Which of the following is not used as an explosive?
(A) Trinitrotoluene
(B) Trinitrobenzene
(C) Picric acid
(D) Nitrobenzene
Answer
268.2k+ views
Hint: An explosive substance is a solid, liquid, or mixture that, by chemical reaction, is capable of releasing gas at a temperature, pressure, and speed high enough to harm the environment. An organic compound is said to be explosive if its explosion leads to highly stable compounds with the release of energy.
Complete Step by Step Solution:
Trinitrotoluene (TNT) is a solid organic compound that is mostly used as an explosive. It is a pale yellow colour. It is not very sensitive to shock and requires a detonator to explode. It is the most popular chemical explosive and is widely employed in weapons and demolitions for these reasons.
Trinitrobenzene is a strong explosive when it is dry. Harmful nitrogen oxides are produced when trinitrobenzene is exposed to heat or fire. Though more explosive than TNT, trinitrobenzene is too costly. Military and commercial mining operations utilise it mainly as a highly explosive compound.
Picric acid is also mainly used as an explosive. Dried-out picric acid should be handled with caution because it could explode if exposed to heat, flame, friction, or shock. When combined with bases, metals, ammonia, concrete, and other oxidising substances, picric acid can react violently and produce salts that are unstable.
However, nitrobenzene is not explosive. It is a stable compound up to about${{450}^{o}}C$.
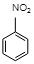
Correct option: (D) Nitrobenzene.
Note: Di or tri nitro compounds are explosive, but mono nitro compounds are not. Nitrobenzene is used in lubricants used in machinery and motors. Nitrobenzene is a very minor component of the pigments, medicines, insecticides, and rubber product manufacturing processes.
Complete Step by Step Solution:
Trinitrotoluene (TNT) is a solid organic compound that is mostly used as an explosive. It is a pale yellow colour. It is not very sensitive to shock and requires a detonator to explode. It is the most popular chemical explosive and is widely employed in weapons and demolitions for these reasons.
Trinitrobenzene is a strong explosive when it is dry. Harmful nitrogen oxides are produced when trinitrobenzene is exposed to heat or fire. Though more explosive than TNT, trinitrobenzene is too costly. Military and commercial mining operations utilise it mainly as a highly explosive compound.
Picric acid is also mainly used as an explosive. Dried-out picric acid should be handled with caution because it could explode if exposed to heat, flame, friction, or shock. When combined with bases, metals, ammonia, concrete, and other oxidising substances, picric acid can react violently and produce salts that are unstable.
However, nitrobenzene is not explosive. It is a stable compound up to about${{450}^{o}}C$.

Correct option: (D) Nitrobenzene.
Note: Di or tri nitro compounds are explosive, but mono nitro compounds are not. Nitrobenzene is used in lubricants used in machinery and motors. Nitrobenzene is a very minor component of the pigments, medicines, insecticides, and rubber product manufacturing processes.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




