NCERT Exemplar Available Free On Vedantu
Free PDF download of NCERT Exemplar for Class 12 Chemistry Chapter 15 - Polymers solved by expert Chemistry teachers on Vedantu as per NCERT (CBSE) Book guidelines. All Chapter 15 - Polymers exercise questions with solutions to help you to revise the complete syllabus and score more marks in your examinations.
Access NCERT Exemplar Solutions for Class 12 Chemistry Chapter 15- Polymers
I. Multiple Choice Questions (Type-I)
1. Which of the following polymers of glucose are stored by animals?
(A) Cellulose
(B) Amylose
(C) Amylopectin
(D) Glycogen
Ans: Option (D)
Explanation:
Glycogen is a branched polymer of glucose found in the liver, muscles, and brain of animals.
2. Which of the following is not a semisynthetic polymer?
(A) cis-polyisoprene
(B) Cellulose nitrate
(C) Cellulose acetate
(D) Vulcanised rubber
Ans: Option (A)
Explanation:
Semi-synthetic polymers are obtained from natural polymers. Cellulose derivatives like cellulose nitrate, cellulose acetate rubber are examples of semi-synthetic polymers.
Cis-polyisoprene is not a semi-synthetic polymer while other examples are semi-synthetic polymer.
3. The commercial name of polyacrylonitrile is ______________.
(A) Dacron
(B) Orlon (acrilan)
(C) PVC
(D) Bakelite
Ans: Option (B)
Explanation:
Polyacrylonitrile is used as a wool substitute in the manufacture of commercial fibers such as orlon or acrilan.
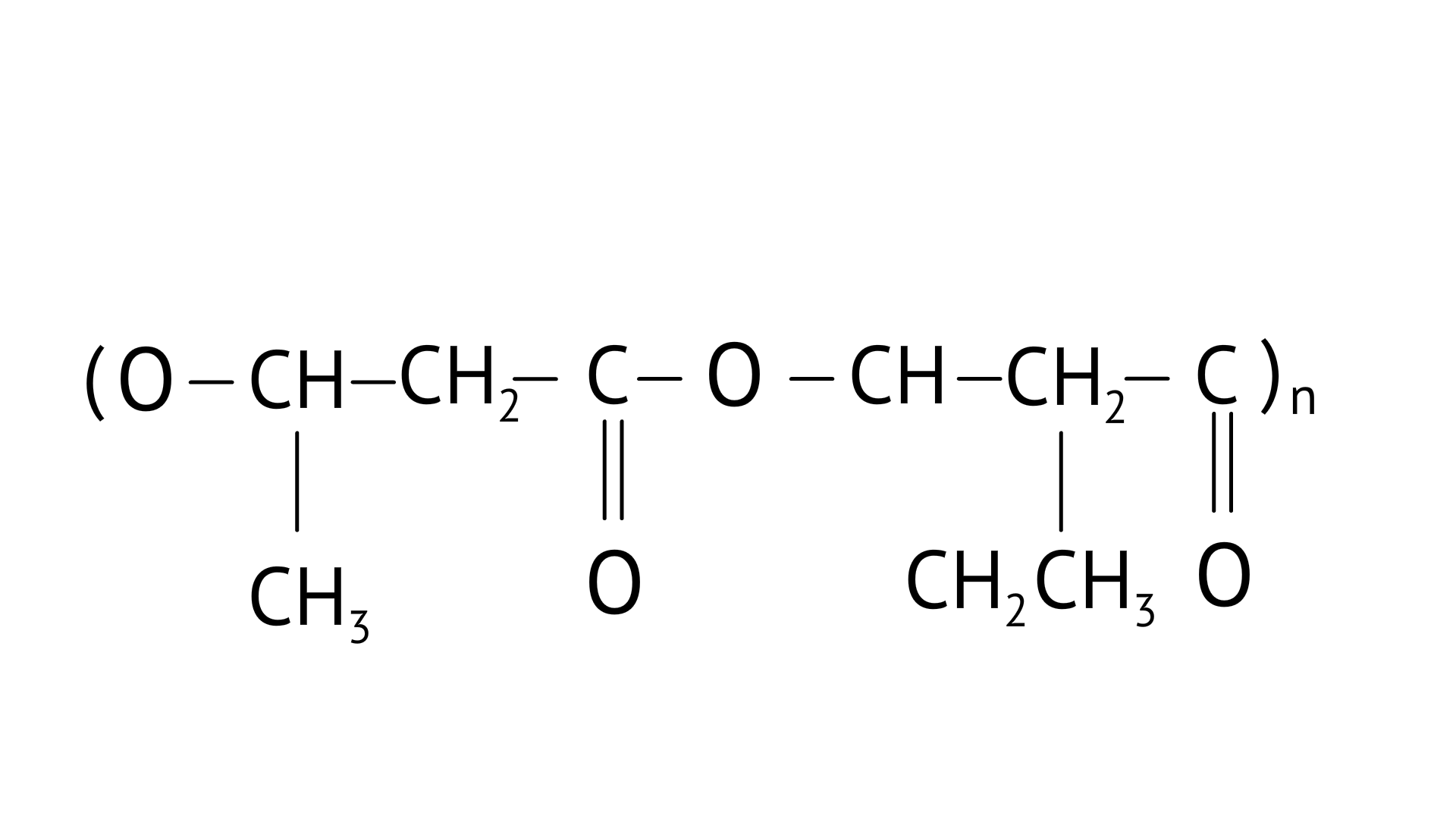
4. Which of the following polymers is biodegradable?
(A)
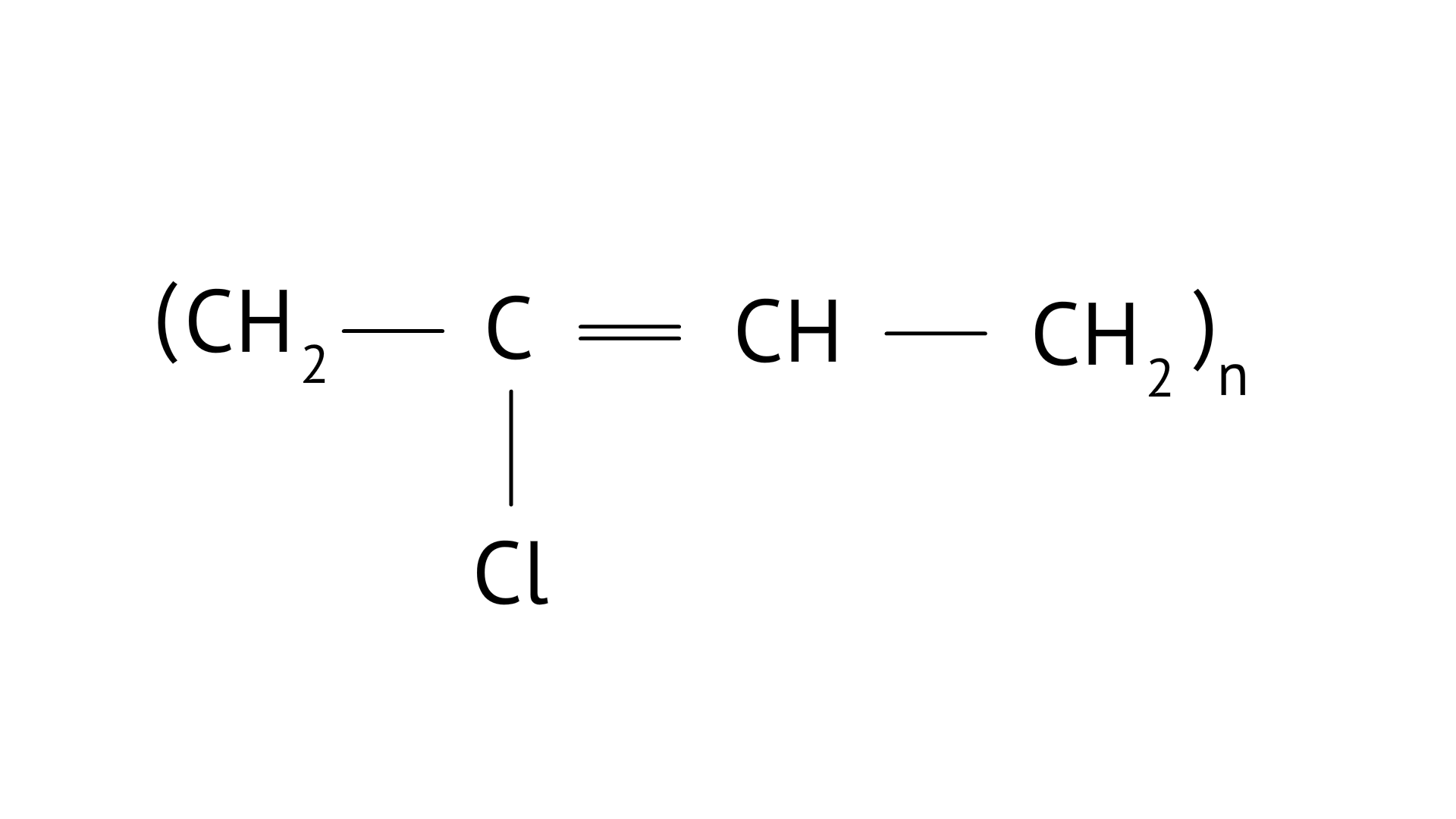
(B)
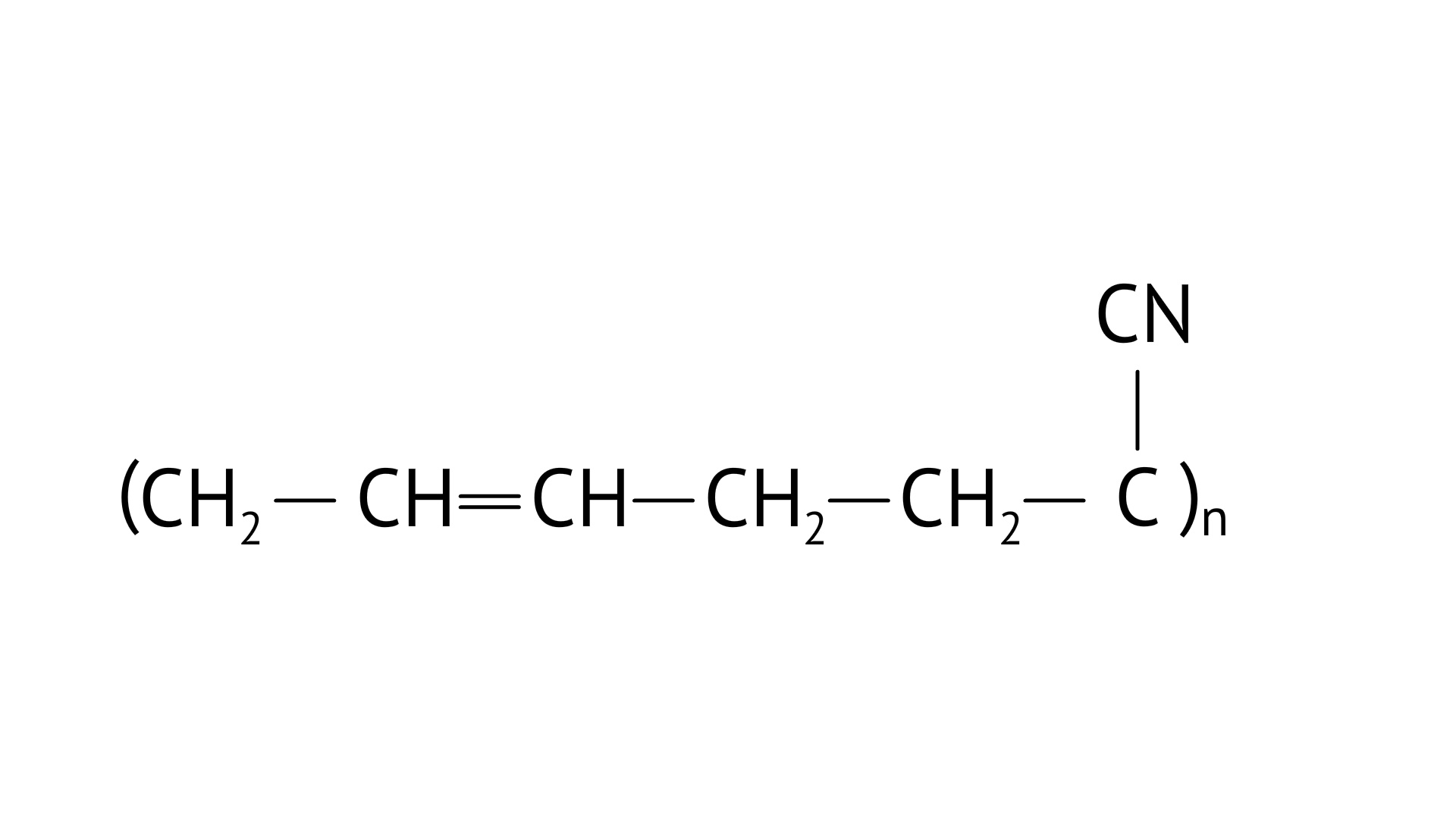
(C)
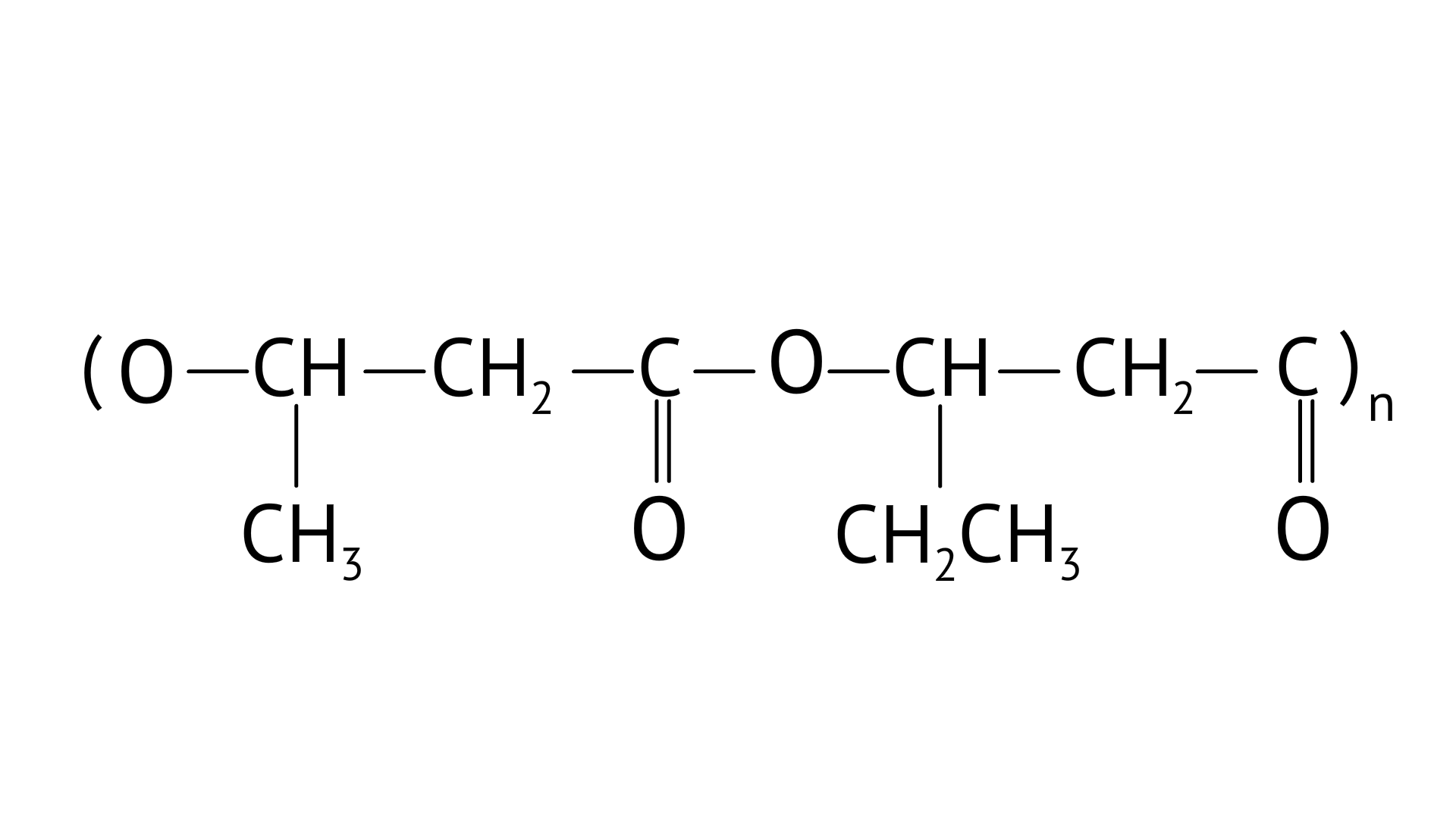
(D)
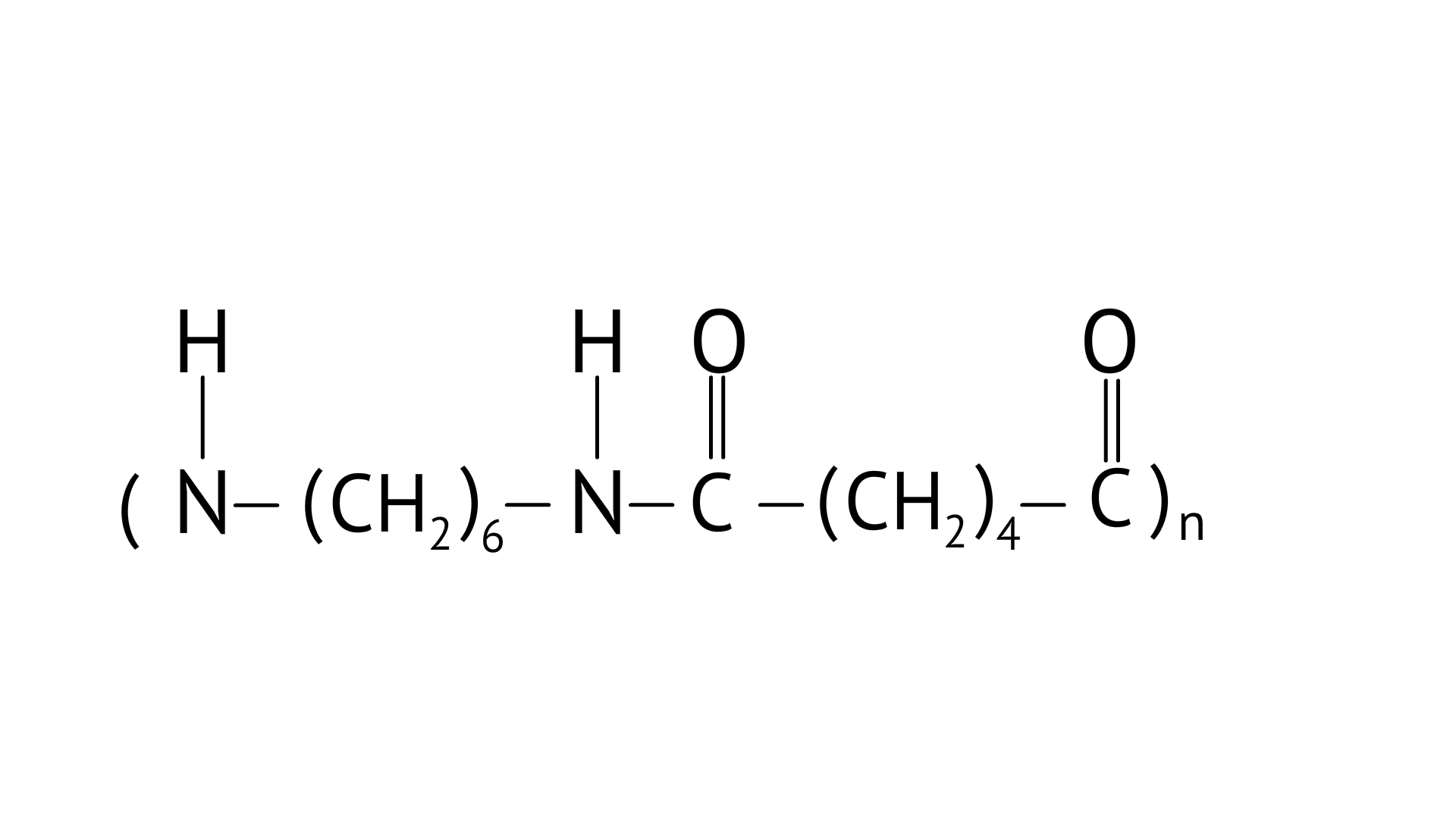
Ans: Option (C)
Explanation:
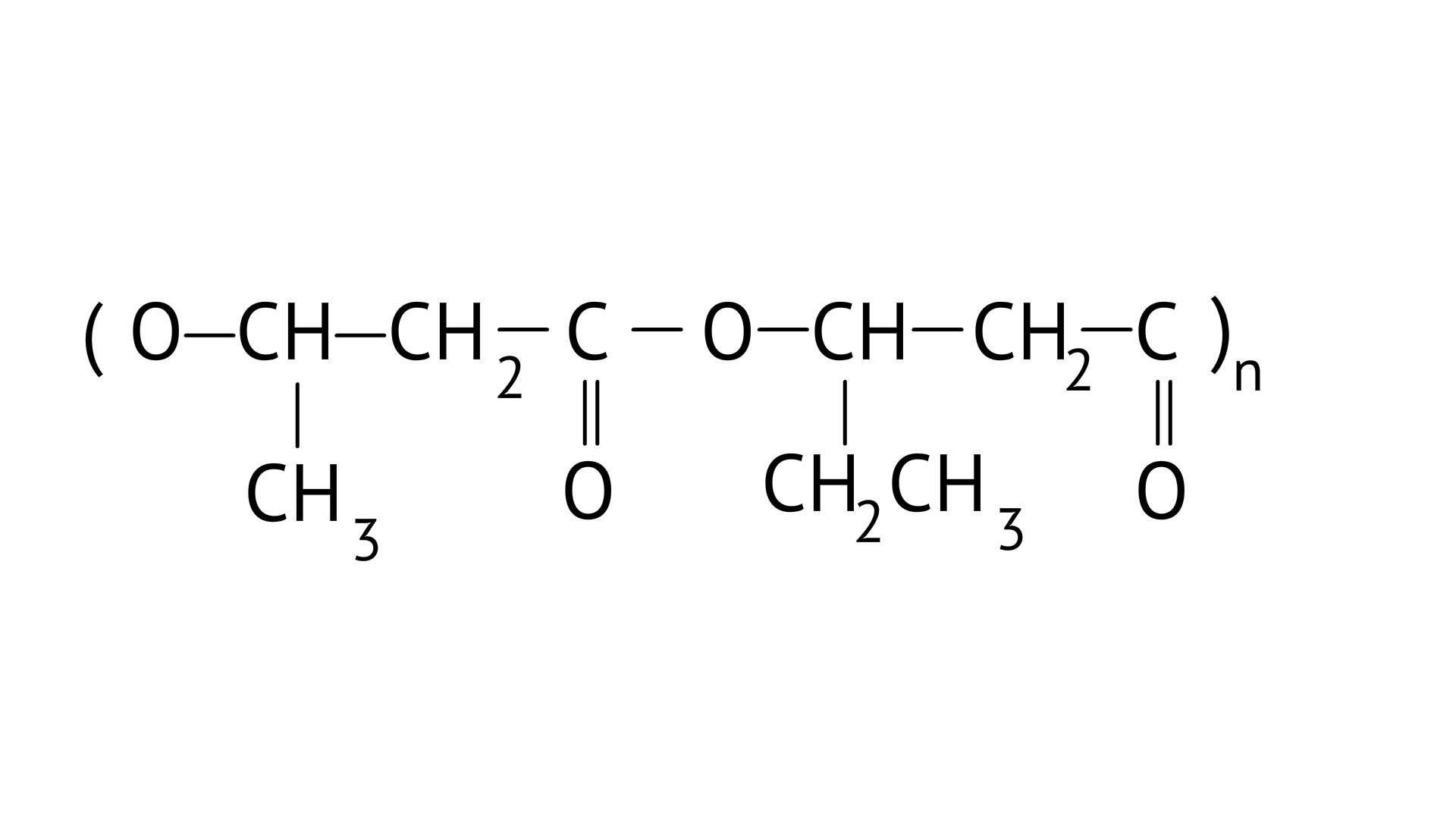
PHBV(Poly beta-hydroxybutyrate co-beta-hydroxy valerate) is a biodegradable polymer. It undergoes bacterial degradation in the environment. The structure is shown as,
5. In which of the following polymers ethylene glycol is one of the monomer units?
(A)
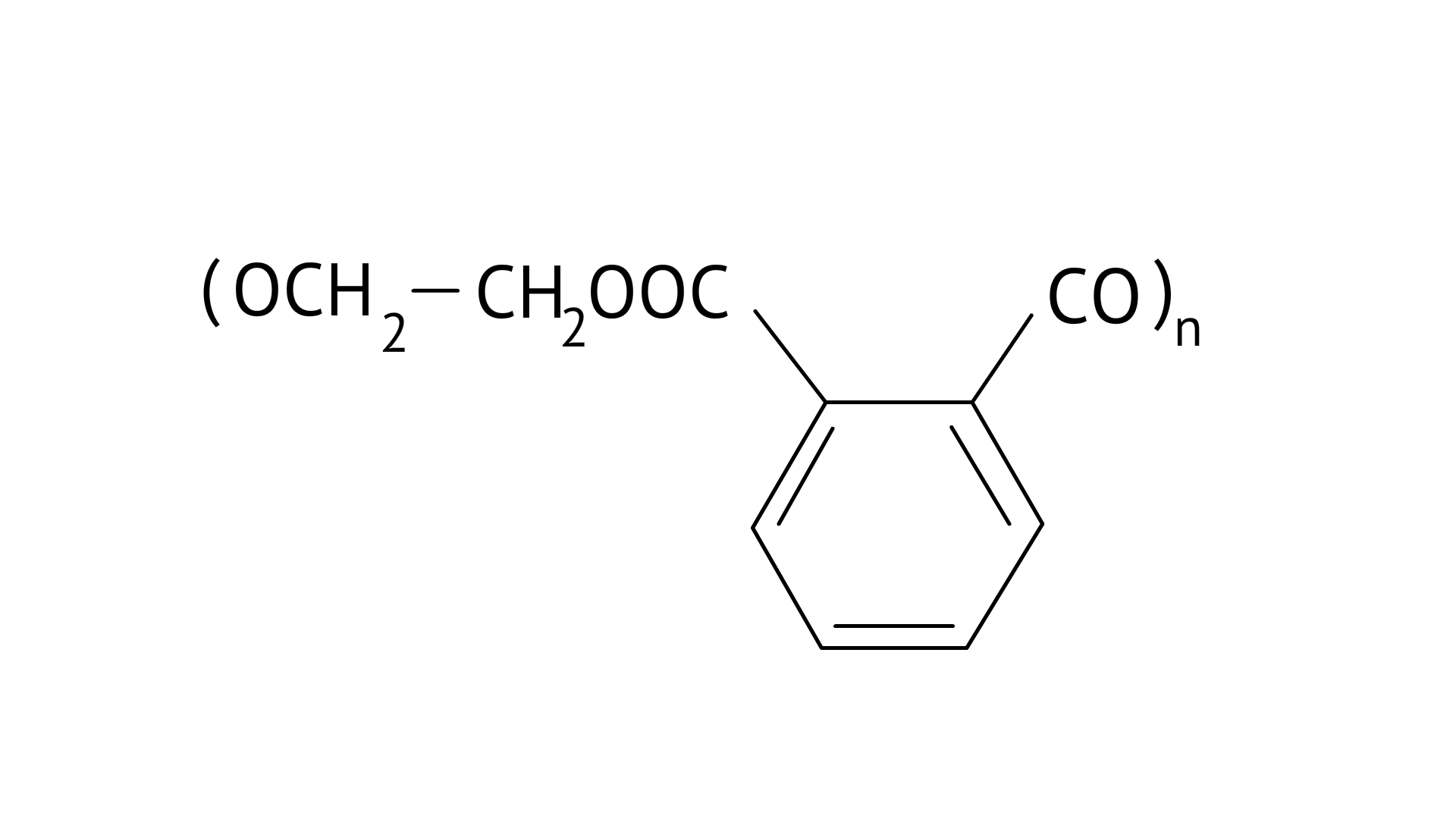
(B)
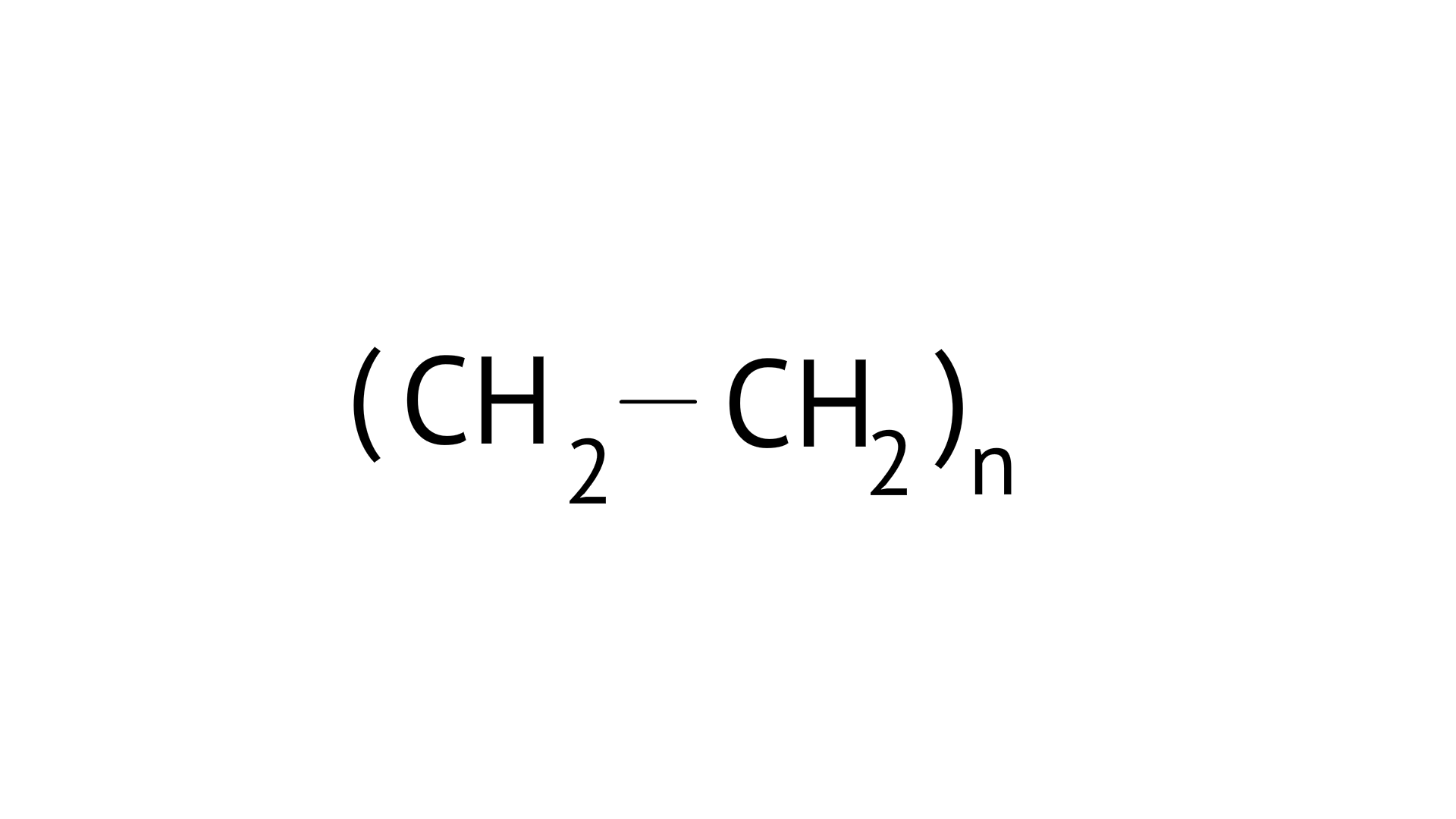
(C)
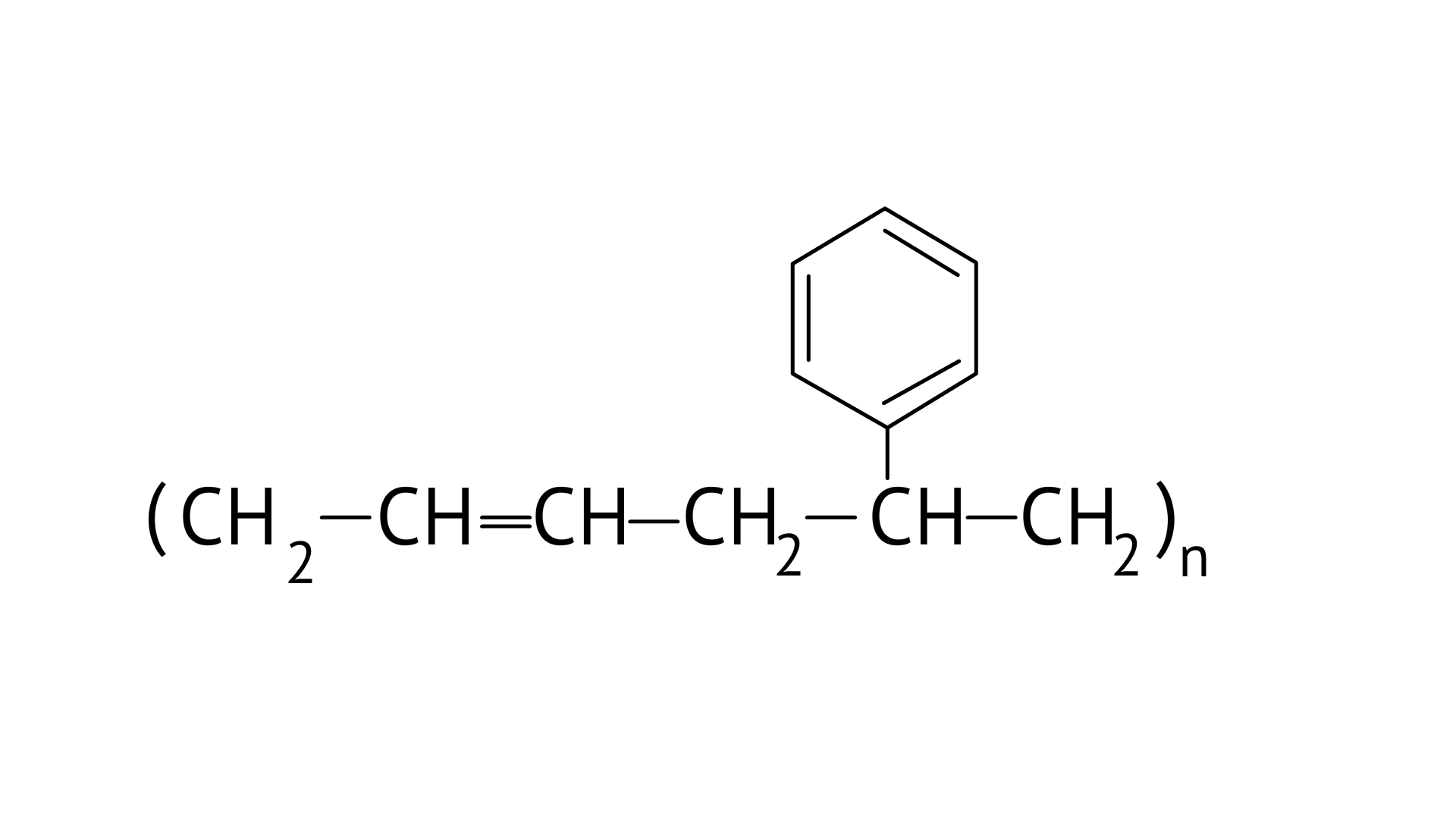
(D)
Ans: Option (A)
Explanation:
Dacron or Terylene is formed by the condensation polymerization of ethylene glycol and phthalic acid with the elimination of water molecules. The structure of Terylene is,
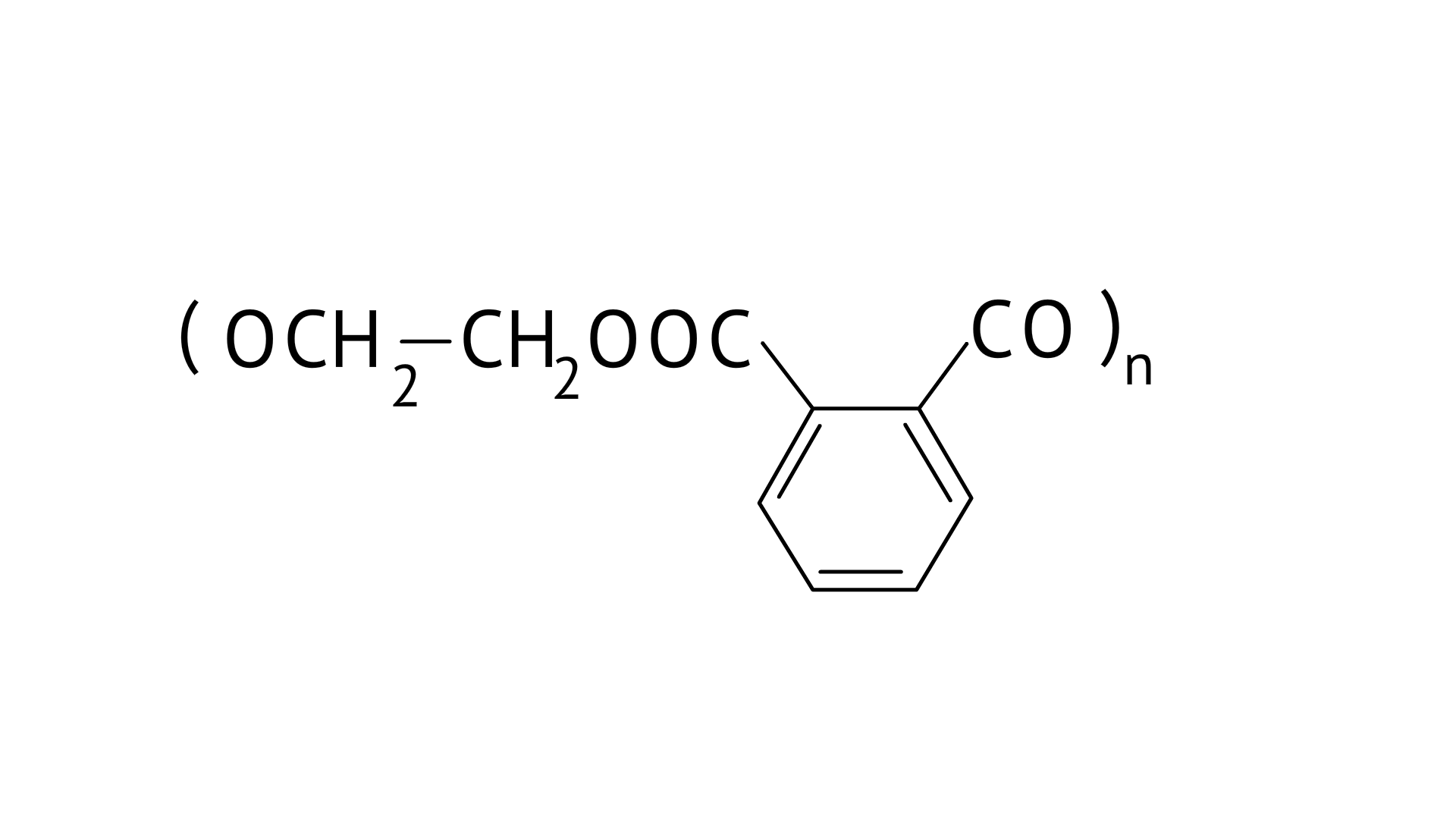
6. Which of the following statements is not true about low density polythene?
(A) Tough
(B) Hard
(C) Poor conductor of electricity
(D) Highly branched structure
Ans: Option (B)
Explanation:
Low-density polythene is obtained from the polymerization of ethane under high pressure and temperature. Low-density polyethylene is tough, a poor conductor of electricity and has a highly branched structure.
Low-density polythene is not hard because branched structure prevents stacking and thus, reduces the intermolecular force of attraction.
So, option (B) is correct.
7.
(A)
(B)

(C)
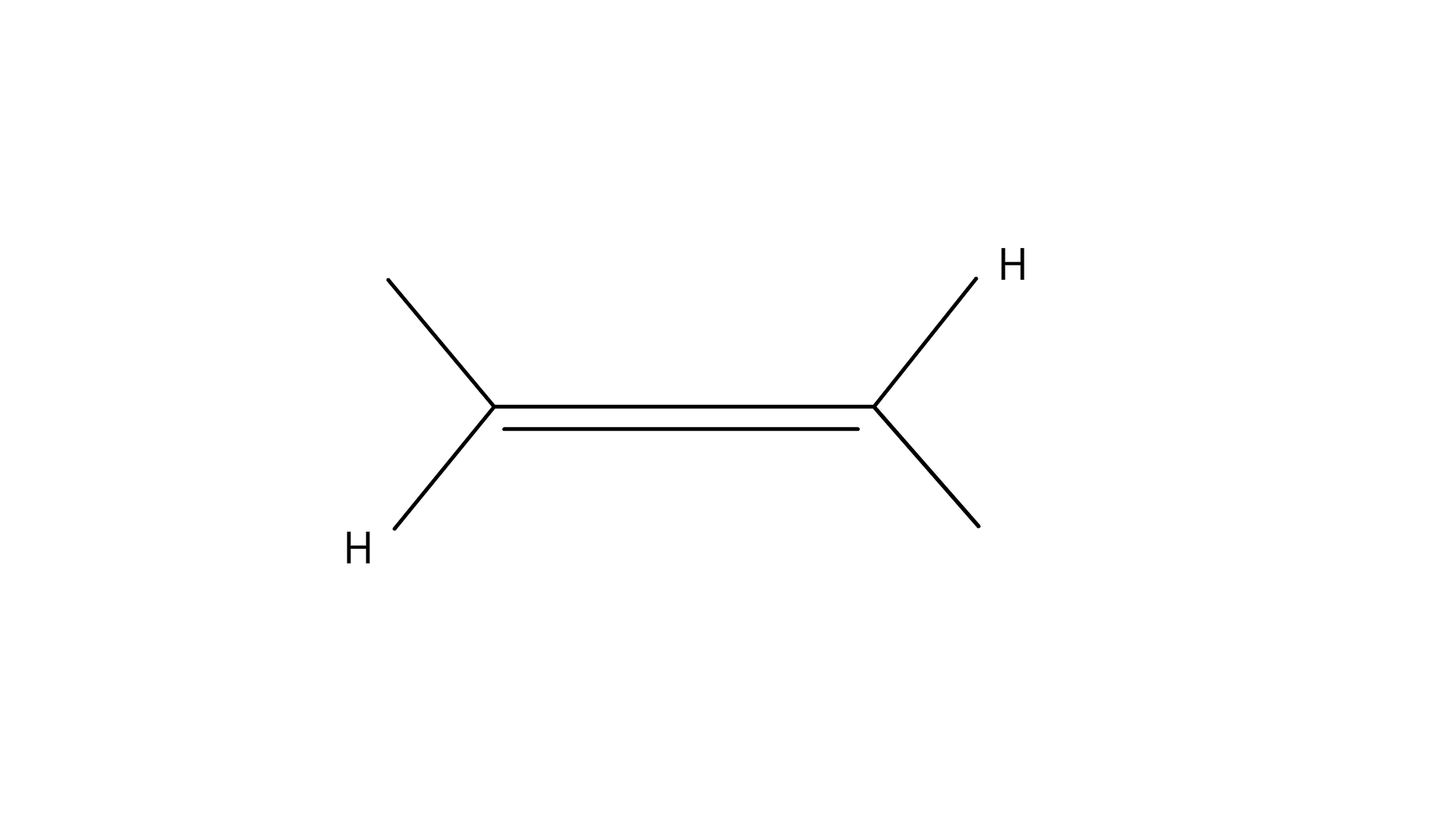
(D)
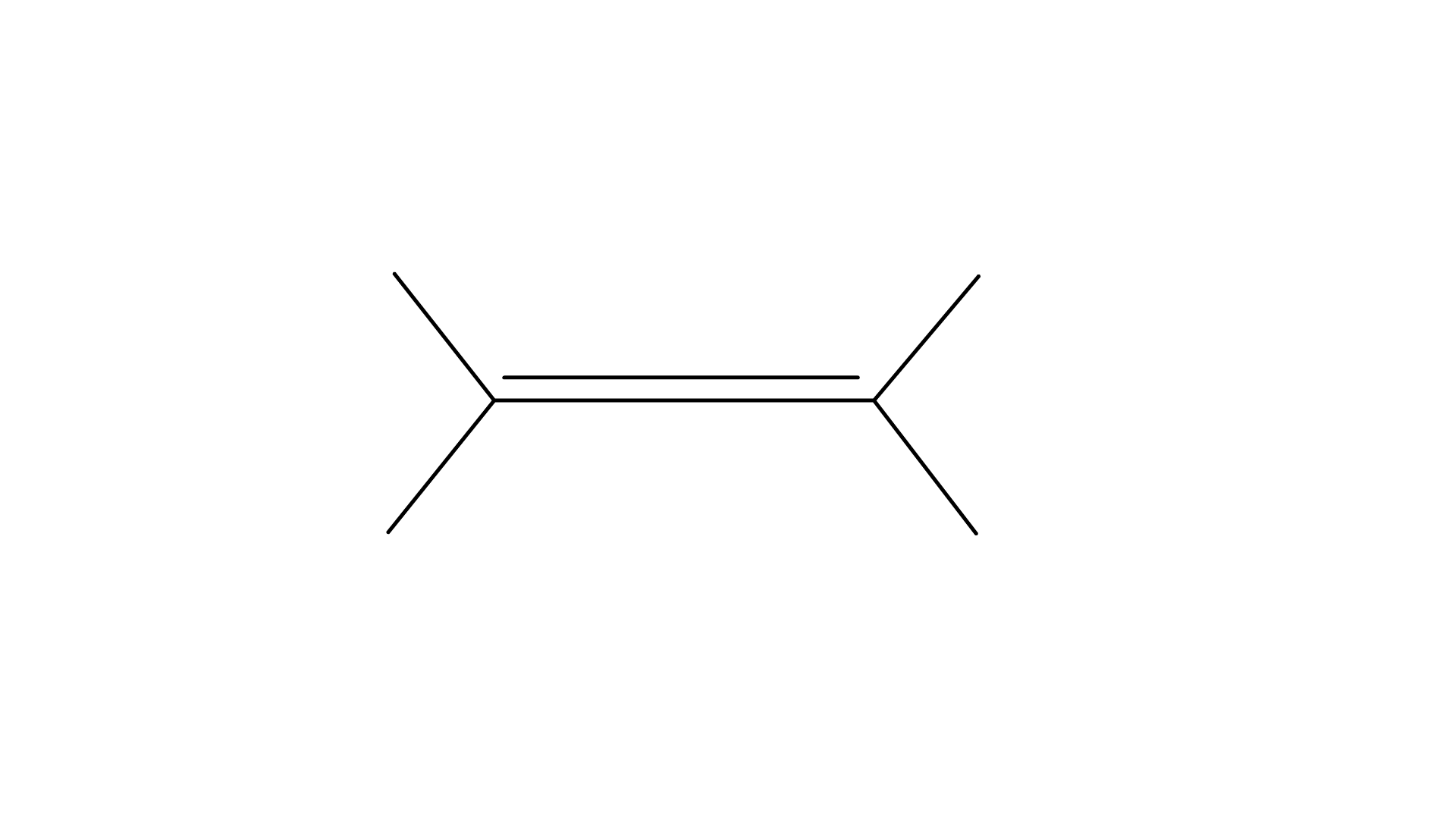
Ans: Option (A)
Explanation:
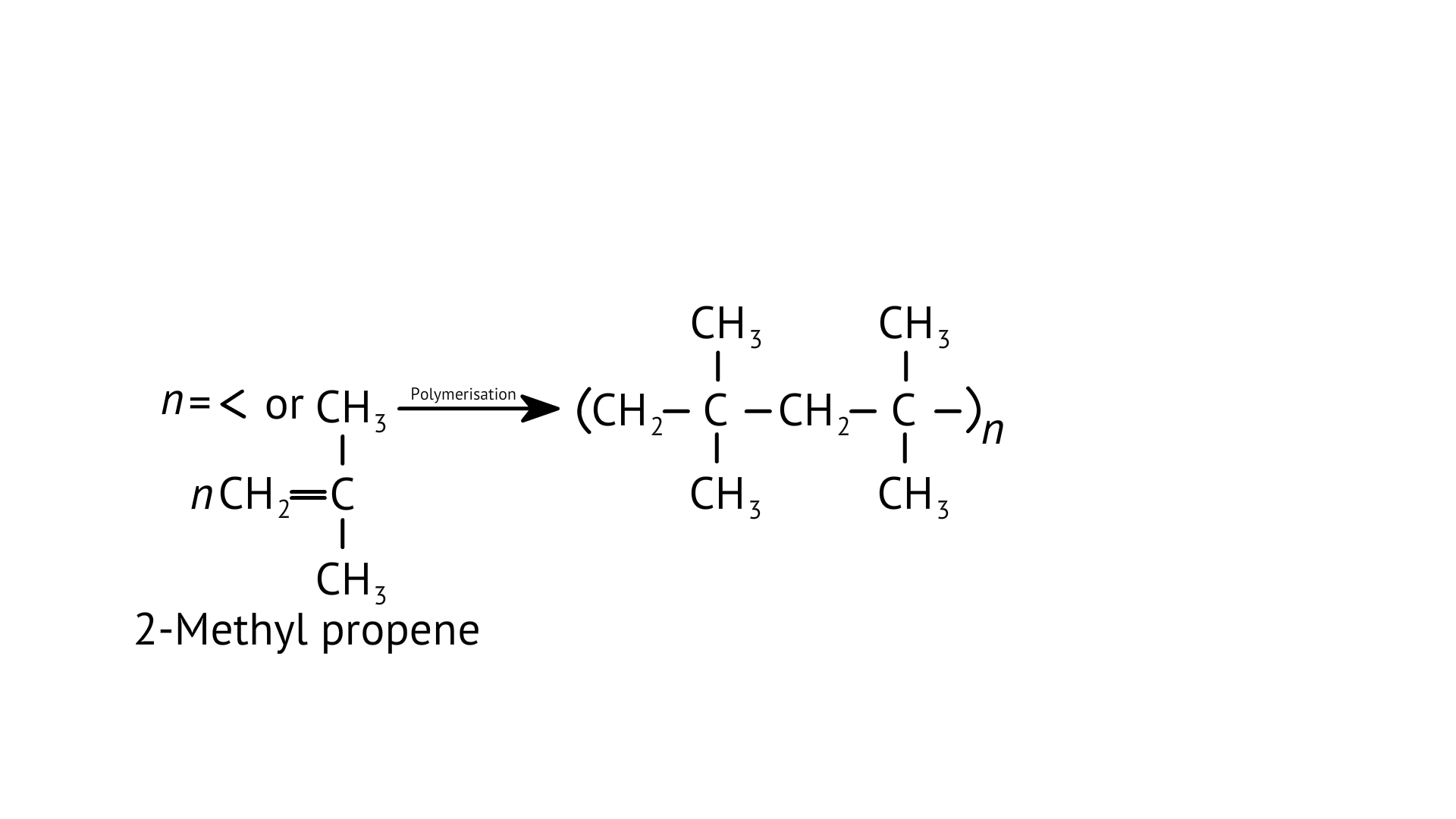
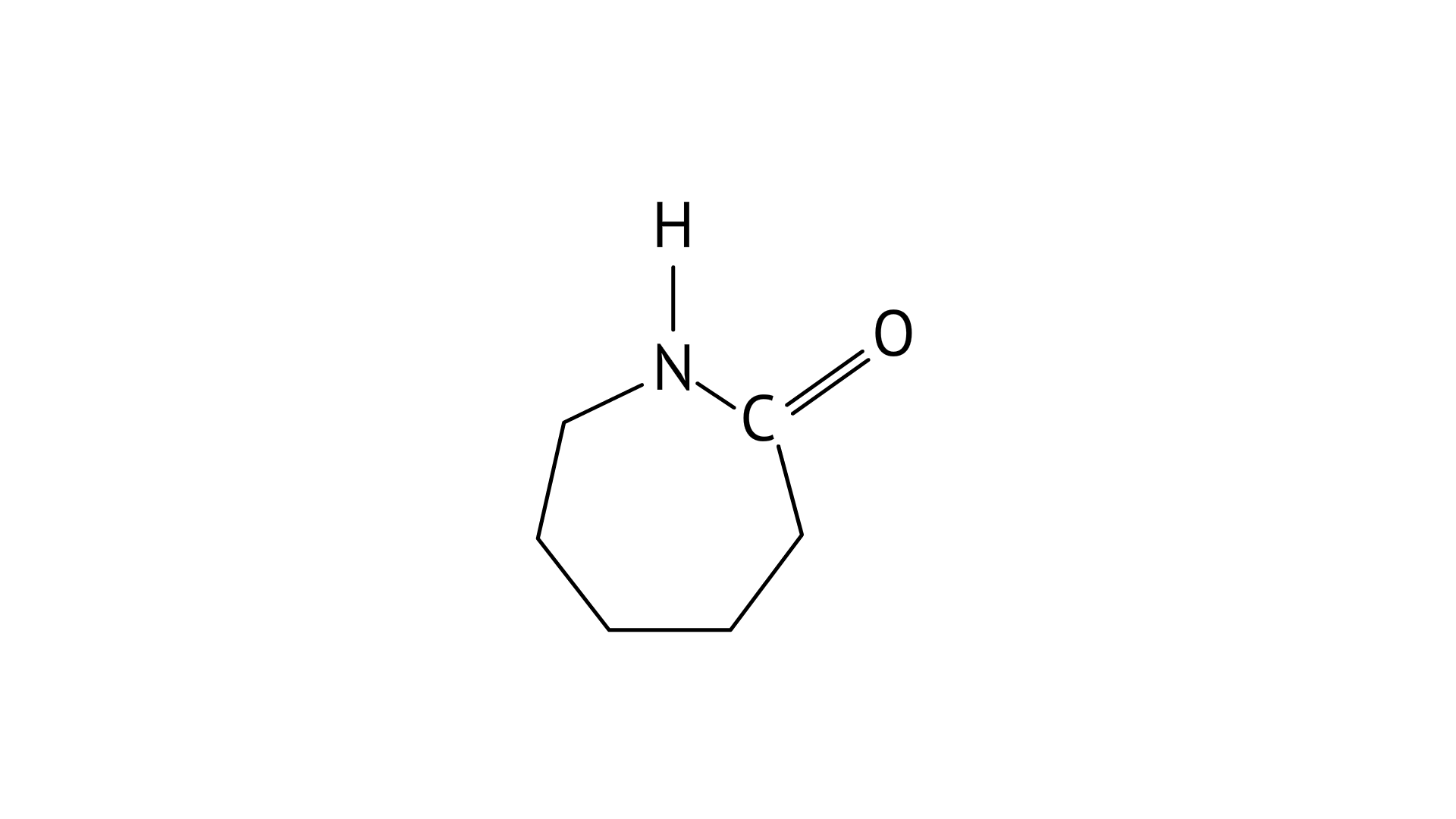
8. Which of the following polymer can be formed by using the following monomer unit?
(A) Nylon-6,6
(B) Nylon 2–nylon 6
(C) Melamine polymer
(D) Nylon-6
Ans: Option (D)
Explanation:
The given structure is caprolactam. Nylon-6 is prepared from the monomer caprolactam. It is obtained by heating caprolactam with water at a high temperature.
II. Multiple Choice Questions (Type-II)
9. Which of the following polymers need at least one di ene monomer for their preparation?
(A) Dacron
(B) Buna-S
(C) Neoprene
(D) Novolac
Ans: Option (B) and (C)
Explanation:
Buna-S and neoprene are the polymers that needs at least one diene monomeric unit. Buna- S is a polymer formed from the polymerization of 1,3-butadiene and styrene. While, Neoprene is formed by the polymerisation of chloroprene.
10. Which of the following are characteristics of thermosetting polymers?
(A) Heavily branched cross linked polymers.
(B) Linear slightly branched long chain molecules
(C) Become infusible on moulding so cannot be reused.
(D) Soften on heating and hardened on cooling, can be reused.
Ans: Option (A) and (C)
Explanation:
Thermosetting polymers are cross linked polymers. These polymers on heating undergo cross linking in moulds. They become infusible on heating so cannot be reused again.
11. Which of the following polymers are thermoplastic?
(A) Teflon
(B) Natural rubber
(C) Neoprene
(D) Polystyrene
Ans: Option (A) and (D)
Explanation:
Thermoplastic polymers are linear or slight branched-chain polymers. This type of polymer softens on heating and hardens on cooling. For example, Teflon and polystyrene are thermoplastic polymers.
So, options (A) and (D) are correct.
12. Which of the following polymers are used as fiber?
(A) Polytetrafluoroethane
(B) Polychloroprene
(C) Nylon
(D) Terylene
Ans: Option (C) and (D)
Explanation:
Fibers are thread-forming solids. They possess high tensile strength and high modulus. They have strong intermolecular forces of attraction like hydrogen bonding. Nylon and Terylene are used as fibers.
13. Which of the following are addition polymers?
(A) Nylon
(B) Melamine formaldehyde resin
(C) Orlon
(D) Polystyrene
Ans: Option (A) and (D)
Explanation:
The formation of addition polymers is accomplished through the repeated addition of monomer molecules with double or triple bonds, such as polythene from ethene and polypropene from propene. Homopolymers are addition polymers formed by the polymerisation of a single monomeric species.
14. Which of the following polymers are condensation polymers?
(A) Bakelite
(B) Teflon
(C) Butyl rubber
(D) Melamine formaldehyde resin
Ans: Option (A) and (D)
Explanation:
Condensation polymers are formed by the addition of more than one monomeric unit. Bakelite and Melamine formaldehyde resin are condensation polymers. Bakelite is a polymer formed from phenol and formaldehyde. Melamine formaldehyde resin is a polymer formed from melamine and formaldehyde.
15. Which of the following monomers form biodegradable polymers?
(A) 3-hydroxybutanoic acid+ 3-hydroxypentanoic acid
(B) Glycine+Aminocaproic acid
(C) Ethylene glycol + Phtahlic acid
(D) Caprolactum
Ans: Option (A) and (B)
Explanation:
The polymers that are not harmful to the environment are biodegradable polymers. PHBV(Poly beta-hydroxybutyrate co-beta-hydroxy valerate) is a biodegradable polymer. It undergoes bacterial degradation in the environment. PHBV is obtained by the polymerization of 3-hydroxybutyric acid and 3-hydroxypentanoic acid.
Nylon-2,Nylon-6 polymers are also biodegradable. It is obtained by the polymerization of glycine and aminocaproic acid.
16. Which of the following are examples of synthetic rubber?
(A) Polychloroprene
(B) Polyacrylonitrile
(C) Buna-N
(D) cis-polyisoprene
Ans: Option (A) and (C)
Explanation:
Synthetic rubbers are either homopolymers or copolymers of 1,3-butadiene or its derivatives with another unsaturated monomer. Polychloroprene or neoprene and Buna-N are examples of synthetic rubber.
17. Which of the following polymers can have strong intermolecular forces?
(A) Nylon
(B) Polystyrene
(C) Rubber
(D) Polyesters
Ans: Option (A) and (D)
Explanation:
Polyamides: These polymers with amide linkages are important examples of synthetic fibers and are known as nylons.
Polyesters: These are the polycondensation products of dicarboxylic acids and diols.
Dacron or terylene is the most well-known example of a polyester.
18. Which of the following polymers have vinylic monomer units?
(A) Acrilan
(B) Polystyrene
(C) Nylon
(D) Teflon
Ans: Option (A), (B) and (D)
Explanation:
Polyacrylonitrile is formed by the addition polymerisation of acrylonitrile in the presence of a peroxide catalyst. Polyacrylonitrile is used as a wool substitute in the manufacture of commercial fibers such as orlon or acrilan.
Polystyrene has the monomer unit styrene.
Teflon is made by heating tetrafluoroethene at high pressures with a free radical or persulphate catalyst.
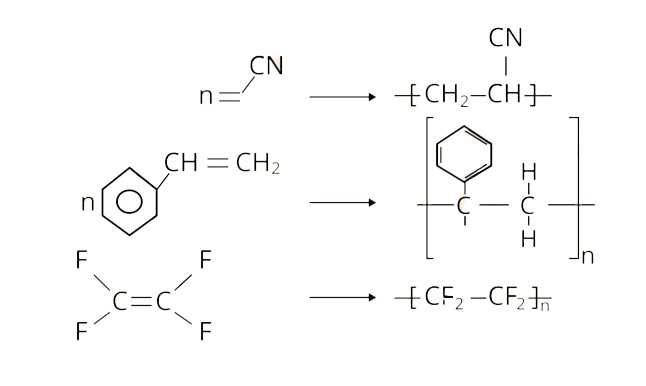
19. Vulcanisation makes rubber ______________.
(A) more elastic
(B) soluble in inorganic solvent
(C) crystalline
(D) more stiff
Ans: Option (A) and (D)
Explanation:
Vulcanisation is a process used to improve the physical properties of natural rubber. This method involves heating a mixture of raw rubber, sulphur, and an appropriate additive to temperatures ranging from 373 K to 415 K. Sulphur forms cross links at the reactive sites of double bonds during vulcanisation, stiffening the rubber.
Short Answer Type
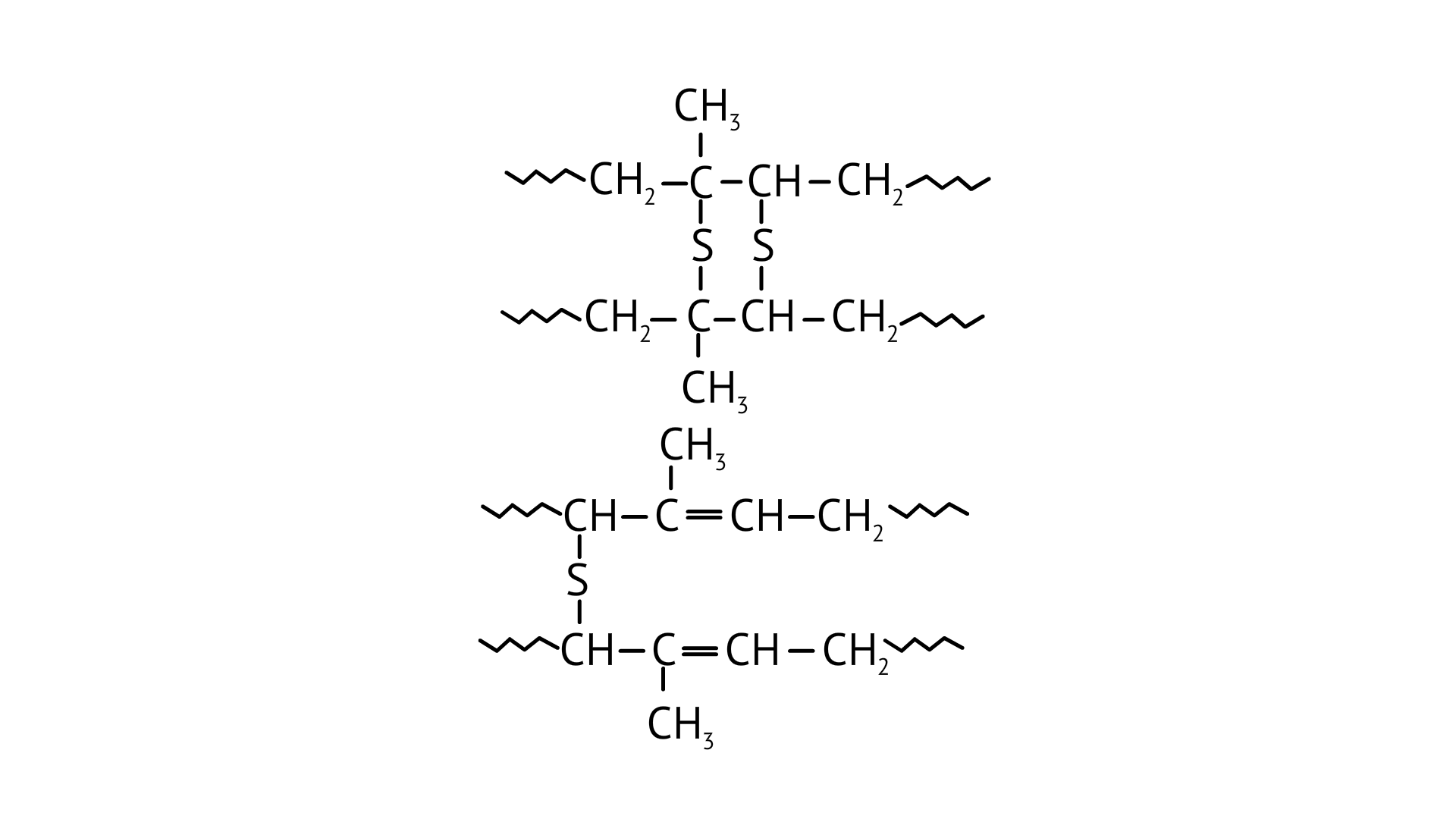
20. A natural linear polymer of 2-methyl-1, 3-butadiene becomes hard on treatment with sulphur between 373 to 415K and —S—S— bonds are formed between chains. Write the structure of the product of this treatment?
Ans: While manufacturing tyre rubber, 5 % sulfur is used as a crosslinking agent. A natural linear polymer of 2-methyl-1, 3-butadiene becomes hard on treatment with sulfur and forms vulcanised rubber. The structure of the product formed is,
21. Identify the type of polymer.
—A—A—A—A—A—A—
Ans: Addition polymers formed by the polymerization of a single monomeric species are known as homopolymers. The above-given polymer is a homopolymer.
22. Identify the type of polymer.
—A—B—B—A—A—A—B—A—
Ans: Addition polymers formed by the polymerization from two different monomers are co-polymers. The above-given polymer is a co-polymer. For example Buna-N, Buna-S.
23. Out of chain growth polymerization and step growth polymerisation, in which type will you place the following.
$(-\mathrm{A}-)_{m}+(-\mathrm{A}-)_{n} \rightarrow(-\mathrm{A}-\mathrm{A}-)_{m+n}$
Ans: This is a type of chain-growth polymerization. The molecules of the same monomer or different monomer are added together to form this type of polymer.
24. Identify the type of polymer given in the following figure.
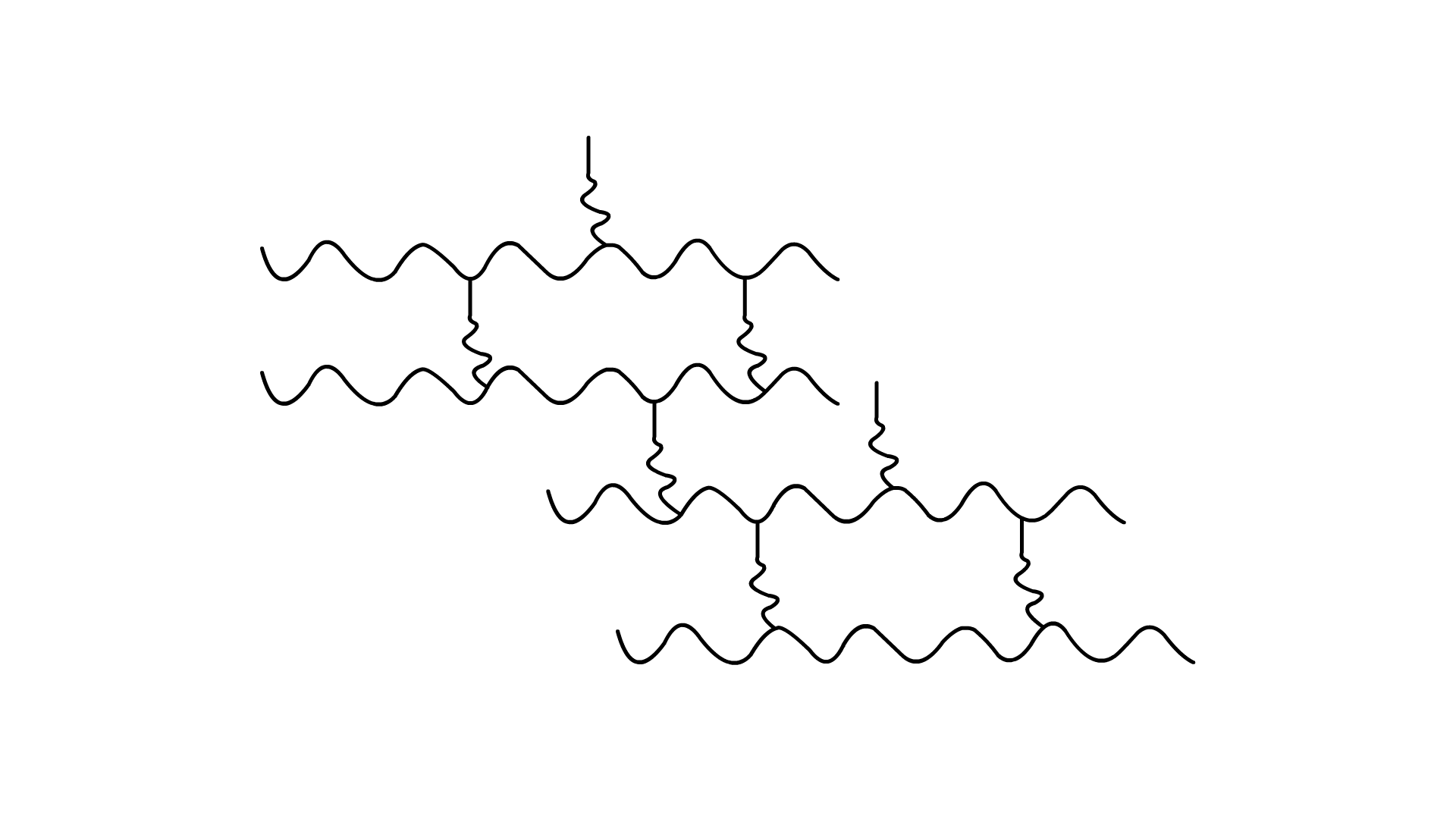
Ans: The polymer shown above is cross-linked or network polymer. They are formed from bi or tri-functional monomers that contain strong covalent bond between polymer chains.
25. Identify the polymer given below :
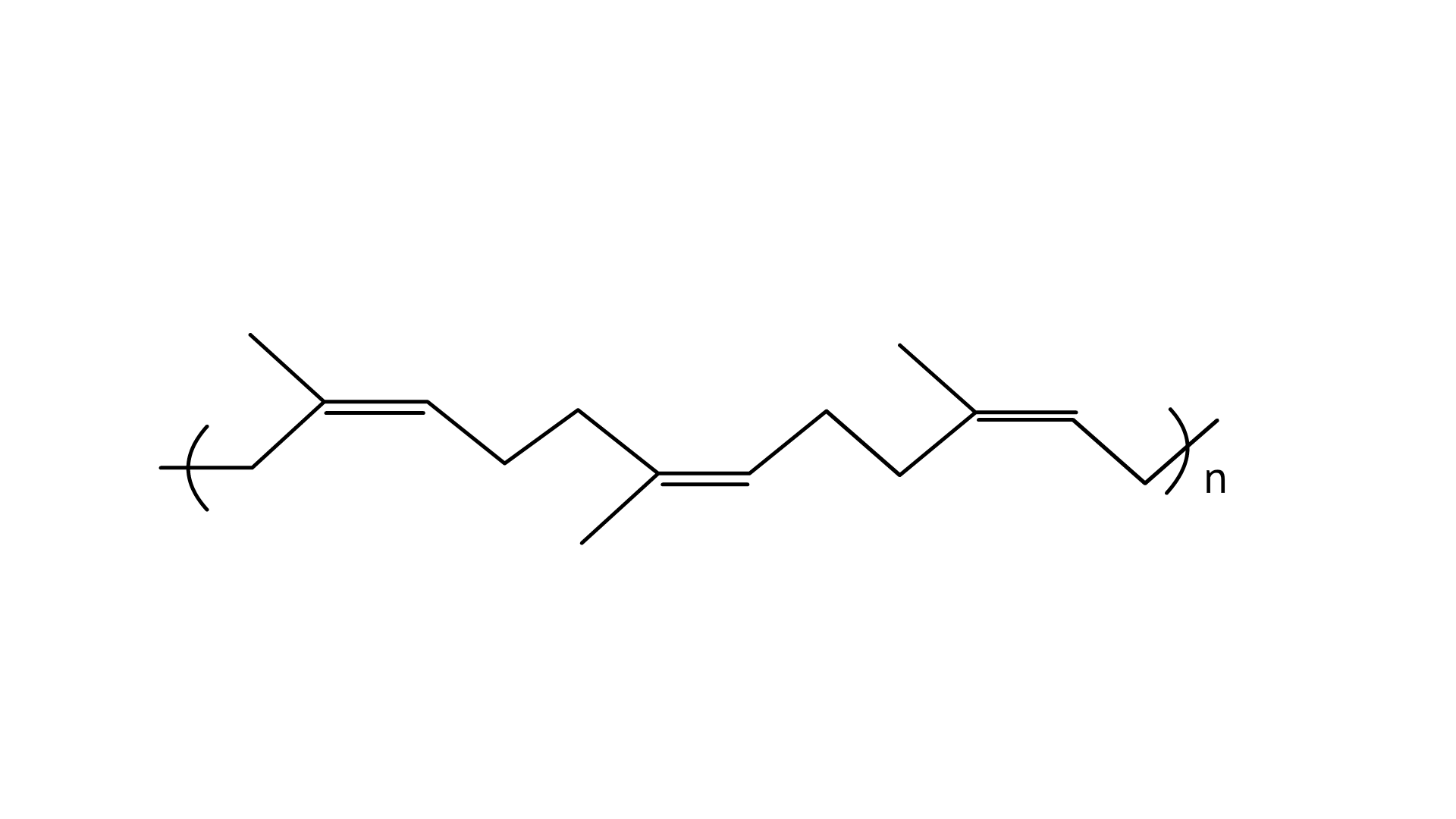
Ans: The cis-polyisoprene molecule is made up of several chains that are held together by weak van der Waals interactions and has a coiled structure. Thus, it can be stretched like a spring and exhibits elastic properties.
26. Why are rubbers called elastomers?
Ans: Rubber is a natural polymer. It is also known as an elastomer due to its elastic properties. It is manufactured from rubber latex.
27. Can an enzyme be called a polymer?
Ans: Enzymes are biocatalysts that are present in the body. They are basically proteins, and they are also considered as polymers.
28. Can nucleic acids, proteins and starch be considered as step growth polymers?
Ans: Yes, nucleic acids, proteins, and starch can be considered step growth polymers because this type of polymerization involves a condensation reaction between two monomers. Loss of simple molecules such as water, alcohol results in this type of polymerization and it results in the formation of high molecular mass polymers.
29. How is the following resin intermediate prepared and which polymer is formed by this monomer unit?
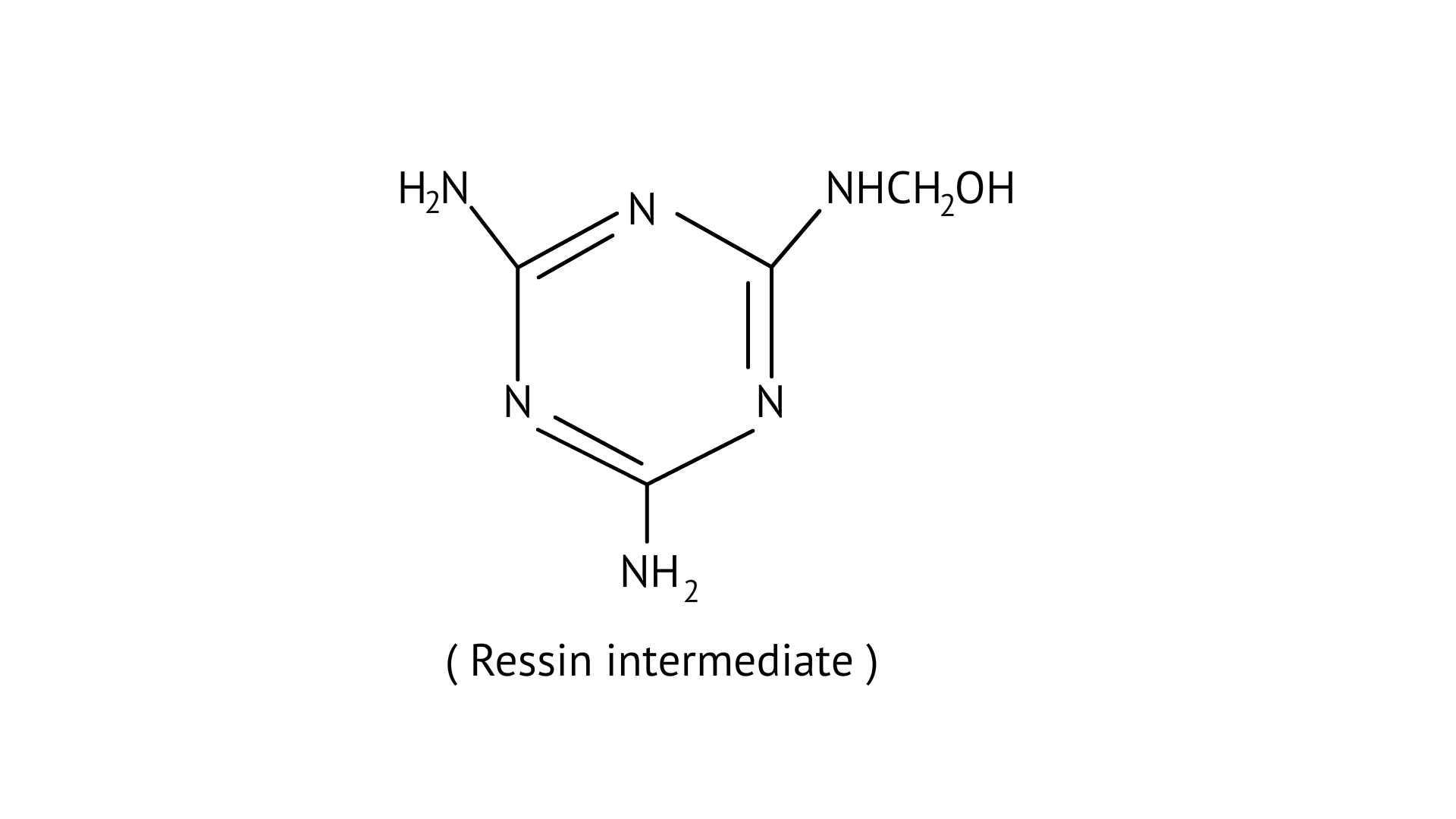
Ans: Melamine formaldehyde polymer is formed by the condensation polymerization of melamine and formaldehyde. This is resin intermediate.
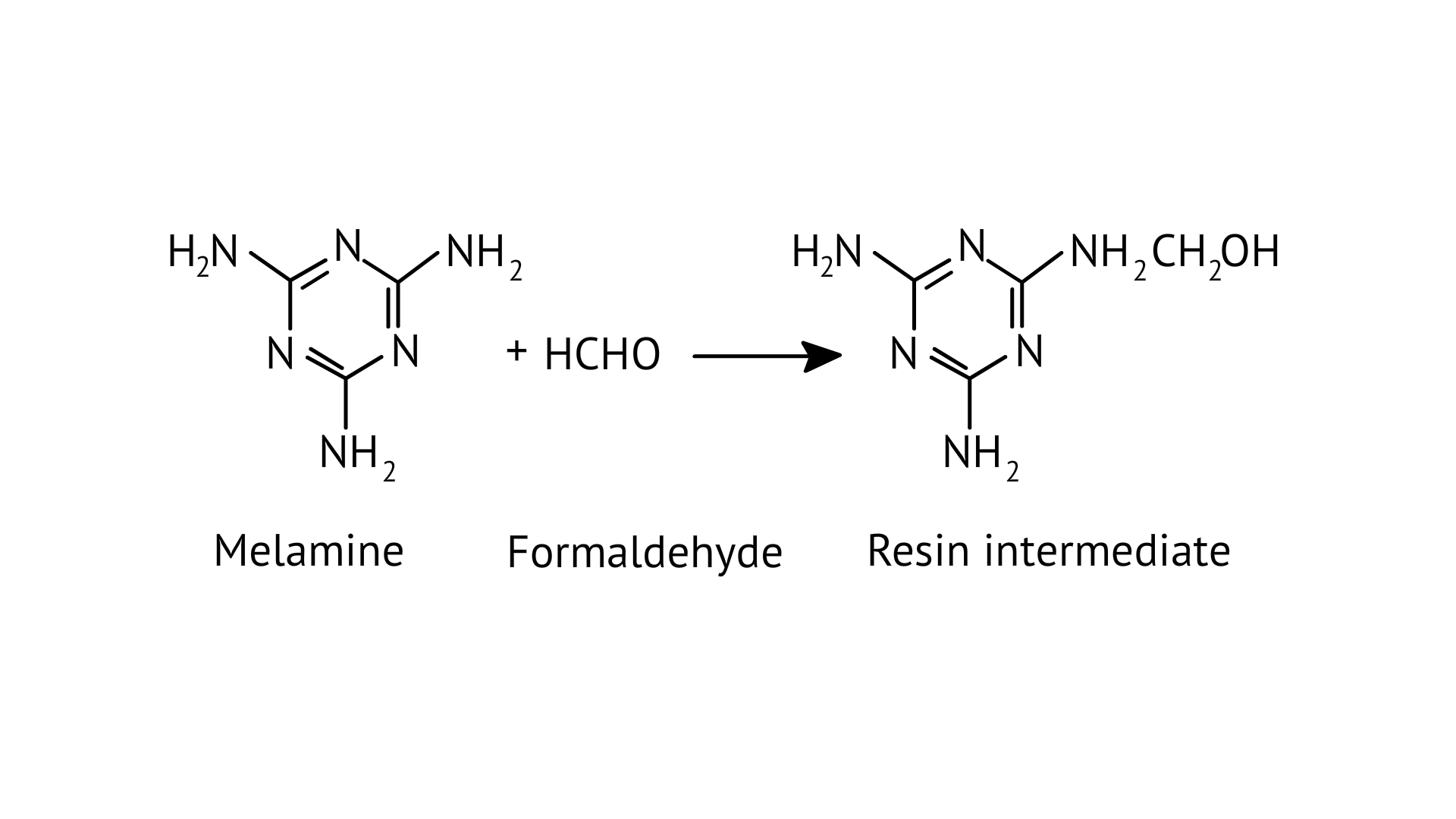
30. To have practical applications why are cross-links required in rubber?
Ans: A vulcanisation process is used to improve the physical properties of natural rubber. This process consists of heating a mixture of raw rubber with sulphur and an appropriate additive at a temperature range between 373 K to 415 k. Sulphur forms cross links at the reactive sites of double bonds during vulcanization, stiffening the rubber.
31. Why does cis-polyisoprene possess elastic property?
Ans: Cis-polyisoprene has a coiled structure which consists of various chains held together by weak Van der Waals force of attraction. It can be stretched like a spring and thus, has elastic properties.
32. What is the structural difference between HDP and LDP? How does the structure account for different behaviour and nature, hence the use of a polymer?
Ans: LDP: The polymerization of ethene under high pressure and temperature in the presence of peroxide catalyst gives low-density polythene. It is obtained through free radical addition and has a high branched structure. It is chemically inert and is a poor conductor of electricity. It is used for manufacturing toys, bottles.
HDP: When addition polymerization of ethene takes place in a hydrocarbon solvent in the presence of a catalyst such as triethylaluminium and Ziegler-Natta catalyst at high temperature and pressure then high-density polythene is formed. It consists of linear molecules. It is also chemically inert. It is used for manufacturing buckets, dustbins.
33. What is the role of benzoyl peroxide in addition polymerisation of alkenes? Explain its mode of action with the help of an example.
Ans: Free radicals are generated from benzoyl peroxide. Alkanes, dienes are polymerized in the presence of free radical inhibitors like benzoyl peroxide, acetyl peroxide. For example, benzoyl peroxide inhibitor is used for the polymerization of ethene to polythene. First, phenyl peroxide radicals are formed which are added to the double bond of ethene generating a new and larger free radical.
34. Which factor imparts crystalline nature to a polymer like nylon?
Ans: Polymers that possess amide linkage are termed as nylon. Fibers have a strong intermolecular force of attraction like hydrogen bonding. It leads to the close packing of the structure which imparts the crystalline nature.
35. Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Ans: Urea-formaldehyde resin is used in laminated sheets. The monomer involved in the formation of urea formaldehyde resin is urea and formaldehyde.
36. Which type of biomolecules have some structural similarity with synthetic polyamides? What is this similarity?
Ans: Proteins have some similar structural similarities with synthetic polyamides. Proteins and polyamides both contain amide linkage.
37. Why should the monomers used in addition to polymerization through free radical pathway be very pure?
Ans: Monomers used in addition to polymerization through free radical pathway should be very pure because even the traces of impurities acts like inhibitors. It leads to the formation of polymers with a shorter chain lengths.
Matching Type
38. Match the polymer of column I with the correct monomer of column II.
Column I | Column II | |
i. High density polythene | a. Isoprene | |
ii. Neoprene | b. Tetrafluoroethene | |
iii. Natural Rubber | c. Chloroprene | |
iv. Teflon | d. Acrylonitrile | |
v. Acrilan | e. Ethene | |
Ans: (i) When addition polymerisation of ethene takes place in a hydrocarbon solvent in the presence of a catalyst such as triethylaluminium and Ziegler-Natta catalyst at high temperature and pressure then high density polythene is formed.
Thus, option (i) from column I is matched with (e) from column II.
(ii) Neoprene is formed by the free radical polymerization of chloroprene.
Thus, option (ii) from column I is matched with (c) from column II.
(iii) Natural rubber is a linear polymer of isoprene units.
Thus, option (iii) from column I is matched with (a) from column II.
(iv) Teflon is formed by heating tetrafluoroethylene at high pressure in the presence of free radical polymerization like persulfate catalyst.
Thus, option (iv) from column I is matched with (b) from column II.
(v) Acrilan is formed by the addition polymerization of acrylonitrile in the presence of a peroxide catalyst.
Thus, option (v) from a column I is matched with (d) from column II.
39. Match the polymers given in Column I with their chemical names given in Column II.
Column I | Column II |
i. Nylon-6 | a. Polyvinyl chloride |
ii. PVC | b. Polyacrylonitrile |
iii. Acrilan | c. Polycaprolactum |
iv. Natural Rubber | d. Low density polythene |
v. LDP | e. cis-polyisoprene |
Ans: (i) Nylon-6 is obtained by heating caprolactam with water at a high temperature.
Thus, option (i) from column I is matched with (c) from column II.
(ii).The chemical name of PVC is polyvinyl chloride.
Thus, option (ii) from column I is matched with (a) from column II.
(iii) Acrilan is formed by the addition polymerization of acrylonitrile in the presence of a peroxide catalyst.
Thus, option (iii) from column I is matched with (b) from column II.
(iv). Natural rubber is a linear polymer of isoprene units. It is also known as cis-polyisoprene.
Thus, option (iv) from column I is matched with (e) from column II.
(v) LDP is known as low density polythene. It is obtained through a free radical addition reaction.
Thus, option (v) from column I is matched with (d) from column II.
40. Match the polymers given in Column I with their commercial names given in Column II.
Column I | Column II |
i. Polyester of glycol and phthalic acid | a. Novolac |
ii. Copolymer of 1,3-butadiene and styrene | b. Glyptal |
iii. Phenol and formaldehyde resin | c. Buna-S |
iv. Polyester of glycol and terephthalic acid | d. Buna-N |
v. Copolymer of 1,3-butadiene and acrylonitrile | e. Dacron |
Ans: (i) Glyptal is formed by the condensation polymerization of polyester of glycol and phthalic acid.
Thus, option (i) from column I is matched with (b) from column II.
(ii) Buna-S is formed from the polymerization of styrene and 1,3-butadiene.
Thus, option (ii) from column I is matched with (c) from column II.
(iii) Novolac is formed as an intermediate in the formation of Bakelite. Bakelite is formed from formaldehyde and phenol.
Thus, option (iii) from column I is matched with (a) from column II.
(iv) Dacron is obtained from the polymerization of polyester of glycol and terephthalic acid.
Thus, option (iv) from column I is matched with (e) from column II.
(v) Buna-N is formed from the polymerization of 1,3-butadiene and acrylonitrile.
Thus, option (v) from column I is matched with (d) from column II.
41. Match the polymers given in Column I with their main applications given in Column II
Column I | Column II |
i. Bakelite | a. Unbreakable crockery |
ii. LDP | b. Non-stick cookwares |
iii. Melamine-formaldehyde resin | c. Packaging material for shock absorbance |
iv. Nylon-6 | d. Electrical switches |
v. Poly tetrafluoro ethane | e. Squeeze bottles |
vi. Polystyrene | f. Tyre, cords |
Ans: (i) Bakelite is used in making combs, electrical switches.
Thus, option (i) from column I is matched with (d) from column II.
(ii) LDP is used in the manufacture of squeezes bottles, toys etc.
Thus, option (ii) from column I is matched with (e) from column II.
(iii) Melamine formaldehyde resin is used in the manufacture of unbreakable crockery.
Thus, option (iii) from column I is matched with (a) from column II.
(iv) Nylon-6 is used in the manufacture of tyre, cords.
Thus, option (iv) from column I is matched with (f) from column II.
(v) Poly tetrafluoro ethane is used in the manufacture of non stick cookware items like pan, cooker.
Thus, option (v) from column I is matched with (b) from column II.
(vi) Polystyrene is used in the manufacture of packaging material.
Thus, option (vi) from column I is matched with (c) from column II.
42. Match the polymers given in Column I with the preferred mode of polymerisation followed by their monomers.
Column I | Column II |
i. Nylon-6,6 | a. Free radical polymerisation |
ii. PVC | b. Ziegler-Natta polymerisation or coordination polymerisation |
iii. HDP | c. Anionic polymerisation |
d. Condensation polymerisation |
Ans:
(i) Nylon-6,6 is obtained by the condensation polymerization of hexamethylenediamine with adipic acid at high pressure and temperature.
Thus, option (i) from column I is matched with (d) from column II.
(ii) Poly vinyl chloride is obtained by the free radical polymerisation.
Thus, option (ii) from column I is matched with (a) from column II.
(iii) When addition polymerisation of ethene takes place in a hydrocarbon solvent in the presence of a catalyst such as triethylaluminium and Ziegler-Natta catalyst at high temperature and pressure then high density polythene is formed.
Thus, option (iii) from column I is matched with (b) from column II.
43. Match the polymers given in Column I with the type of linkage present in them given in Column II.
Column I | Column II |
i. Terylene | a. Glycosidic linkage |
ii. Nylon | b. Ester linkage |
iii. Cellulose | c. Phosphodiester linkage |
iv. Protein | d. Amide linkage |
v. RNA |
Ans:
(i) Terylene or Dacron have ester linkage.
Thus, option (i) from column I is matched with (b) from column II.
(ii) Nylon have amide linkage present in them.
Thus, option (ii) from column I is matched with (d) from column II.
(iii) Cellulose is stored by plants. It contains glycosidic linkage.
Thus, option (iii) from column I is matched with (a) from column II.
(iv) Proteins contains amide linkage.
Thus, option (iv) from column I is matched with (d) from column II.
(v) RNA contains phosphate, sugar and base. They contains phosphodiester linkage.
Thus, option (v) from column I is matched with (c) from column II.
44. Match materials given in Column I with the polymers given in Column II.
Column I | Column II |
i. Natural rubber latex | a. Nylon |
ii. Wood laminates | b. Neoprene |
iii. Ropes and fibers | c. Dacron |
iv. Polyester fabric | d. Melamine-formaldehyde resins |
v. Synthetic rubber | e. Urea-formaldehyde resins |
vi. Unbreakable crockery | f. cis-polyisoprene |
Ans:
(i) Natural rubber is a linear polymer of isoprene units. It is also known as cis-polyisoprene.
Thus, option (i) from column I is matched with (f) from column II.
(ii) Urea formaldehyde resin is used in the manufature of wood laminates.
Thus, option (ii) from column I is matched with (e) from column II.
(iii) Nylon is used in making ropes and fibres.
Thus, option (iii) from column I is matched with (a) from column II.
(iv) Dacron is obtained from the polymerization of polyester of glycol and terphthalic acid.
Thus, option (iv) from column I is matched with (c) from column II.
(v) Neoprene is a type of synthetic rubber. It is formed by the free radical polymerisation of chloroprene.
Thus, option (v) from column I is matched with (b) from column II.
(vi) Melamine formaldehyde resin is used in the manufacture of unbreakable crockery.
Thus, option (vi) from column I is matched with (d) from column II.
45. Match the polymers given in Column I with their repeating units given in Column II.
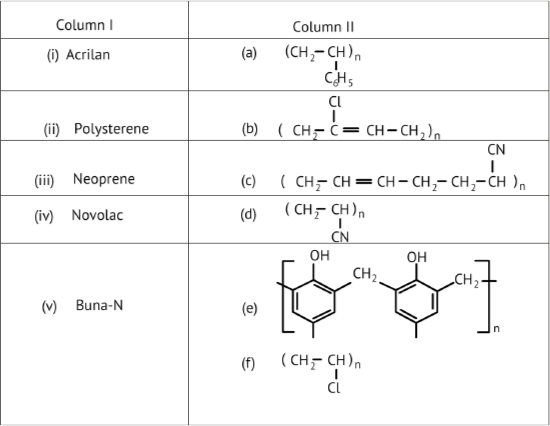
Ans: (i) Acrilan is formed by the addition polymerization of acrylonitrile in the presence of peroxide catalyst.
Thus, option (i) from column I is matched with (d) from column II.
(ii) Polystyrene is formed by the polymerization of styrene monomer.
Thus, option (ii) from column I is matched with (a) from column II.
(iii) Neoprene is a type of synthetic rubber. It is formed by the free radical polymerisation of chloroprene.
Thus, option (iii) from column I is matched with (b) from column II.
(iv) Novolac is formed as an intermediate in the formation of Bakelite. Bakelite is formed from formaldehyde and phenol.
Thus, option (iv) from column I is matched with (e) from column II.
(v) Buna-N is formed from the polymerization of 1,3-butadiene and acrylonitrile.
Thus, option (v) from column I is matched with (c) from column II.
Assertion and Reason Type
Note: In the following questions a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices.
(i) Assertion and reason both are correct statement but reason does not explain assertion.
(ii) Assertion and reason both are correct statements and reason explains the assertion.
(iii) Both assertion and reason are wrong statement.
(iv) Assertion is correct statement and reason is wrong statement.
(v) Assertion is wrong statement and reason is correct statement.
46. Assertion : Rayon is a semi synthetic polymer and is taken as a better choice than cotton fabric. Reason : Mechanical and aesthetic properties of cellulose can be improved by acetylation.
Ans: (ii) Assertion and reason both are correct statements and reason explains the assertion.
Explanation: Rayon is a semi synthetic polymer. It is preferred over cotton fabric because its properties are improved by acetylation.
47. Assertion : Most of the Synthetic polymers are not biodegradable. Reason : Polymerisation process induces toxic character in organic molecules.
Ans: (iv) Assertion is correct statement and reason is wrong statement. (v) Assertion is wrong statement and reason is correct statement.
Explanation: Most of the Synthetic polymers are not biodegradable. A large number of the polymers are resistant to the degradation process of the environment and therefore, are responsible for the accumlation of polymeric solid waste material.
48. Assertion : Olefinic monomers undergo addition polymerisation. Reason : Polymerisation of vinylchloride is initiated by peroxides/ persulphates.
Ans: (i) Assertion and reason both are correct statement but reason does not explain assertion.
Explanation: Olefinic monomers undergo addition polymerisation. The molecules of the same monomer or different monomer are added together to form this type of polymer. This mode of polymerisation leads to an increase in chain length.
49. Assertion : Polyamides are best used as fibres because of high tensile strength. Reason : Strong intermolecular forces (like hydrogen bonding within polyamides) lead to close packing of chains and increase the crystalline character, hence, provide high tensile strength to polymers.
Ans: (ii) Assertion and reason both are correct statements and reason explains the assertion.
Explanation: Polyamides are best used as fibres because of high tensile strength. They have strongest intermolecular forces of attraction like hydrogen bonding. It lead to close packing of chains and increase the crystalline character.
50. Assertion : For making rubber synthetically, isoprene molecules are polymerised. Reason : Neoprene (a polymer of chloroprene) is a synthetic rubber.
Ans: (v) Assertion is wrong statement and reason is correct statement.
Explanation: Synthetic rubber is a type of rubber polymer that are capable of getting stretched to twice of its length. They are homopolymers of either 1,3-butadiene derivatives or copolymers of butadiene. Polychloroprene or neoprene is a type of synthetic rubber.
51. Assertion : Network polymers are thermosetting. Reason : Network polymers have high molecular mass.
Ans: (i) Assertion and reason both are correct statements but reason does not explain assertion.
Explanation: Thermosetting polymers are cross linked polymers. They are highly branched polymers with high molecular masses. These polymers on heating undergo cross linking in moulds. They become infusible on heating.
52. Assertion : Polytetrafluoroethene is used in making non-stick cookwares. Reason : Fluorine has highest electronegativity.
Ans: (i) Assertion and reason both are correct statement but reason does not explain assertion.
Explanation: Tetrafluoroethene on heating with a free radical at high pressure forms teflon. It s chemically inert and resistant. It is used in making oil seals and gaskets ussed for non stick surfaces.
Long Answer Type
53. Synthetic polymers do not degrade in the environment for a long time. How can biodegradable synthetic polymers be made. Differentiate between biopolymers and biodegradable polymers and give examples of each type.
Ans: 1.Poly-hydroxybutyrate-co-β-hydroxyvalerate (PHBV)
It's made by copolymerization 3-hydroxybutanoic acid and 3-hydroxypentanoic acid. PHBV is used in specialty packaging, orthopaedic devices, and controlled drug release. In the environment, PHBV is degraded by bacteria.
2. Nylon 2-Nylon 6: It is a biodegradable alternating polyamide copolymer of glycine and amino caproic acid.
Biopolymers are natural polymers found in plants and animals such as protein, fat, cellulose, and so on.
Biodegradable polymers are polymers that contain functional groups that are similar to those found in biopolymers. They are not resistant to the processes of environmental degradation.
54. Differentiate between rubbers and plastics on the basis of intermolecular forces.
Ans: In rubber, the polymer chain are held together by weakest force of attraction. While, in plastic, the strong intermolecular force of attraction is present between elastomers and fibres.
In rubber, a few crosslinks are introduced between the chain, while plastics are linear or slightly branched molecules which gets soft on heating and hard on cooling.
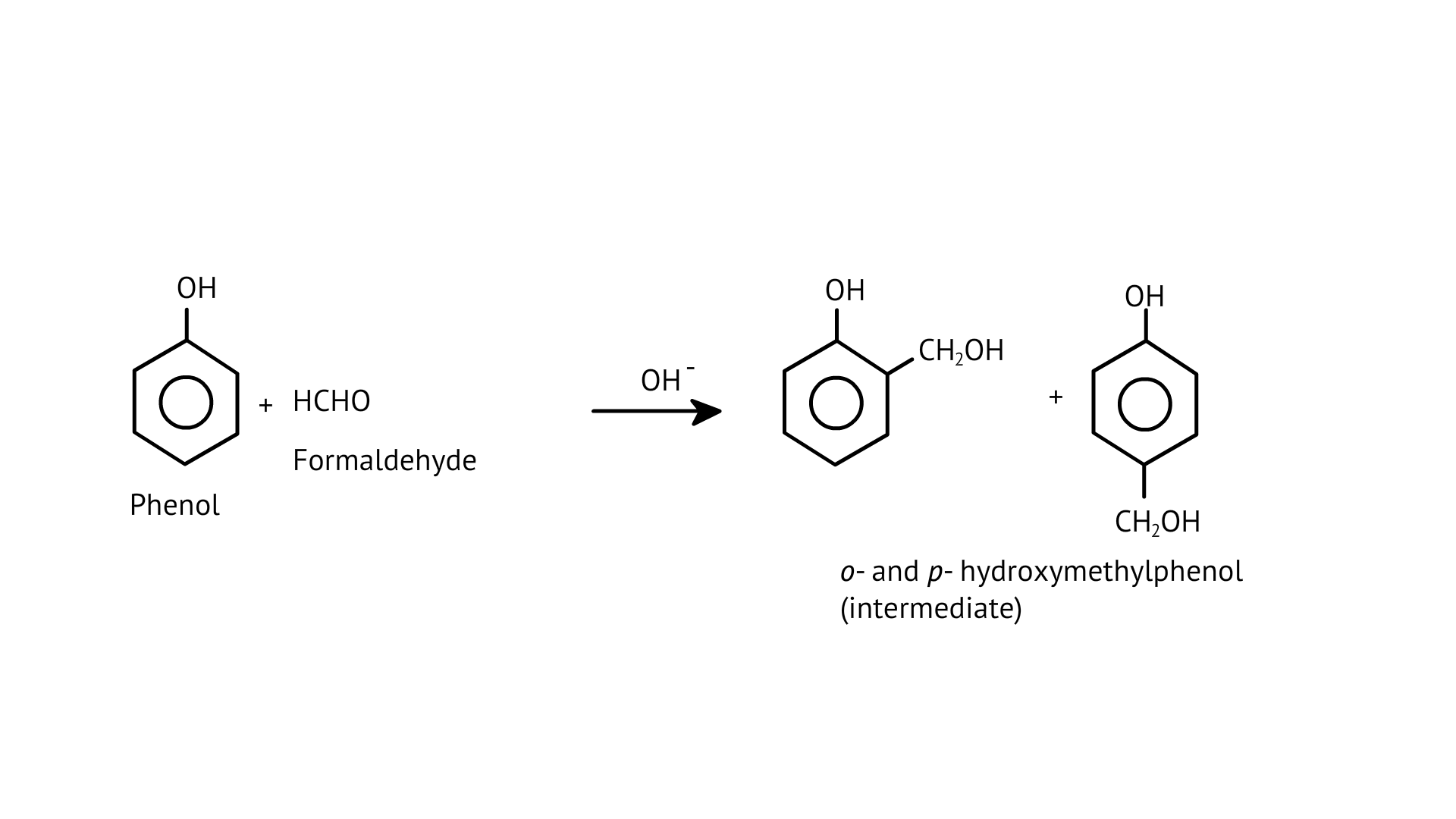
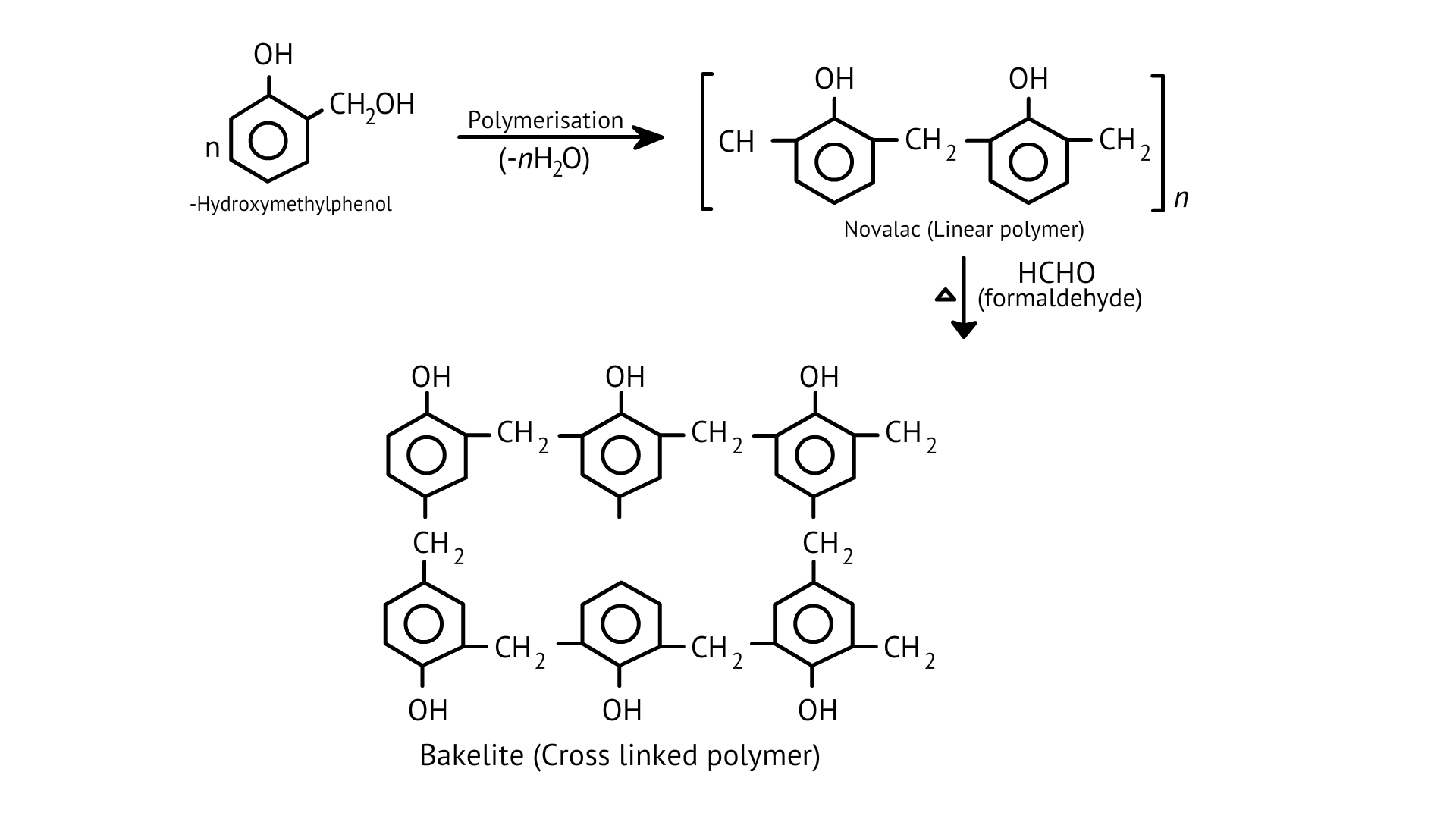
55. Phenol and formaldehyde undergo condensation to give a polymer (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
Ans: The oldest synthetic polymers are phenol-formaldehyde polymers. These are made by combining phenol and formaldehyde in the presence of either an acid or a base catalyst. The reaction begins with the formation of o-and/or p hydroxymethyl phenol derivatives, which then react with phenol to form compounds with shaving rings connected by –CH2 groups. The first product could be a linear product, such as Novolac.
When heated with formaldehyde, Novolac crosslinks to form Bakelite, an infusible solid mass.
It is used to make combs, phonograph records, electrical switches, and utensil handles.
The polymer A is Novolac and polymer B is Bakelite.
56. Low density polythene and high density polythene, both are polymers of ethene but there is marked difference in their properties. Explain.
Ans:
LDP | HDP |
1. It is obtained by polymerizing ethene under high pressures of 1000 to 2000 atmospheres at temperatures ranging from 350 K to 570 K in the presence of traces of dioxygen or a peroxide initiator (catalyst). | 1. It is formed when ethene is added polymerised in a hydrocarbon solvent in the presence of a catalyst such as triethylaluminium and titanium tetrachloride (Ziegler Natta catalyst) at temperatures ranging from 333 K to 343 K and pressures ranging from 6-7 atmospheres. |
2. LDP has a highly branched structure as a result of free radical addition and H-atom abstraction. | 2. HDP is made up of linear molecules and has a high density as a result of close packing. |
3. Low density polythene is chemically inert, tough, and flexible, but it is a poor conductor of electricity. | 3. It is also chemically inert and tougher than sand. |
57. Which of the following polymers soften on heating and harden on cooling? What are the polymers with this property collectively called? What are the structural similarities between such polymers? Bakelite, urea-formaldehyde resin, polythene, polyvinyl, polystyrene.
Ans: Thermoplastic polymer
Thermoplastic polymers are linear or slight branched-chain polymers. In this type of polymer, the strong intermolecular force of attraction is present between elastomers and fibers. This type of polymer softens on heating and hardens on cooling. For example, polythene, polystyrene and polyvinyl.
Thermosetting polymer
Thermosetting polymers are cross-linked polymers. These polymers on heating undergo cross-linking in molds. They become infusible on heating so, cannot be reused again. For example, Bakelite and urea-formaldehyde resins.
Summary of Chapter 15
Chapter 15 begins with an introduction to Polymers. Polymers are high molecular weight molecules composed of molecular weight components. We then move on to the classification synthesis. Mechanism of Polymerization has then been touched upon after which we come to some of the important polymers such as polythene, nylon, polyester, bakelite and natural rubber. Each of these has been elaborated upon along with everyday examples.
The chapter ends with the descriptions of bio-degradable (cellulose, proteins, starch) and non-biodegradable polymers(synthetic polymers).
FAQs on NCERT Exemplar for Class 12 Chemistry - Polymers - Free PDF Download
1. Where can I find relevant material on Polymers for class 12 Chemistry- Chapter 15?
You can find appropriate study material on Vedantu if you go to NCERT Exemplar for Class 12 Chemistry Chapter 15 - Polymers. The solved book of exercise questions is a great mode of revision for all students. Polymers have been explained with lots of examples and questions of all kinds related to the chapter are present in the book. Make sure that you solve all questions to pass examinations with flying colours. The study material present in the book will help you interestingly understand Chemistry too.
2. How do I study before my class 12 Chemistry examinations if I haven’t already studied chapter 15 previously?
A quick way for you would be to refer to Vedantu’s portal and scan all the study material. If you haven’t had the time to study the chapter from your textbook, you can go to this site and study from here. It has a summarised version of notes to read from and also has NCERT Exemplar for Class 12 Chemistry Chapter 15 - Polymers. This book of solved exercises will enable you to do well in your tests as all the questions here are similar to the ones that will come for your actual examinations. Free study material available on Vedantu for download. These can be accessed anytime and anywhere for free!
3. What is Nylon in Chapter 15 of Class 12 Chemistry?
Nylon belongs to a family of synthetic polymers which are made of polyamides. Its texture is similar to silk. A detailed description of what nylon is has been provided in NCERT Exemplar for Class 12 Chemistry Chapter 15 - Polymers. This book has all the required information that one needs to decipher the topics and sub-topics of the chapter. You can find this on Vedantu’s website and also the app. Vedantu is indeed a leading educational platform for those who are on the lookout for premium quality e-learning platforms.
4. How will I make revision notes for Class 12 Chemistry- Chapter 15 on Polymers?
You can compare and compile notes for the same by visiting NCERT Exemplar for Class 12 Chemistry Chapter 15 - Polymers. This book of solved exercises is available on Vedantu. It has been created as per the NCERT guidelines and is a masterpiece.
Solving these questions will enlighten you more on the subject and making notes will also get smoother after having solved these. One will then know which the most crucial topics are and make notes accordingly. Notes are of massive help before tests.
5. How trustworthy is an e-learning platform Vedantu for class 12 students?
Vedantu is completely trustworthy as an e-learning platform. It has a vast study material to choose from and has all the matters that’s needed by students of either board. Accessing the material provided on Vedantu is also quite smooth as most of the stuff is provided online for students to choose from and then study. You can download the app and access it from your phones too for the sake of convenience. It is quite handy for those who would love to study even while they’re commuting and do not have access to a laptop. Vedantu aims to educate each child who has a thirst for knowledge and wishes to learn.

























