, and their functions in plant cells. With surprising facts and interactive elements, you'll gain a deeper understanding of these fascinating organelles and their impact on both plant biology and everyday life.
Understanding Plastids: Definition and Function
Plastids definition: Plastids are specialised, double-membrane organelles in plant cells responsible for manufacturing and storing food, pigments, and other vital compounds. They are crucial for processes like photosynthesis and nutrient storage, making them indispensable for plant survival.
What is plastids?
Plastids are organelles that contribute to key cellular functions, such as converting sunlight to energy (in chloroplasts) and storing compounds (in leucoplasts).
Function of plastids:
Photosynthesis: Chloroplasts capture light energy and transform it into chemical energy.
Pigment Storage: Chromoplasts store pigments that lend leaves, flowers, and fruits their vibrant colours.
Nutrient Storage: Leucoplasts store starches, proteins, and lipids, ensuring plants have the energy and building blocks they need.
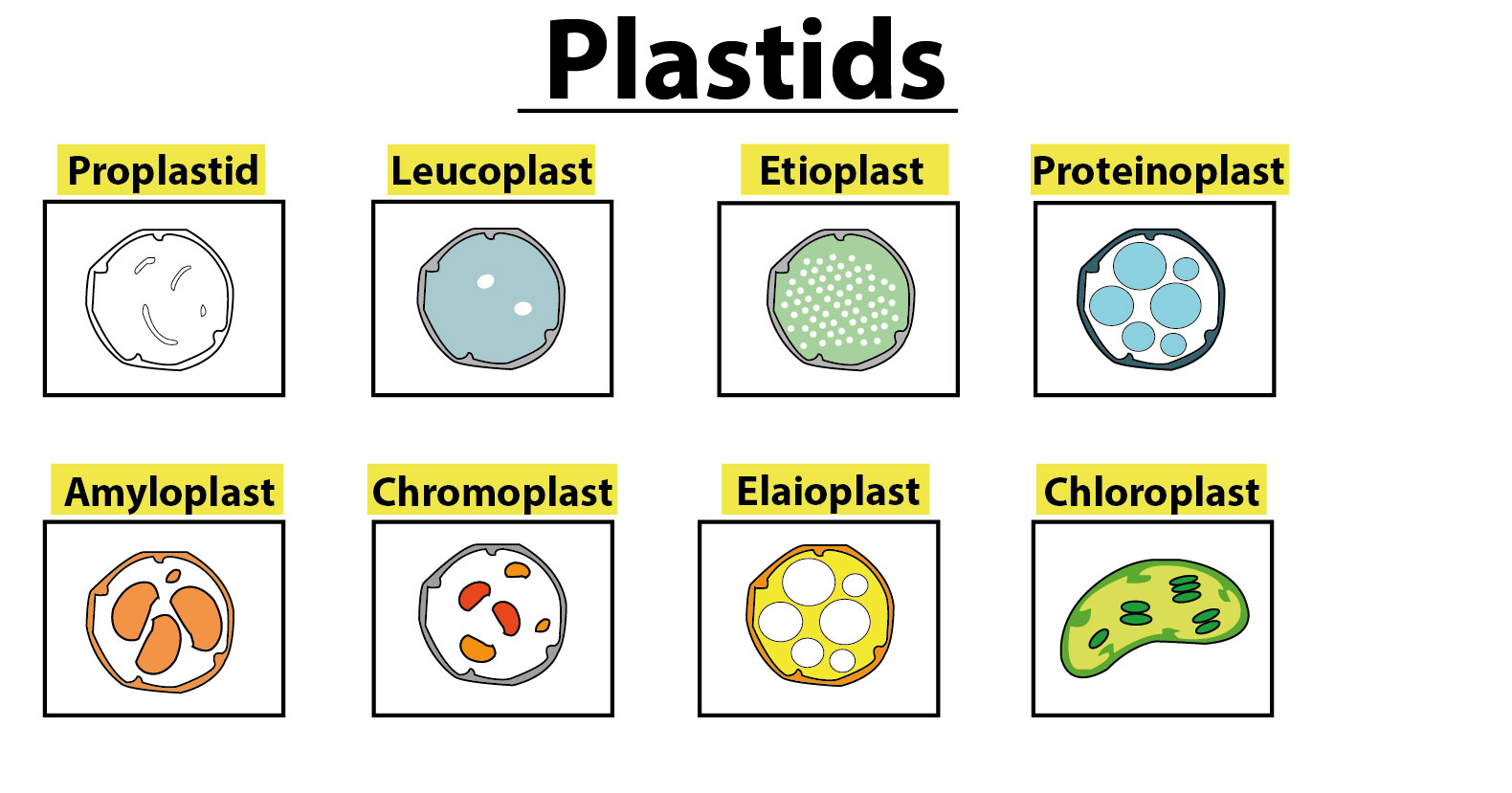
Types of Plastids and Their Specialisations
Types of plastids vary based on the presence of pigments and their developmental stages. The main types include:
Chloroplasts
Structure of plastids: Chloroplasts are biconvex, semi-porous, double-membrane organelles located mainly in the mesophyll of plant cells.
Role: They are the primary sites for photosynthesis, converting light energy into sugars.
Chromoplasts
Plastids in plant cells: Chromoplasts, found in flowers, ageing leaves, and fruits, store carotenoid pigments.
Significance: They provide the bright colours necessary for attracting pollinators and enhancing visual appeal.
Gerontoplasts
Transition Role: Gerontoplasts are ageing chloroplasts that transition as the leaf's photosynthetic activity declines, especially during autumn.
Leucoplasts
Non-Pigmented Organelles: These are colourless plastids found in non-photosynthetic tissues like roots.
Subtypes:
Amyloplasts: Specialised in storing and synthesising starch.
Proteinoplasts: Involved in the storage of proteins, often found in seeds.
Elaioplasts: Dedicated to the storage of fats and oils.
Students can also read out detailed article on the Difference between mitochondria and plastids to understand the concept better.
- Inheritance and the Uniparental Trait
Plastids are inherited uniparentally. In angiosperms, plastids are passed on through the female gamete, while in many gymnosperms, the male pollen is the source. This unique mode of inheritance underscores the critical role of plastids in plant development and evolution.
Test your Knowledge on Plastids!
Take this short quiz and then “Check your answers” to see how well you understand plastids.
Question: What are plastids primarily responsible for in plant cells?
Question: Name the type of plastid that is essential for photosynthesis.
Question: Which plastid is known for storing pigments that give fruits and flowers their colour?
Question: What is the function of leucoplasts in non-photosynthetic parts of the plant?
Question: How are plastids generally inherited in angiosperms?
Check Your Answers:
Manufacturing and storing food, pigments, and other compounds.
Chloroplasts.
Chromoplasts.
They store starch, proteins, or lipids.
Through the female gamete (uniparental inheritance).
Fun Facts about Plastids
Fact 1: Despite being small, plastids can transform into different forms (e.g., chloroplasts converting into chromoplasts) as plants mature.
Fact 2: Plastids contain their own DNA, highlighting their evolutionary history as once free-living organisms.
Fact 3: The vibrant colours of autumn leaves are partly due to the transformation of chloroplasts into gerontoplasts.
Real-World Applications
Plastids are not just academic curiosities—they have real-world significance:
Agriculture: Understanding plastids helps in developing crops with enhanced nutritional qualities and improved stress resistance.
Biotechnology: Manipulating plastid functions is a key strategy in producing biofuels and biopharmaceuticals.
Horticulture: The role of chromoplasts in colour production is exploited to breed visually appealing fruits and ornamental plants.


FAQs on Plastids in Plant Cells: Types, Structure, and Key Roles
1. What are plastids and why are they essential for a plant cell?
Plastids are double-membrane-bound organelles found in the cells of plants and algae. They are essential because they perform critical functions like photosynthesis (converting light energy into chemical energy), storage of food reserves like starch and lipids, and synthesis of pigments that give colour to flowers and fruits, aiding in pollination.
2. What are the three main types of plastids based on their function?
The three main types of plastids, classified by their pigments and functions, are:
- Chloroplasts: Contain chlorophyll and carotenoids; they are the primary sites of photosynthesis.
- Chromoplasts: Contain fat-soluble carotenoid pigments like carotene and xanthophylls, giving yellow, orange, or red colours to parts of the plant like fruits and flowers.
- Leucoplasts: These are colourless plastids that store nutrients. They include amyloplasts (store starch), elaioplasts (store oils and fats), and proteinoplasts (store proteins).
3. What are the key structural components of a chloroplast?
A chloroplast is enclosed by a double membrane. The inner fluid-filled space, called the stroma, contains numerous membrane-bound flattened sacs called thylakoids. Thylakoids are arranged in stacks, known as grana. The stroma also contains its own small, circular DNA, ribosomes, and the enzymes required for synthesising carbohydrates during photosynthesis.
4. How can one type of plastid transform into another? Give an example.
Plastids are developmentally related and can change from one form to another based on the plant's needs and environmental cues. For example, when a raw green tomato ripens, its chloroplasts (rich in chlorophyll) differentiate into red chromoplasts by breaking down chlorophyll and synthesising large amounts of the red pigment lycopene. This changes the fruit's colour from green to red.
5. What is the significance of plastids having their own DNA and ribosomes?
The presence of their own circular DNA and 70S ribosomes is highly significant as it supports the endosymbiotic theory. This theory suggests that plastids originated from free-living photosynthetic prokaryotes that were engulfed by an early eukaryotic cell. This genetic material allows plastids to synthesise some of their own proteins and replicate independently of the cell's nucleus, making them semi-autonomous organelles.
6. How do chromoplasts and chloroplasts differ in their primary role in a plant?
The primary difference lies in their function. Chloroplasts are responsible for photosynthesis; their main role is to capture light energy and convert it into chemical energy in the form of glucose. In contrast, chromoplasts are non-photosynthetic and are primarily involved in synthesising and storing pigments. Their key role is to provide colour to flowers and fruits, which helps in attracting pollinators and animals for seed dispersal.
7. Are all plastids coloured? Explain why or why not.
No, not all plastids are coloured. Leucoplasts are a type of plastid that are colourless because they lack pigments. Their primary function is not photosynthesis or colouration, but the storage of food materials. For instance, amyloplasts in potato tubers are leucoplasts that store large amounts of starch, a colourless carbohydrate.










