
Which of the following is an example of metallic crystalline solids?
(a) \[C\]
(b) \[W\]
(c) \[Si\]
(d) \[AgCl\]
Answer
244.5k+ views
Hint: The solid is a state of matter, in which the constituent particles remain closely packed with the minimum amount of kinetic energy. Generally, solids are recognized for their rigid and hard nature. In most cases they have cubic crystal structures.
Complete Step by Step Solution:
In metallic solids the constituents’ particles i.e., metal atoms are fused together by metallic bonds.
In metallic solids, the positively charged ions are evenly distributed in the crystal and surrounded by free mobile electrons.
In metallic solid the mobile electrons are not held between a couple of atoms i.e., they can delocalize. On the other hand, the metallic solid possesses a sea of electrons everywhere (Image 1).
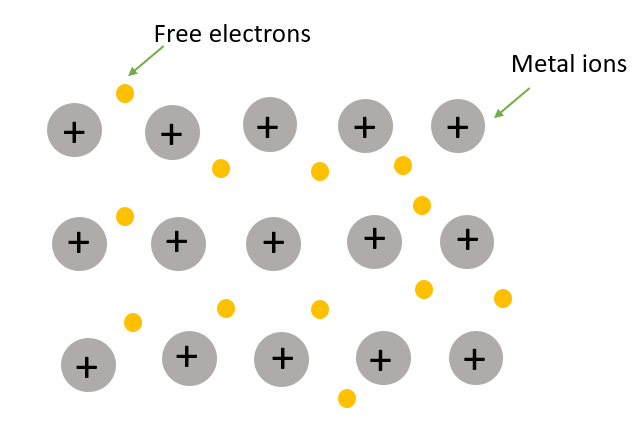
Image 1: Structure of metallic solid.
In metallic solid each atom can share one or more electrons to the sea of mobile electrons. These behaviours cause an increase in the electrical and thermal conductivity of metallic solids.
Metallic crystalline solids are malleable and ductile i.e., they can easily convert into sheets and wires respectively.
Unlike covalent solids, they are stiff or brittle in nature because the cations can slide into the sea of electrons without breaking any bond.
Metallic solids such as copper, gold, zinc, tungsten, etc. are examples of such solids.
Therefore, option (b) will be the correct answer because silicon, carbon and \[AgCl\] are not metals. While \[W\] is a metal.
Note: The metallic solid can be pure, or they can be the combination of two or more than two metals. Bronze is alloy and it has the mixture of two metals i.e., copper and tin.
Complete Step by Step Solution:
In metallic solids the constituents’ particles i.e., metal atoms are fused together by metallic bonds.
In metallic solids, the positively charged ions are evenly distributed in the crystal and surrounded by free mobile electrons.
In metallic solid the mobile electrons are not held between a couple of atoms i.e., they can delocalize. On the other hand, the metallic solid possesses a sea of electrons everywhere (Image 1).

Image 1: Structure of metallic solid.
In metallic solid each atom can share one or more electrons to the sea of mobile electrons. These behaviours cause an increase in the electrical and thermal conductivity of metallic solids.
Metallic crystalline solids are malleable and ductile i.e., they can easily convert into sheets and wires respectively.
Unlike covalent solids, they are stiff or brittle in nature because the cations can slide into the sea of electrons without breaking any bond.
Metallic solids such as copper, gold, zinc, tungsten, etc. are examples of such solids.
Therefore, option (b) will be the correct answer because silicon, carbon and \[AgCl\] are not metals. While \[W\] is a metal.
Note: The metallic solid can be pure, or they can be the combination of two or more than two metals. Bronze is alloy and it has the mixture of two metals i.e., copper and tin.
Recently Updated Pages
JEE Main 2026 Admit Card OUT LIVE Soon| Session 2 Direct Download Link

JEE Main 2026 Session 2 City Intimation Slip Expected Soon: Check How to Download

JEE Main 2026 Session 2 Application Form: Reopened Registration, Dates & Fees

JEE Main 2026 Session 2 Registration (Reopened): Last Date, Fees, Link & Process

WBJEE 2026 Registration Started: Important Dates Eligibility Syllabus Exam Pattern

Types of Solutions in Chemistry: Explained Simply

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Understanding the Angle of Deviation in a Prism

Understanding Differential Equations: A Complete Guide

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




