Aromatization of n-hexane gives:
(A) Benzene
(B) Toluene
(C) Methane
(D) A mixture of octanes
Answer
558.9k+ views
Hint: n-hexane is normal hexane or hexane and is a straight chain alkane having six carbon atoms. It has the molecular formula ${{\text{C}}_{\text{6}}}{{\text{H}}_{{\text{14}}}}$ . The structure of n-hexane is shown below.
Aromatization is a chemical reaction which involves the formation of an aromatic compound from a single non – aromatic precursor. It is usually achieved by dehydrogenation.
Complete step by step answer:
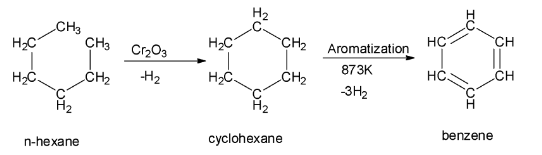
In the process of aromatization of n-hexane, the n-hexane at first is treated with vanadium oxide or chromium (III) oxide. This treatment gives a cyclic hexane or cyclohexane. This cyclohexane formed in the first step then undergoes dehydrogenation and aromatization to give benzene as the final product. The reaction of aromatization of n – hexane to give benzene is shown below:
So, the option A is correct.
Toluene can be obtained from benzene through Friedel - Craft’s alkylation process. But it cannot be directly obtained from n – hexane through the aromatization of n – hexane as aromatization of n – hexane gives benzene as the product. So, the option B is not correct.
The third option is methane which is not an aromatic compound. Since aromatization means the formation of ‘aromatic’ compounds from non – aromatic compounds, therefore methane is not acceptable. So, the option C is also not correct.
The last option is a mixture of octanes which are again not aromatic compounds. Therefore, the option D is also not correct.
So, the option A is correct.
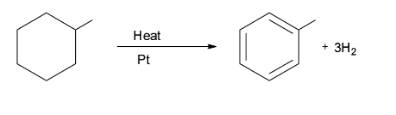
Note: The aromatization reaction has a huge application in the oil refining process. For example, it is observed in the conversion of naphthene into aromatics which is catalyzed by platinum. This is illustrated in the conversion of methylcyclohexane into toluene. This reaction is used in the production of gasoline from petroleum.

Aromatization is a chemical reaction which involves the formation of an aromatic compound from a single non – aromatic precursor. It is usually achieved by dehydrogenation.
Complete step by step answer:
In the process of aromatization of n-hexane, the n-hexane at first is treated with vanadium oxide or chromium (III) oxide. This treatment gives a cyclic hexane or cyclohexane. This cyclohexane formed in the first step then undergoes dehydrogenation and aromatization to give benzene as the final product. The reaction of aromatization of n – hexane to give benzene is shown below:

So, the option A is correct.
Toluene can be obtained from benzene through Friedel - Craft’s alkylation process. But it cannot be directly obtained from n – hexane through the aromatization of n – hexane as aromatization of n – hexane gives benzene as the product. So, the option B is not correct.
The third option is methane which is not an aromatic compound. Since aromatization means the formation of ‘aromatic’ compounds from non – aromatic compounds, therefore methane is not acceptable. So, the option C is also not correct.
The last option is a mixture of octanes which are again not aromatic compounds. Therefore, the option D is also not correct.
So, the option A is correct.
Note: The aromatization reaction has a huge application in the oil refining process. For example, it is observed in the conversion of naphthene into aromatics which is catalyzed by platinum. This is illustrated in the conversion of methylcyclohexane into toluene. This reaction is used in the production of gasoline from petroleum.

Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




