What Is a Hypotonic Solution in Osmosis with Examples and Diagram
Tonicity refers to a solution's relative solute concentration in comparison to another solution. A hypotonic solution is a type of tonicity. It is one in which the concentration of soluble chemicals outside the cell is less than that inside the cell.
In a hypotonic solution, the solute concretion is always smaller than the cell. There is less solvent because there is a high concentration of solute inside the cells (water).
What is Hypotonic Solution?
Hypo denotes low, hence a hypotonic solution is one that has a higher water content than solute concentration. A solution is a molecularly dispersed mixture of one or more compounds in a sufficient quantity of dissolving solvent. A solute is a dissolved material in a solution. What enters and leaves the cell is regulated and controlled by the cell membrane. A selectively permeable membrane is a membrane that enables certain materials to pass through but not others.
In a hypotonic solution, the solute concretion is always smaller than the cell. There is less solvent because there is a high concentration of solute inside the cells (water).
Osmosis is a process in which water passes through a semipermeable membrane from a high concentration to a low concentration. The fact that the solute concentration is low in this solution implies that the solvent concentration is high. Water flows from the outside to the inside of the cell.
Hypertonic and Hypotonic Solution
A hypertonic solution is one in which the concentration of solutes in the solution is higher than the concentration of the cell in the solution. Saltwater is an example of a hypertonic solution. Whereas a hypotonic solution is a solution that has more water outside the cell. Water then travels into the cell from the solution.
Both animal and plant cells have been shown to be affected by hypertonic and hypotonic solutions. Hypertonic solution has a higher solute content and less water than a cell. There is a net migration of water from inside to outside the cell because the concentration of water is higher within the cell. Osmosis allows water to leave the cell. The loss of water causes the cells in a hypertonic solution to shrink. The animal cell will shrivel up in a hypertonic solution. This is known as crenation. The plant cell becomes less stiff in a hypertonic solution, which is known as plasmolysis.
In a hypotonic solution, The water enters the cell due to high solute concentration inside the cell. The water is more in this hypotonic solution and solute concentrations are low.
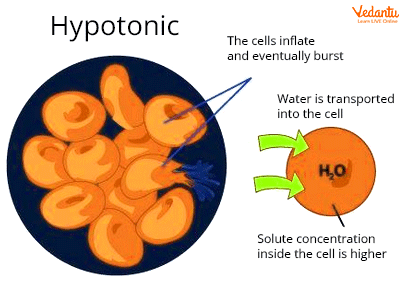
Image: Hypotonic solution
Hypotonic Solution Example
Hypotonic solutions involve distilled and clean water. Hypotonic solutions include 0.45 per cent saline and 5% dextrose in water (this is technically isotonic, but once the dextrose is absorbed, it operates on the body as if it were hypotonic).
When Cells are Placed in a Hypotonic Solution
A hypotonic solution contains a lower concentration of solutes and a higher concentration of solvent. Water fills a cell through osmosis when it is placed in a hypotonic solution. Due to the lack of a cell wall, animal cells enlarge and explode.
During hypotonic solution, the animal cell ruptures. This is called hemolysis. The plant cell would load up and become much stiffer in a hypotonic solution, and this is known as turgor pressure. The plant cell has a cell wall that exerts reverse pressure on the cell membrane.
Administration of Hypotonic Solution
Hypotonic solutions are frequently employed to dilute extracellular fluid and rehydrate cells in individuals with hypertonic fluid imbalances, as well as to treat gastric fluid loss and dehydration caused by severe diuresis. This type of solution contains no calories or other electrolytes, but it does contain free water, salt, and chloride. The cell has a low amount of extracellular solute and wants to shift into the cell to utilise standard osmosis. This promotes cell swelling, which can result in the cell bursting or lysing.
0.45 percent saline (1/2 NS), 0.225 percent saline (1/4 NS), and 0.33 percent saline (1/3 NS) are hypotonic solutions. When a cell is dehydrated and fluids must be reintroduced, hypotonic solutions are used. This happens when a patient develops diabetic ketoacidosis (DKA) or hyperosmolar hyperglycemia.
When we talk about Intravenous (IV) fluids, we usually mean that the water wants to exit the intravascular region and enter the Red Blood Cells (RBCs). The most common cause of giving 0.45 percent salinity is real dehydration, which occurs when the body loses solely water and no electrolytes (this is different from fluid volume deficit, when the body loses both water and electrolytes). The body already has a normal amount of electrolytes in dehydration. Therefore, there is no need to add extra to the IV solution. Only some of the patient's water needs to be refilled.
Conclusion
A hypotonic solution has the feature that it has a low solute concentration as compared to the cell. This causes cell swelling, and this property is used in some clinical trials to treat patients. This type of solution is provided intravenously and helps to regain the normal state of the cell and body. This article helps to understand the properties of the hypotonic solution and its meaning. It has provided all the information regarding hypotonic solutions with examples.


FAQs on Hypotonic Solution and Its Effect on Cells
1. What is a hypotonic solution in biology?
A hypotonic solution is a solution that has a lower solute concentration than the cell, causing water to move into the cell by osmosis.
- Water moves from high water potential to low water potential.
- The cell gains water and may swell.
- Common in discussions of cell transport and tonicity.
2. What happens to a cell in a hypotonic solution?
When placed in a hypotonic solution, a cell absorbs water and swells due to osmosis.
- In animal cells, excessive swelling can cause cell lysis (bursting).
- In plant cells, the cell becomes turgid but does not burst due to the cell wall.
3. Why do plant cells not burst in a hypotonic solution?
Plant cells do not burst in a hypotonic solution because their cell wall provides structural support and prevents overexpansion.
- Water enters the cell by osmosis.
- The central vacuole fills with water.
- Turgor pressure builds against the cell wall, stabilizing the cell.
4. What is the difference between hypotonic, hypertonic, and isotonic solutions?
The difference lies in solute concentration relative to the cell and the direction of water movement.
- Hypotonic solution: Lower solute concentration; water enters the cell.
- Hypertonic solution: Higher solute concentration; water leaves the cell.
- Isotonic solution: Equal solute concentration; no net water movement.
5. Can you give an example of a hypotonic solution?
An example of a hypotonic solution is distilled water compared to the cytoplasm of a cell.
- Distilled water has very low solute concentration.
- Water enters the cell by osmosis.
- Animal cells may burst, while plant cells become turgid.
6. How does osmosis work in a hypotonic solution?
In a hypotonic solution, osmosis causes water to move into the cell through a selectively permeable membrane.
- The surrounding solution has higher water potential.
- The cell interior has lower water potential.
- Water moves down its concentration gradient into the cell.
7. What is hemolysis in a hypotonic solution?
Hemolysis is the bursting of red blood cells when placed in a hypotonic solution.
- Water enters the red blood cell by osmosis.
- The cell swells due to lack of a cell wall.
- The plasma membrane ruptures, releasing hemoglobin.
8. What is turgor pressure in a hypotonic solution?
Turgor pressure is the pressure exerted by water inside a plant cell against its cell wall in a hypotonic solution.
- Water enters the central vacuole.
- The vacuole expands.
- The plasma membrane presses against the cell wall.
9. Is a hypotonic solution dangerous for human cells?
Yes, a hypotonic solution can be dangerous for human cells because it may cause them to swell and burst.
- Human cells lack a rigid cell wall.
- Excess water enters by osmosis.
- Severe swelling can lead to cell lysis.
10. How do you identify if a solution is hypotonic to a cell?
A solution is hypotonic to a cell if it has a lower solute concentration than the cell’s cytoplasm, causing water to enter the cell.
- Compare solute concentrations inside and outside the cell.
- Observe cell swelling under a microscope.
- Look for increased turgidity in plant cells or lysis in animal cells.










