The compound formed when an alcoholic solution of ethylene dibromide is heated with granulated zinc is:
(A) Ethene
(B) Ethyne
(C) Ethane
(D) Bromoethane
Answer
264.3k+ views
Hint:An elimination reaction is a type of organic reaction in which two atoms or substituents that are on adjacent carbon atoms are removed from a molecule in either one step or two-step mechanism
Complete step by step solution:
> In the question it is given that ethylene dibromide is heated with granulated zinc.
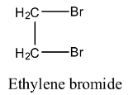
- The structure of the ethylene dibromide is as follows.
- The reaction of an alcoholic solution of ethylene dibromide is heated with granulated zinc is as follows
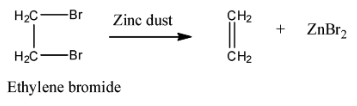
- The above reaction is called a dehalogenation reaction or elimination reaction.
- Zinc removes bromine atoms as zinc bromide (\[ZnB{{r}_{2}}\]).
Coming to given options, option B, Ethyne, means acetylene. But the product formed is not ethylene. So, it is wrong.
Coming to option C, ethane, but the product formed is acetylene. So, it is wrong.
Coming to option D, bromo ethane, it is also wrong,
Coming to option A, Ethene, means ethylene. This option is correct because the product formed in the above reaction is ethylene.
So, the correct option is A.
Note: Don’t be confused between ethane, Ethene, Ethyne. All are not the same.
Ethane-it is called alkane and a saturated compound.
Ethene-it is called alkene and ethylene. It is an unsaturated compound.
Ethyne- it is called as alkyne and called as acetylene. It is an unsaturated compound.
Complete step by step solution:
> In the question it is given that ethylene dibromide is heated with granulated zinc.
- The structure of the ethylene dibromide is as follows.

- The reaction of an alcoholic solution of ethylene dibromide is heated with granulated zinc is as follows

- The above reaction is called a dehalogenation reaction or elimination reaction.
- Zinc removes bromine atoms as zinc bromide (\[ZnB{{r}_{2}}\]).
Coming to given options, option B, Ethyne, means acetylene. But the product formed is not ethylene. So, it is wrong.
Coming to option C, ethane, but the product formed is acetylene. So, it is wrong.
Coming to option D, bromo ethane, it is also wrong,
Coming to option A, Ethene, means ethylene. This option is correct because the product formed in the above reaction is ethylene.
So, the correct option is A.
Note: Don’t be confused between ethane, Ethene, Ethyne. All are not the same.
Ethane-it is called alkane and a saturated compound.
Ethene-it is called alkene and ethylene. It is an unsaturated compound.
Ethyne- it is called as alkyne and called as acetylene. It is an unsaturated compound.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




