The compound A with following sequence of reaction gave benzoic acid

A. Nitrobenzene
B. Aniline
C. Benzaldehyde
D. Amides
Answer
259.2k+ views
Hint: When aniline undergoes a reaction with nitrous acid at the temperature of 273K -278K results in the formation of benzenediazonium chloride. The production of nitrous acid is due to the reaction of sodium nitrite with hydrochloric acid.
Complete Step by Step Solution:
In the given question, the reactant A undergoes reaction with \[{\rm{NaN}}{{\rm{O}}_{\rm{2}}}/{\rm{HCl}}\] to form B. The then product formed B reacts with KCN to form C. The acidic hydrolysis of C results in the formation of benzoic acid.
Let’s understand the diazotization reaction in detail. In the diazotization reaction, the primary aromatic amine converts into diazonium salt. Here, A undergoes undergoes reaction with \[{\rm{NaN}}{{\rm{O}}_{\rm{2}}}/{\rm{HCl}}\]. So, the reaction is,
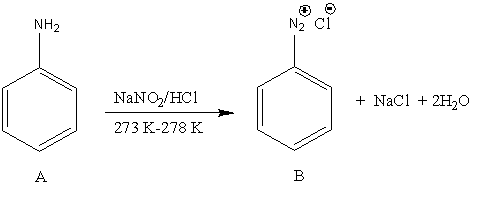
Image: Reaction of aniline with \[{\rm{NaN}}{{\rm{O}}_{\rm{2}}}/{\rm{HCl}}\]
In the second reaction, B undergoes a reaction with KCN. The reaction of diazonium chloride with KCN results in the formation of cyanide. So, the reaction is,
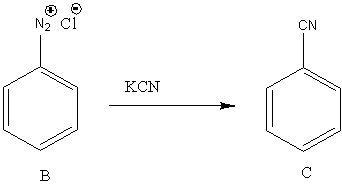
Image: Reaction of diazonium chloride with KCN
So, product C is benzonitrile.
Let’s understand the third reaction. The acidic hydrolysis of benzonitrile gives benzoic acid.
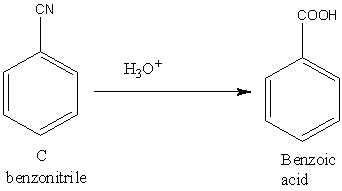
Image: Acidic hydrolysis of benzonitrile
So, the complete reaction is,
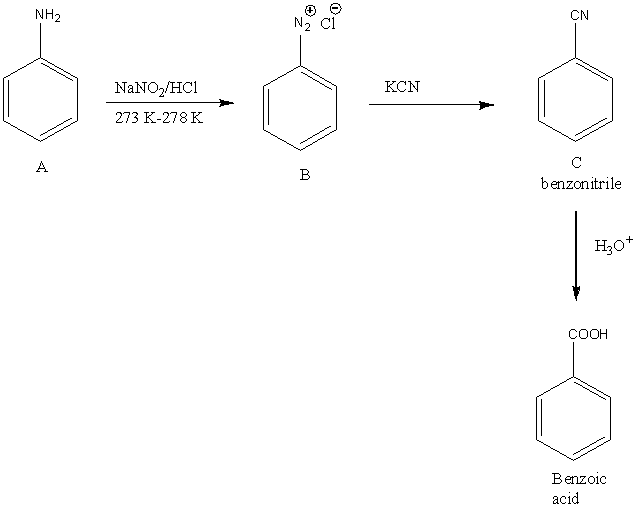
Image: The reaction of aniline to form benzoic acid
Hence, the reactant A is aniline, i.e, \[{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{N}}{{\rm{H}}_{\rm{2}}}\] .
Note: The Sandmeyer reaction is the one in which chloride, bromide and cyanide ion can be introduced in the benzene ring in the presence of copper ion. And, we know, the acidic hydrolysis of cyanide gives the carboxylic acid.
Complete Step by Step Solution:
In the given question, the reactant A undergoes reaction with \[{\rm{NaN}}{{\rm{O}}_{\rm{2}}}/{\rm{HCl}}\] to form B. The then product formed B reacts with KCN to form C. The acidic hydrolysis of C results in the formation of benzoic acid.
Let’s understand the diazotization reaction in detail. In the diazotization reaction, the primary aromatic amine converts into diazonium salt. Here, A undergoes undergoes reaction with \[{\rm{NaN}}{{\rm{O}}_{\rm{2}}}/{\rm{HCl}}\]. So, the reaction is,

Image: Reaction of aniline with \[{\rm{NaN}}{{\rm{O}}_{\rm{2}}}/{\rm{HCl}}\]
In the second reaction, B undergoes a reaction with KCN. The reaction of diazonium chloride with KCN results in the formation of cyanide. So, the reaction is,

Image: Reaction of diazonium chloride with KCN
So, product C is benzonitrile.
Let’s understand the third reaction. The acidic hydrolysis of benzonitrile gives benzoic acid.

Image: Acidic hydrolysis of benzonitrile
So, the complete reaction is,

Image: The reaction of aniline to form benzoic acid
Hence, the reactant A is aniline, i.e, \[{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{\rm{5}}}{\rm{N}}{{\rm{H}}_{\rm{2}}}\] .
Note: The Sandmeyer reaction is the one in which chloride, bromide and cyanide ion can be introduced in the benzene ring in the presence of copper ion. And, we know, the acidic hydrolysis of cyanide gives the carboxylic acid.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




