Structural formula for lewisite is
A.
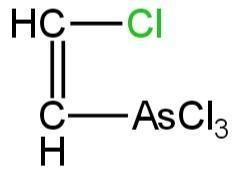
B.
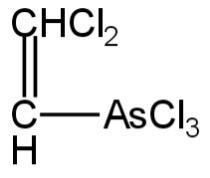
C.
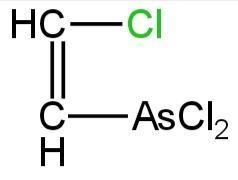
D. None of these
Answer
268.5k+ views
Hint: A molecule of AsCl3 gets added to acetylene giving lewisite. It is an organoarsenic compound i.e., including organic units, ethene, and arsenic. It was employed by many countries for its usage as a chemical weapon, behaving as a blister agent and lung irritant.
Complete Step by Step Solution:
Lewisite is a compound that is readied by the addition of a molecule of arsenic trichloride to acetylene in the presence of a desirable catalyst.
This catalyst mainly is aluminium chloride.
\[AsC{l_3} + {C_2}{H_2} \to ClCHCHAsC{l_2}\]
Lewisite undergoes hydrolysis in water to form hydrochloric acid and chlorovinyl arsenious oxide which is a less strong blister agent.
\[{\rm{ClCHCHAsC}}{{\rm{l}}_{\rm{2}}}{\rm{ + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O}} \to {\rm{ClCHCHAs}}{\left( {{\rm{OH}}} \right)_{\rm{2}}}{\rm{ + 2 HCl}}\]
This reaction is quickened by alkaline solutions and forms acetylene and trisodium arsenate.
Lewisite reacts with metals to yield hydrogen gas. It is combustible, but difficult to ignite.
It is an arsenic-based compound that was formulated to be a powerful chemical to be used in the war.
Susceptibility to lewisite commonly influences the eyes, skin, and respiratory tract which happen nearly immediately after the following contact.
As a chemical combat agent, it can be circulated as a liquid, aerosol, or vapour and can be utilised to provoke fatalities and for area denial.
So, the chemical formula of lewisite is\[ClCHCHAsC{l_2}\]. So, its structure is C.
So, option C is correct.
Note: Lewisite was made in 1904 by Julius Arthur Nieuwland during research for his Ph.D. In his thesis, he depicted a reaction between acetylene and arsenic trichloride, which guided the formation of lewisite. Revelation to the ensuing compound made Nieuwland so sick he was in the hospital for many days.
Complete Step by Step Solution:
Lewisite is a compound that is readied by the addition of a molecule of arsenic trichloride to acetylene in the presence of a desirable catalyst.
This catalyst mainly is aluminium chloride.
\[AsC{l_3} + {C_2}{H_2} \to ClCHCHAsC{l_2}\]
Lewisite undergoes hydrolysis in water to form hydrochloric acid and chlorovinyl arsenious oxide which is a less strong blister agent.
\[{\rm{ClCHCHAsC}}{{\rm{l}}_{\rm{2}}}{\rm{ + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O}} \to {\rm{ClCHCHAs}}{\left( {{\rm{OH}}} \right)_{\rm{2}}}{\rm{ + 2 HCl}}\]
This reaction is quickened by alkaline solutions and forms acetylene and trisodium arsenate.
Lewisite reacts with metals to yield hydrogen gas. It is combustible, but difficult to ignite.
It is an arsenic-based compound that was formulated to be a powerful chemical to be used in the war.
Susceptibility to lewisite commonly influences the eyes, skin, and respiratory tract which happen nearly immediately after the following contact.
As a chemical combat agent, it can be circulated as a liquid, aerosol, or vapour and can be utilised to provoke fatalities and for area denial.
So, the chemical formula of lewisite is\[ClCHCHAsC{l_2}\]. So, its structure is C.
So, option C is correct.
Note: Lewisite was made in 1904 by Julius Arthur Nieuwland during research for his Ph.D. In his thesis, he depicted a reaction between acetylene and arsenic trichloride, which guided the formation of lewisite. Revelation to the ensuing compound made Nieuwland so sick he was in the hospital for many days.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




