The maximum number of NO2 group that can introduce by nitration of benzene is usually
A. 4
B. 2
C. 3
D. 6
Answer
261k+ views
Hint: Group NO2 is electrophile and thus, during the nitration of benzene this group will attach at the position where there electron density is more. There are three positions in benzene, ortho, meta, and para. Ortho is 1,2 relation, meta is 1,3 relation and para is 1,4 relation as per numbering. Find no. of electron rich positions to get the maximum number of NO2 groups to introduce.
Complete Step by Step Solution:
To know that how much NO2 group will attach on benzene it is required to draw resonating structures of nitrobenzene which firstly form after the first nitration such as
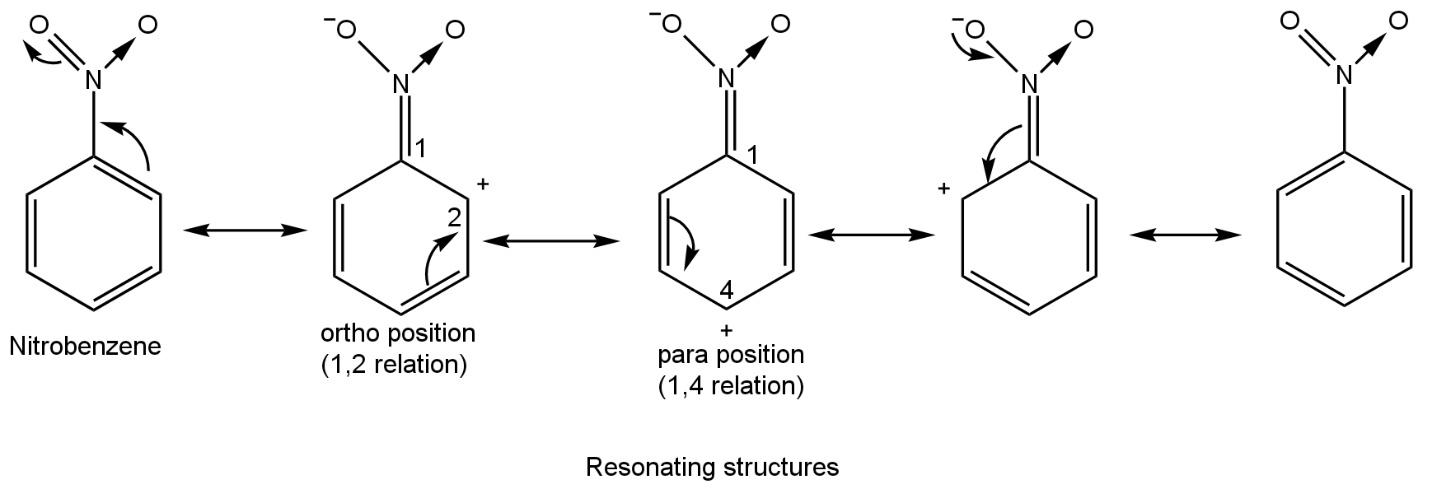
From all resonating structures of nitrobenzene, positive charge resides on ortho and para position with respect to carbon number 1 which is directly attached to the nitro group. Thus, electron density at ortho and para will be less and at meta will be relatively more. Thus, further nitration leads to attaching the NO2 group at meta position.
So, NO2 can attach to meta position with respect to carbon 1 (directly attached to NO2 group). As there are two meta positions thus two NO2 groups can join at this position. Altogether three NO2 groups can be introduced by nitration of benzene.
Thus, the correct option is B.
Note: After nitration of benzene, HNO2 group is attached directly to the ring. As nitrogen is attached to the benzene ring and also directly attached to the oxygen atom, this group will behave as an electron withdrawing group and attract the whole electron density of the ring towards itself.
Complete Step by Step Solution:
To know that how much NO2 group will attach on benzene it is required to draw resonating structures of nitrobenzene which firstly form after the first nitration such as

From all resonating structures of nitrobenzene, positive charge resides on ortho and para position with respect to carbon number 1 which is directly attached to the nitro group. Thus, electron density at ortho and para will be less and at meta will be relatively more. Thus, further nitration leads to attaching the NO2 group at meta position.
So, NO2 can attach to meta position with respect to carbon 1 (directly attached to NO2 group). As there are two meta positions thus two NO2 groups can join at this position. Altogether three NO2 groups can be introduced by nitration of benzene.
Thus, the correct option is B.
Note: After nitration of benzene, HNO2 group is attached directly to the ring. As nitrogen is attached to the benzene ring and also directly attached to the oxygen atom, this group will behave as an electron withdrawing group and attract the whole electron density of the ring towards itself.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




