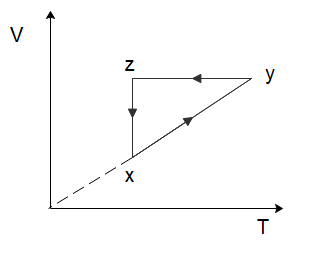
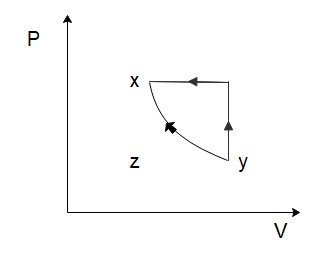
A thermodynamic cycle xyzx is shown on a V−T diagram. The P−V diagram that best describes this cycle is: (Diagrams are schematic and not to scale)
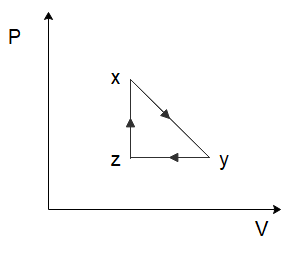
(A)
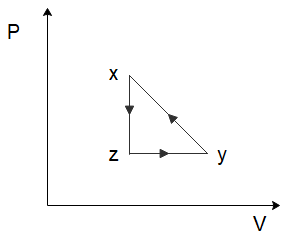
(B)
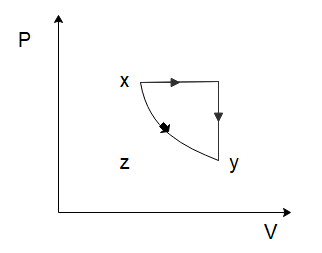
(C)
(D)
Answer
249.6k+ views
Hint: In this solution, we will use the ideal gas formula to determine the relationship between pressure-volume and temperature. For each process, we will determine which quantity remains constant and which quantity changes.
Formula used: In this solution, we will use the following formula:
-Ideal gas law: \[PV = nRT\] where $P$ is the pressure, $V$ is the volume, $n$ is the number of moles, $R$is the gas constant, and $T$ is the temperature
Complete step by step answer:
To solve this problem let us start by considering the process of $x \to y$. From the graph, we can see that both the temperature and the volume of the gas are increasing. Now in the relation \[PV = nRT\], $n$ and $R$ are constants. So, if the temperature and the volume both increase, the pressure would have to remain constant for the equation to hold true. So, for the process $x \to y$, the pressure will remain constant while the volume increases. Among the options given to us, this is only shown in option (C).
So the correct option is C
Note: Often, we cannot determine the correct choice from just one process, so let us determine the graphs of the other processes as well.
In the process \[y \to z\], we can see that the volume of the gas remains constant, while the temperature of the gas decreases. Again, from the ideal gas law, we can deduce that the pressure of the gas would have to decrease to ensure the ideal gas law holds true. So, the pressure decreases, and the volume of the gas remains constant which is again only shown in option (C).
Formula used: In this solution, we will use the following formula:
-Ideal gas law: \[PV = nRT\] where $P$ is the pressure, $V$ is the volume, $n$ is the number of moles, $R$is the gas constant, and $T$ is the temperature
Complete step by step answer:
To solve this problem let us start by considering the process of $x \to y$. From the graph, we can see that both the temperature and the volume of the gas are increasing. Now in the relation \[PV = nRT\], $n$ and $R$ are constants. So, if the temperature and the volume both increase, the pressure would have to remain constant for the equation to hold true. So, for the process $x \to y$, the pressure will remain constant while the volume increases. Among the options given to us, this is only shown in option (C).
So the correct option is C
Note: Often, we cannot determine the correct choice from just one process, so let us determine the graphs of the other processes as well.
In the process \[y \to z\], we can see that the volume of the gas remains constant, while the temperature of the gas decreases. Again, from the ideal gas law, we can deduce that the pressure of the gas would have to decrease to ensure the ideal gas law holds true. So, the pressure decreases, and the volume of the gas remains constant which is again only shown in option (C).
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
Understanding the Angle of Deviation in a Prism

Understanding Centrifugal Force in Physics

Understanding Charging and Discharging of Capacitors

Understanding Electromagnetic Waves and Their Importance

The least count of a stop watch is 02s The time of class 11 physics JEE_Main

A Polaroid examines two adjacent plane polarised beams class 11 physics JEE_Main

Other Pages
NCERT Solutions For Class 11 Physics Chapter 3 Motion In A Plane - 2025-26

NCERT Solutions For Class 11 Physics Chapter 4 Laws Of Motion - 2025-26

NCERT Solutions For Class 11 Physics Chapter 10 Thermal Properties Of Matter - 2025-26

NCERT Solutions For Class 11 Physics Chapter 13 Oscillations - 2025-26

CBSE Notes Class 11 Physics Chapter 5 - Work Energy and Power - 2025-26

Class 11 JEE Main Physics Mock Test 2025




