Which of the following on hydrolysis forms acetic acid
A.\[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CN}}\]
B.\[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{OH}}\]
C.\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{OH}}\]
D.\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{N}}{{\rm{H}}_2}\]
Answer
249.9k+ views
Hint: Hydrolysis refers to the breaking down of chemical bonds by a molecule of water. Water or hydroxyl ion acts as a nucleophile in this type of reaction.
Complete Step by Step Solution:
Acetic acid, also recognized as ethanoic acid, is an acidic, uncoloured liquid.
It is the simplest carboxylic acid containing two carbon atoms and thus has the name 'ethanoic acid'.
A. \[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CN}}\]
This is ethanenitrile or acetonitrile also known as methyl cyanide.
This is an uncoloured liquid and is the simplest organic nitrile.
The acidic hydrolysis of acetonitrile will produce acetic acid.
This hydrolysis happens in the presence of hydrochloric acid.
The initial step involves the formation of ethanamide.
The next step involves the formation of ethanoic acid and ammonium chloride.
The reaction happens as follows: \[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CN + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O + HCl}} \to {\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{COOH + N}}{{\rm{H}}_{\rm{4}}}{\rm{Cl}}\]
So, A is correct.
B. \[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{OH}}\]
This is methanol or methyl alcohol.
This is the simplest organic alcohol compound.
Methanol on hydrolysis does not give acetic acid.
So, B is incorrect.
C. \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{OH}}\]
This is ethanol or ethyl alcohol.
This is the next organic alcohol compound after methanol.
Ethanol on hydrolysis produces ethanal or acetaldehyde.
The reaction occurs as follows:
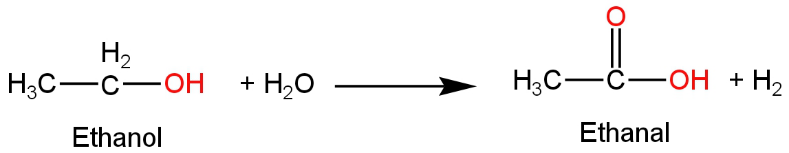
Image: Hydrolysis of ethanol
This does not produce acetic acid as the product.
So, C is incorrect.
D. \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{N}}{{\rm{H}}_2}\]
This is ethanamine or ethylamine.
It is a simple amine compound.
Ethylamine on diazotization by nitrous acid preceded by hydrolysis will give ethanol.
When this compound is treated with sodium nitrite and hydrochloric acid, the amine group is transformed into ethyl diazonium salt which then on hydrolysis will give ethanol.
The reaction happens as follows:
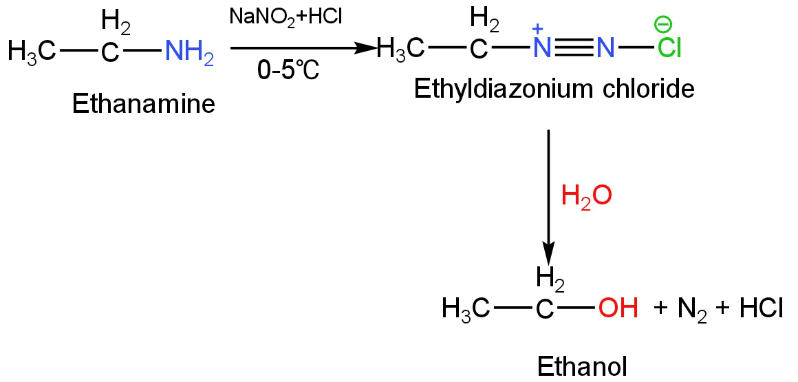
Image: Reaction of ethyl amine with nitrous acid.
So, this compound on hydrolysis will not give acetic acid.
So, D is incorrect.
So, option A is correct.
Note: Acetylene in the reaction with HgOH and dilute sulfuric acid leads to the formation of vinyl alcohol. Vinyl alcohol tautomerizes to form acetaldehyde. Acetaldehyde on oxidation forms acetic acid.
Complete Step by Step Solution:
Acetic acid, also recognized as ethanoic acid, is an acidic, uncoloured liquid.
It is the simplest carboxylic acid containing two carbon atoms and thus has the name 'ethanoic acid'.
A. \[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CN}}\]
This is ethanenitrile or acetonitrile also known as methyl cyanide.
This is an uncoloured liquid and is the simplest organic nitrile.
The acidic hydrolysis of acetonitrile will produce acetic acid.
This hydrolysis happens in the presence of hydrochloric acid.
The initial step involves the formation of ethanamide.
The next step involves the formation of ethanoic acid and ammonium chloride.
The reaction happens as follows: \[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CN + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O + HCl}} \to {\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{COOH + N}}{{\rm{H}}_{\rm{4}}}{\rm{Cl}}\]
So, A is correct.
B. \[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{OH}}\]
This is methanol or methyl alcohol.
This is the simplest organic alcohol compound.
Methanol on hydrolysis does not give acetic acid.
So, B is incorrect.
C. \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{OH}}\]
This is ethanol or ethyl alcohol.
This is the next organic alcohol compound after methanol.
Ethanol on hydrolysis produces ethanal or acetaldehyde.
The reaction occurs as follows:

Image: Hydrolysis of ethanol
This does not produce acetic acid as the product.
So, C is incorrect.
D. \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{N}}{{\rm{H}}_2}\]
This is ethanamine or ethylamine.
It is a simple amine compound.
Ethylamine on diazotization by nitrous acid preceded by hydrolysis will give ethanol.
When this compound is treated with sodium nitrite and hydrochloric acid, the amine group is transformed into ethyl diazonium salt which then on hydrolysis will give ethanol.
The reaction happens as follows:

Image: Reaction of ethyl amine with nitrous acid.
So, this compound on hydrolysis will not give acetic acid.
So, D is incorrect.
So, option A is correct.
Note: Acetylene in the reaction with HgOH and dilute sulfuric acid leads to the formation of vinyl alcohol. Vinyl alcohol tautomerizes to form acetaldehyde. Acetaldehyde on oxidation forms acetic acid.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




