



What is the Nitrogen Cycle and its Importance in the Ecosystem?
The nitrogen cycle is a vital biogeochemical cycle that describes how nitrogen moves between the atmosphere, soil, water, and living organisms. It ensures the continuous availability of nitrogen in usable forms for plants and animals.
“The nitrogen cycle is defined as the circulation and transformation of nitrogen in different chemical forms through the ecosystem.”
Nitrogen is one of the most essential elements for life because it is a key component of:
Proteins
DNA and RNA
Chlorophyll
Although nitrogen gas (N₂) makes up nearly 78–80% of the atmosphere, most organisms cannot directly use it. Therefore, nitrogen must be converted into usable forms like ammonia, nitrites, and nitrates through various biological and chemical processes.
It involves several processes that convert nitrogen into different forms so that it can be:
Absorbed by plants
Used by animals
Returned to the atmosphere
This continuous transformation is essential for maintaining ecosystem balance and productivity.
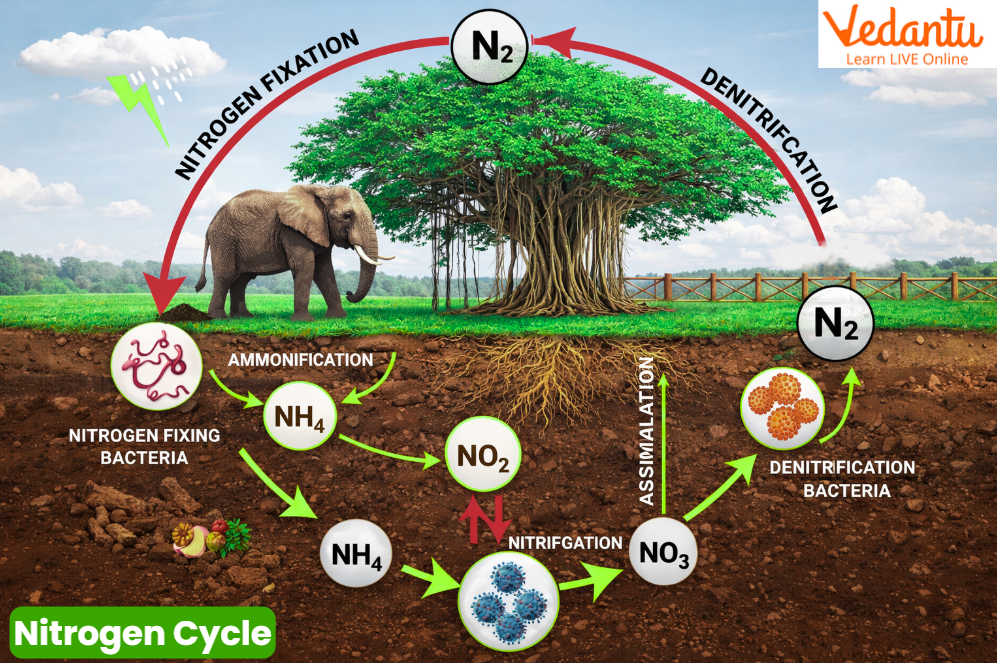
Nitrogen Cycle Steps
The nitrogen cycle steps include five major processes:
Nitrogen Fixation
Nitrification
Assimilation
Ammonification
Denitrification
(+ Anammox as an additional pathway)
Each step is mainly carried out by microorganisms and is essential for maintaining nitrogen balance.
1. Nitrogen Fixation
Nitrogen fixation is the process of converting atmospheric nitrogen (N₂) into ammonia (NH₃), which can be used by plants.
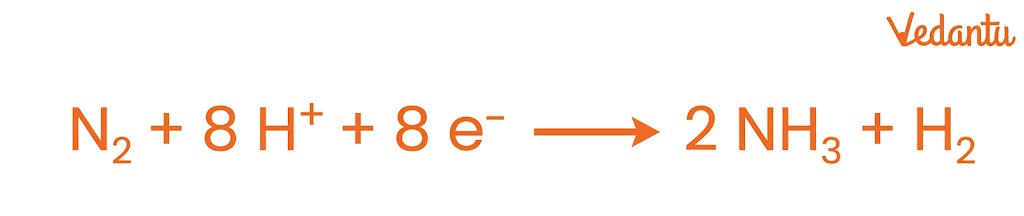
Key Points:
Requires high energy (ATP) due to strong triple bond in N₂
Carried out mainly by bacteria like Rhizobium
Occurs in root nodules of leguminous plants
Enzyme involved: Nitrogenase
Types:
Biological fixation (by microbes)
Abiotic fixation (lightning)
Industrial fixation (Haber process)
Only after fixation does nitrogen become biologically available.
2. Nitrification
Nitrification is the conversion of ammonia into nitrites and then nitrates.
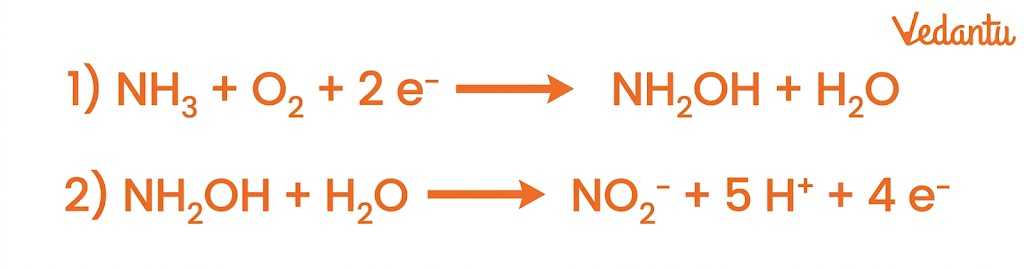
Steps:
Ammonia → Nitrite (NO₂⁻) by ammonia-oxidizing bacteria
Nitrite → Nitrate (NO₃⁻) by nitrite-oxidizing bacteria
Important Features:
Occurs under aerobic conditions
Carried out by bacteria such as Nitrosomonas and Nitrobacter
Produces a small amount of energy
Nitrates formed are easily absorbed by plants.
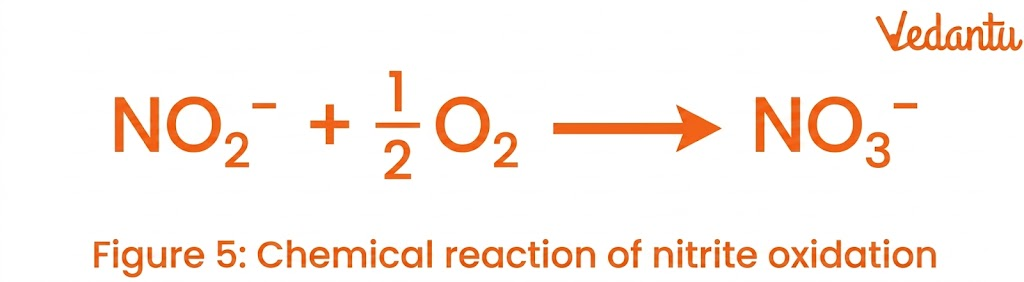
3. Assimilation (Assimilation Nitrogen Cycle)
Assimilation in the nitrogen cycle refers to the uptake of nitrates or ammonia by plants.
Process:
Plants absorb nitrates from soil
Convert them into amino acids and proteins
Animals obtain nitrogen by consuming plants
This step transfers nitrogen into the food chain and is essential for growth.
4. Ammonification
Ammonification is the conversion of organic nitrogen into ammonia.
Occurs When:
Plants and animals excrete waste
Dead organisms decompose
Key Agents:
Bacteria and fungi
Importance:
Returns nitrogen back to soil
Makes ammonia available again for nitrification
5. Denitrification
Denitrification is the process of converting nitrates back into nitrogen gas.
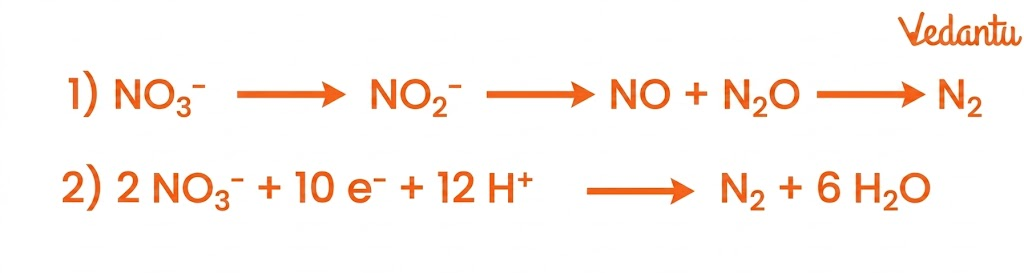
Key Features:
Occurs under anaerobic conditions
Carried out by bacteria like Pseudomonas
Produces gases like N₂ and N₂O
Importance:
Returns nitrogen to atmosphere
Maintains nitrogen balance
Nitrous oxide released is a greenhouse gas and contributes to pollution.
6. Anammox (Anaerobic Ammonia Oxidation)
Anammox is a special process where:
Ammonia reacts with nitrite
Produces nitrogen gas directly
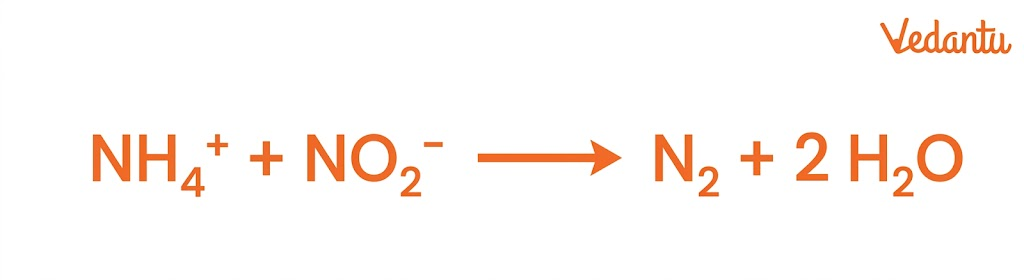
Key Points:
Occurs in anaerobic environments
Performed by specific bacteria
Plays a role in nitrogen loss in oceans
This is an important addition to the traditional nitrogen cycle.
Importance of Nitrogen Cycle
The importance of the nitrogen cycle lies in maintaining life on Earth.
Supplies usable nitrogen to plants
Maintains soil fertility
Supports food chain
Regulates ecosystem productivity
Helps in protein and DNA synthesis
Without the nitrogen cycle, life processes would stop due to lack of usable nitrogen.
Nitrogen Cycle in Ecosystem
The nitrogen cycle in ecosystem plays a central role in:
Terrestrial Ecosystems:
Controls plant growth
Maintains soil nutrients
Influences biodiversity
Aquatic Ecosystems:
Regulates nutrient balance
Prevents eutrophication (when balanced)
Supports aquatic life
Nitrogen availability often limits primary productivity in ecosystems.
Human Impact on Nitrogen Cycle
Human activities have significantly altered the nitrogen cycle.
Major Activities:
Use of nitrogen fertilizers
Burning fossil fuels
Industrial nitrogen fixation (Haber-Bosch process)
Effects:
Increased nitrogen levels in ecosystems
Soil nutrient imbalance
Water pollution (nitrate leaching)
Algal blooms and oxygen depletion
Loss of biodiversity
Increased greenhouse gases
Human activity has doubled global nitrogen fixation, causing major ecological changes.
Environmental Consequences
Excess nitrogen leads to:
Eutrophication in water bodies
Hypoxia (low oxygen conditions)
Harmful algal blooms
Acidification of freshwater systems
Changes in food web structure
It can also increase the risk of diseases in humans and wildlife.
FAQs on Nitrogen Cycle: Steps, Diagram, Processes and Human Impact Explained
1. What are the 5 stages of the nitrogen cycle?
The nitrogen cycle steps are:
Nitrogen fixation
Nitrification
Assimilation
Ammonification
Denitrification
These five stages together make nitrogen available to living organisms and then return it to the atmosphere.
2. What is the nitrogen cycle and explain it?
The nitrogen cycle is the natural process through which nitrogen moves between the atmosphere, soil, water, plants, animals, and microorganisms. Since most organisms cannot use atmospheric nitrogen directly, it must be converted into usable forms like ammonia, nitrites, and nitrates. Through fixation, nitrification, assimilation in nitrogen cycle, ammonification, and denitrification, nitrogen is continuously recycled in the ecosystem.
3. What are the 7 parts of the nitrogen cycle?
The seven parts often included in the nitrogen cycle are:
Nitrogen fixation
Nitrification
Assimilation
Ammonification
Denitrification
Anammox
Dissimilatory nitrate reduction to ammonium
For school and NEET-level study, the main focus is usually on the first five major nitrogen cycle steps.
4. Is the nitrogen cycle a gaseous cycle?
Yes, the nitrogen cycle is considered a gaseous biogeochemical cycle because its main reservoir is the atmosphere, where nitrogen is present as nitrogen gas (N₂).
5. What is NO2 NO3 NH3 NH4?
These are common chemical forms of nitrogen found in the nitrogen cycle in ecosystem:
NO₂⁻ = Nitrite
NO₃⁻ = Nitrate
NH₃ = Ammonia
NH₄⁺ = Ammonium
These forms are produced and used during different steps of the nitrogen cycle.
6. Why is the nitrogen cycle important?
The importance of the nitrogen cycle lies in making nitrogen available to living organisms. Nitrogen is needed for proteins, DNA, RNA, chlorophyll, and many other biomolecules. The cycle also maintains soil fertility, supports plant growth, helps nutrient recycling, and keeps nitrogen balanced in the atmosphere and ecosystem.
7. What is the main component of nitrogen?
The main form of nitrogen in nature is dinitrogen gas (N₂). It is the major nitrogen form present in the atmosphere, but most living organisms cannot use it directly until it is fixed into other compounds.
8. What is assimilation in the nitrogen cycle?
Assimilation in the nitrogen cycle is the process in which plants absorb nitrates or ammonia from the soil and convert them into proteins and other organic compounds. Animals then obtain this nitrogen by eating plants or other animals.



































