The number of isomers in ${C_4}{H_{10}}O$ are:
a.)7
b.)8
c.)6
d.)5
Answer
269.1k+ views
Hint:
Draw the different structures possible using this formula. The number is the sum of isomers both with alcohol as a functional group and ether.
Complete step by step answer:
This question is about the number of isomers in ${C_4}{H_{10}}O$. First, let’s see what structural isomers are:
The structural isomers are those in which atoms remain same while they differ in bonding with each other. The number of atoms of each type are the same. Now, for the compound given to us which is a four-carbon compound with one Oxygen atom. We know that for such a compound two functional groups are possible which are alcohol and ether.
Now, let’s start drawing the structure of alcohol first.
The first molecule, we will draw with all four carbons in a single chain. It will be n- Butanol.
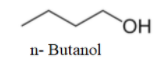
We can see that one more molecule with straight chain can be drawn as-
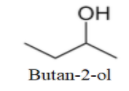
Now, as no more straight chain molecule can be formed; so, we will make branched molecules as-
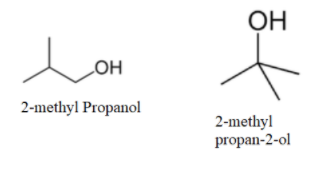
We can see that no more molecule with alcohol as a functional group can be made. So, now we will make ethers as-
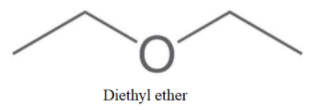
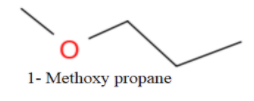
Now, no more straight chain ether is possible, So, we will form branching as-
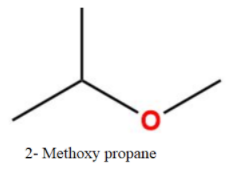
Thus, the total number of isomers are-
Alcohols=4
Ethers=3
Total isomers=7
So, option a.) is the correct answer.
Note:
If we try to make more number of straight chain alcohol, it will form the same molecule only. Example is-
Both these will form one molecule only.

Draw the different structures possible using this formula. The number is the sum of isomers both with alcohol as a functional group and ether.
Complete step by step answer:
This question is about the number of isomers in ${C_4}{H_{10}}O$. First, let’s see what structural isomers are:
The structural isomers are those in which atoms remain same while they differ in bonding with each other. The number of atoms of each type are the same. Now, for the compound given to us which is a four-carbon compound with one Oxygen atom. We know that for such a compound two functional groups are possible which are alcohol and ether.
Now, let’s start drawing the structure of alcohol first.
The first molecule, we will draw with all four carbons in a single chain. It will be n- Butanol.

We can see that one more molecule with straight chain can be drawn as-

Now, as no more straight chain molecule can be formed; so, we will make branched molecules as-

We can see that no more molecule with alcohol as a functional group can be made. So, now we will make ethers as-


Now, no more straight chain ether is possible, So, we will form branching as-

Thus, the total number of isomers are-
Alcohols=4
Ethers=3
Total isomers=7
So, option a.) is the correct answer.
Note:
If we try to make more number of straight chain alcohol, it will form the same molecule only. Example is-
Both these will form one molecule only.

Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




