Full name of DDT is:
(A) 1,1,1-trichloro-2,2-bis(p-chlorophenyl) ethane
(B) 1,1-dichloro-2,2-diphenyl trimethylethane
(C) 1,1-dichloro-2,2-diphenyl trichloroethane
(D) None of these
Answer
264.3k+ views
Hint – You can start by defining the full form of DDT and draw a well labeled of its molecular structure. Then move on to how DDT was discovered and where it is used. Especially remember to point out what harm it can do for us and the environment.
Complete step by step solution:
> The structure of DDT is shown below:
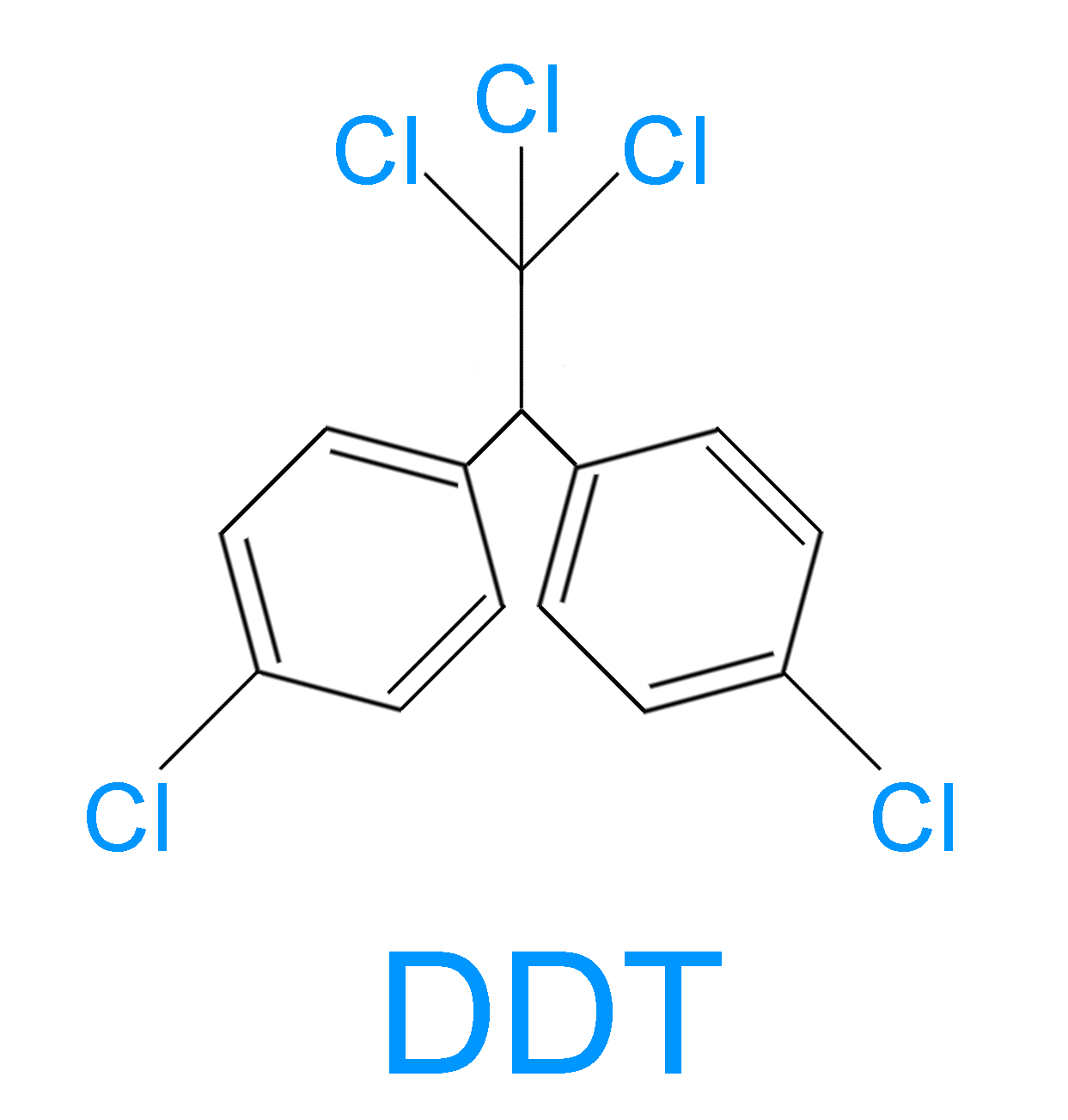
> DDT was synthesized for the first time in 1874 by an Austrian chemist Othmar Zeidler. But it was only in 1939 that a Swiss chemist Paul Hermann discovered its use as an insecticide.
> DDT has a great significance in world history. In World War II when the world was engulfed in war, diseases like malaria wreaked havoc among the soldiers and civilians. So DDT came as a potential solution to reduce the number of mosquitos (the vectors for malaria causing pathogens).
> The reaction by which DDT is produced is as follows
$2{C_6}{H_5}Cl(chlorobenzene) + {C_2}HOC{l_3}(chloral)\xrightarrow{{}}{C_{14}}{H_9}C{l_5}(DDT) + {H_2}O$
> As you can see chlorobenzene and chloral are used as the raw material for the process of making 1,1-dichloro-2,2-diphenyl trichloroethane.
> DDT should be handled carefully, as DDT can disturb the endocrine system of humans. It is also considered as a carcinogen (substances that can potentially cause the formation of cancerous cells). In case of shorter periods of time it does not harm that much and is classified by the US National Toxicology Program as “moderately toxic”.
Therefore option (A) 1,1,1-trichloro-2,2-bis(p-chlorophenyl) ethane is the correct answer.
Note - DDT became available for public sale in 1945 for use as an agricultural pesticide and household insecticide. By 1944 we started hearing concerns that excessive use of DDT might do more harm than good. It was reported that excessive use of DDT can not only kill insects but also kills fish, birds and other forms of life.
Complete step by step solution:
> The structure of DDT is shown below:

> DDT was synthesized for the first time in 1874 by an Austrian chemist Othmar Zeidler. But it was only in 1939 that a Swiss chemist Paul Hermann discovered its use as an insecticide.
> DDT has a great significance in world history. In World War II when the world was engulfed in war, diseases like malaria wreaked havoc among the soldiers and civilians. So DDT came as a potential solution to reduce the number of mosquitos (the vectors for malaria causing pathogens).
> The reaction by which DDT is produced is as follows
$2{C_6}{H_5}Cl(chlorobenzene) + {C_2}HOC{l_3}(chloral)\xrightarrow{{}}{C_{14}}{H_9}C{l_5}(DDT) + {H_2}O$
> As you can see chlorobenzene and chloral are used as the raw material for the process of making 1,1-dichloro-2,2-diphenyl trichloroethane.
> DDT should be handled carefully, as DDT can disturb the endocrine system of humans. It is also considered as a carcinogen (substances that can potentially cause the formation of cancerous cells). In case of shorter periods of time it does not harm that much and is classified by the US National Toxicology Program as “moderately toxic”.
Therefore option (A) 1,1,1-trichloro-2,2-bis(p-chlorophenyl) ethane is the correct answer.
Note - DDT became available for public sale in 1945 for use as an agricultural pesticide and household insecticide. By 1944 we started hearing concerns that excessive use of DDT might do more harm than good. It was reported that excessive use of DDT can not only kill insects but also kills fish, birds and other forms of life.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




