Class 12 Exemplar Free Download on Vedantu
Free PDF download of NCERT Exemplar for Class 12 Chemistry Chapter 13 - Amines solved by expert Chemistry teachers on Vedantu.com as per NCERT (CBSE) Book guidelines. All Chapter 13 - Amines exercise questions with solutions to help you to revise the complete syllabus and score more marks in your examinations.
Access NCERT Exemplar Solutions for Class 12 Chemistry Chapter 13- Amines
MULTIPLE CHOICE QUESTIONS (TYPE-I)
1. Which of the following is a 3° amine?
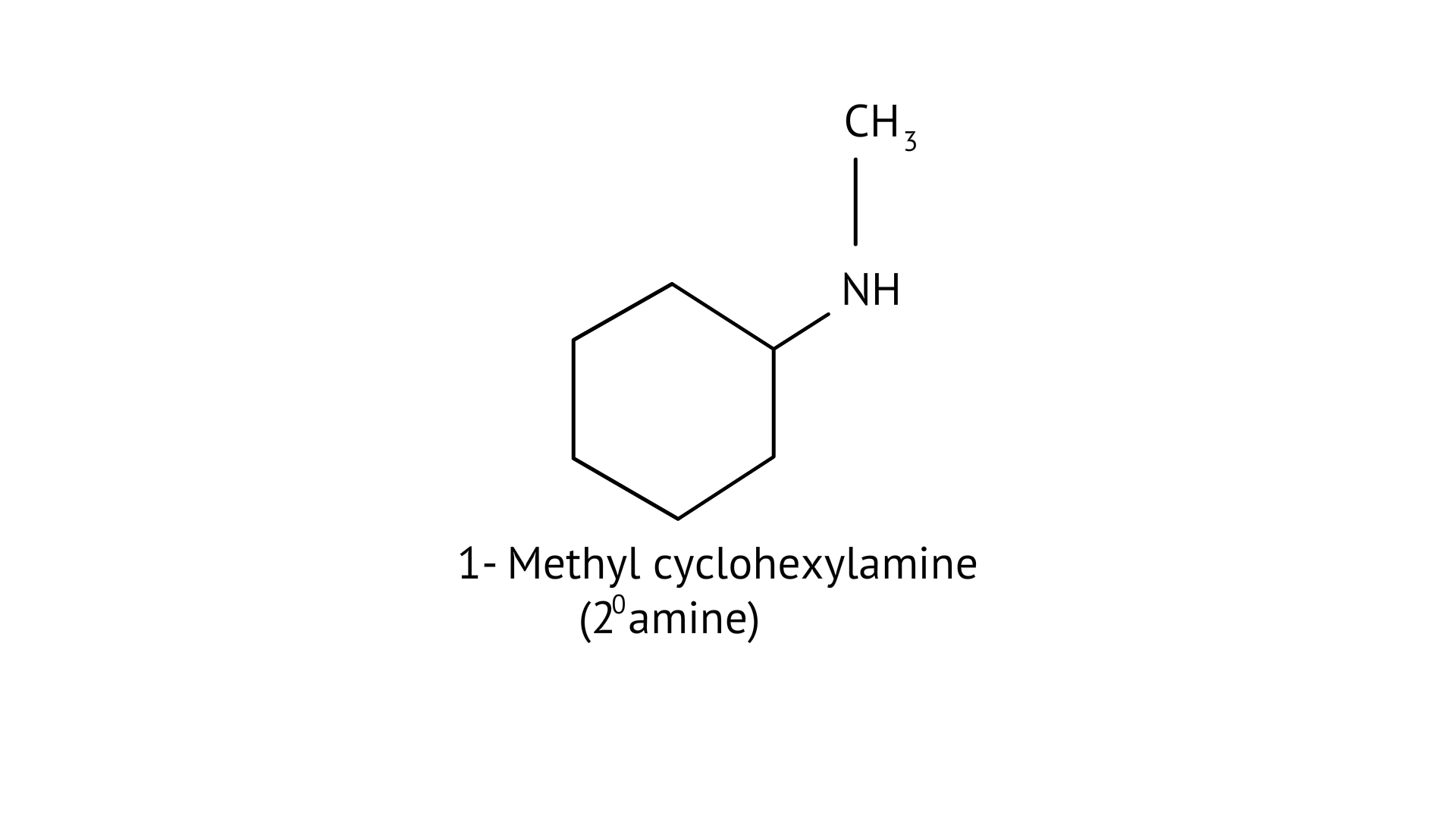
(A) 1-methylcyclohexylamine
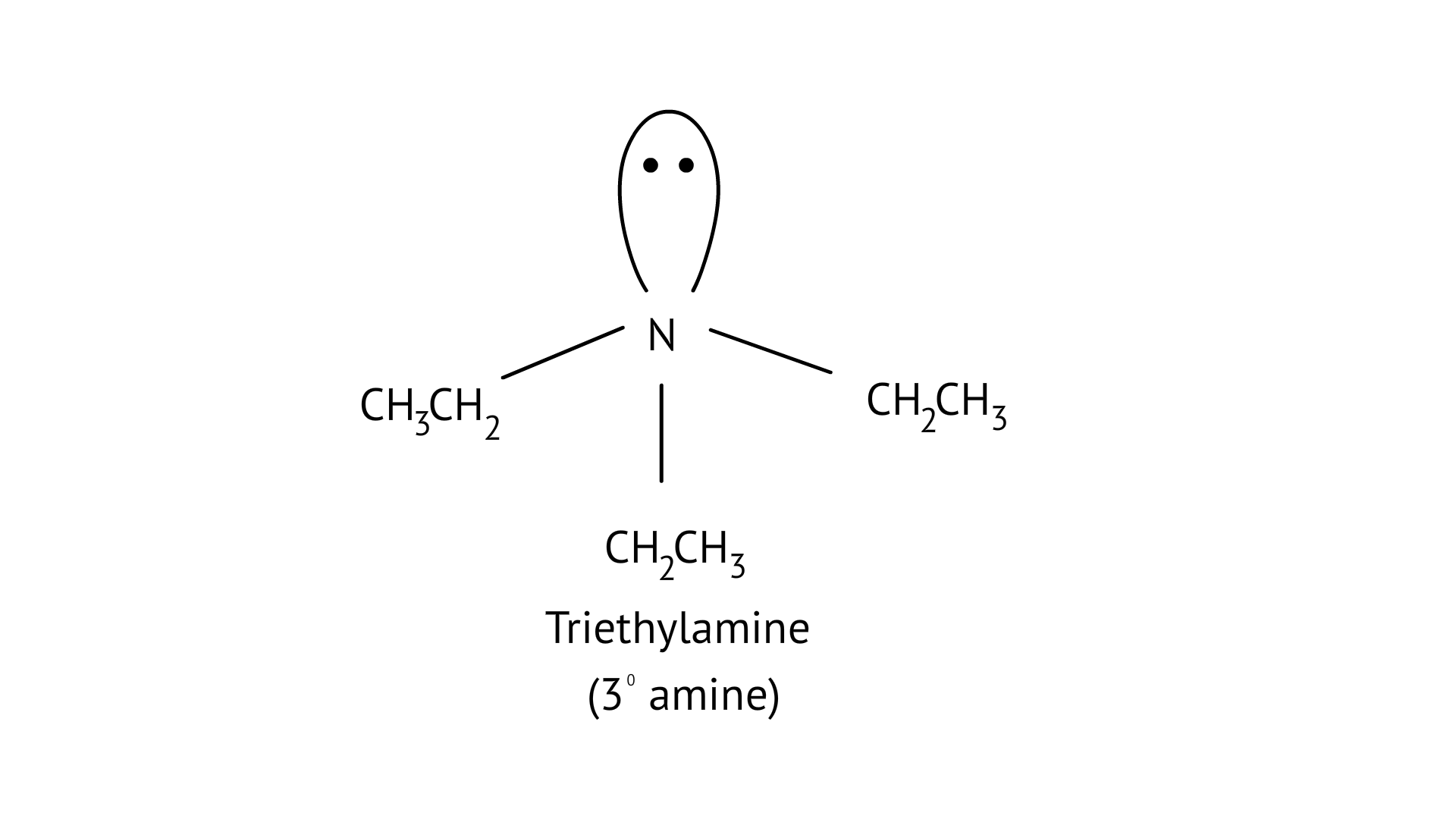
(B) Triethylamine
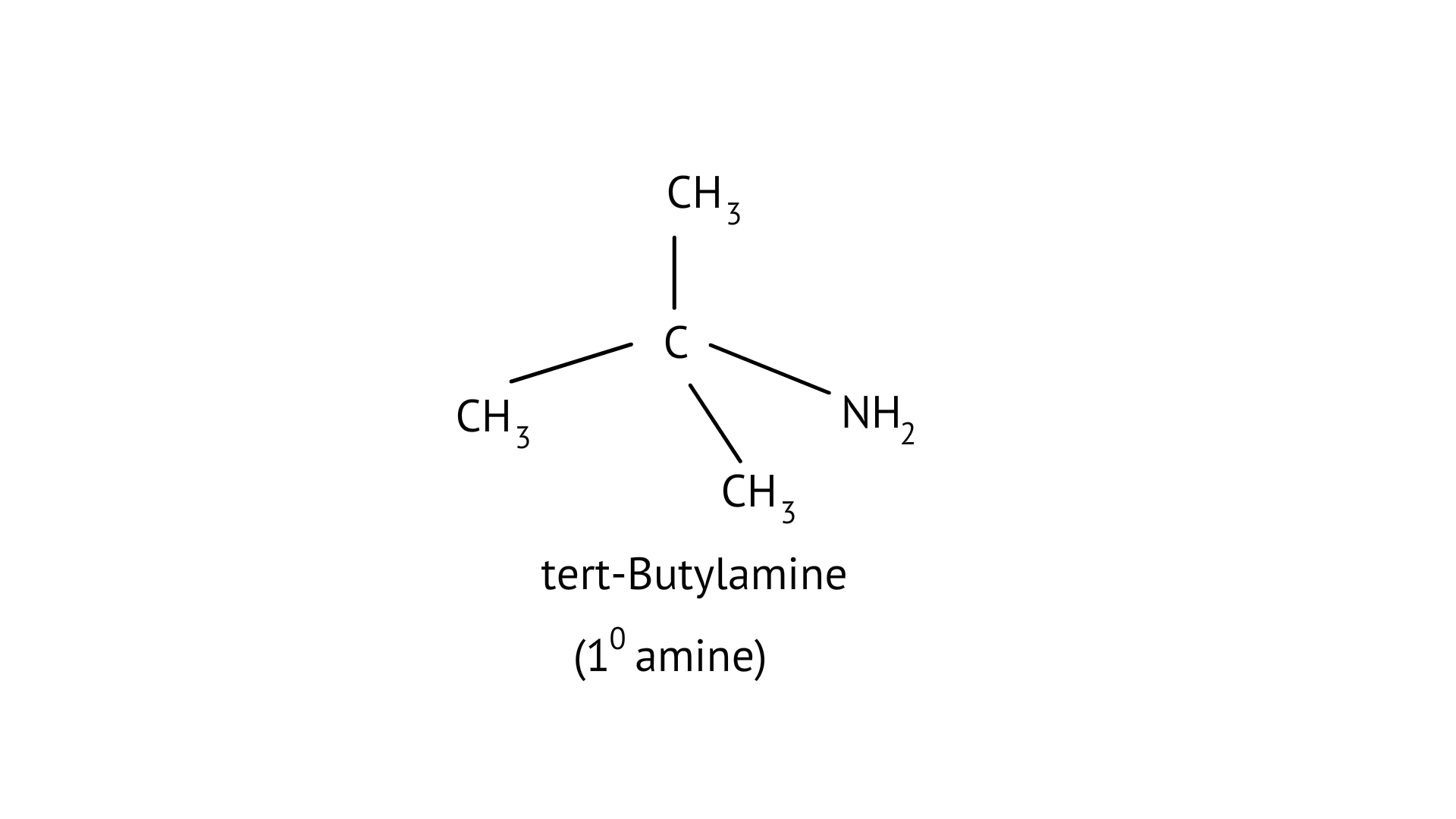
(C) tert-butylamine
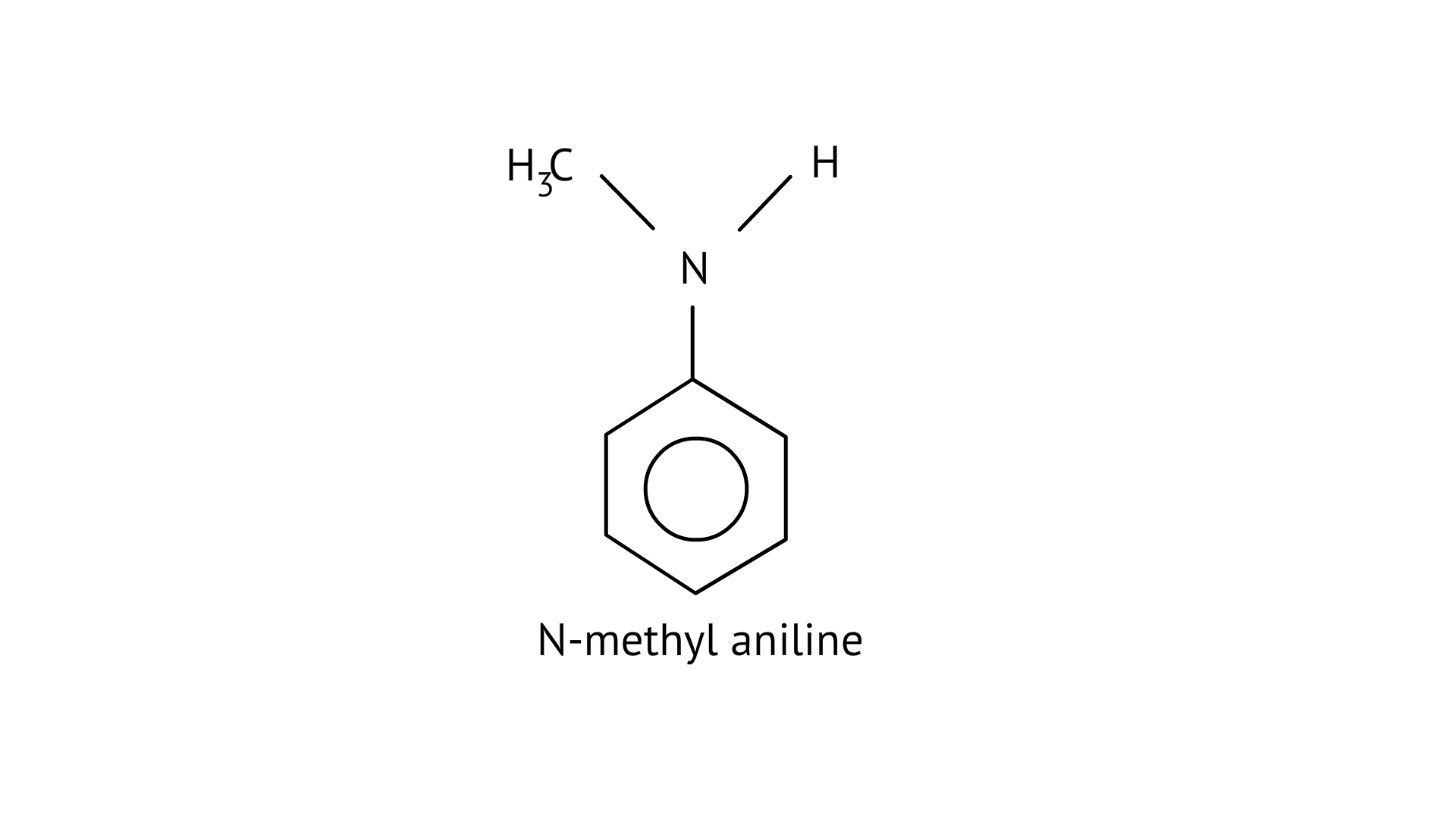
(D) N-methylaniline
Ans: (B) Triethylamine
The structure of the given amines are shown below.
(A)
(B)
(C)
(D)
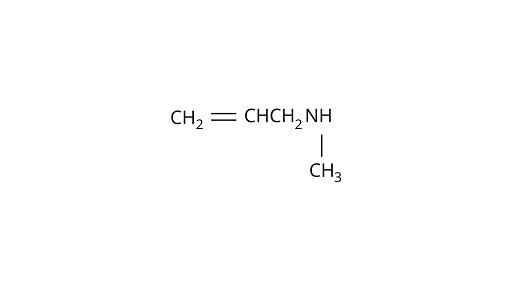
2. The correct IUPAC name for $\mathrm{CH}_{2}=\mathrm{CH}-\mathrm{CH}_{2}-\mathrm{NH}-\mathrm{CH}_{3}$ is
(A) allylmethylamine
(B) 2-amino-4-pentene
(C) 4-aminopent-l-ene
(D) N-méthylprop-2-en-l-amine
Ans: Correct answer: (D)
3. Amongst the following, the strongest base in an aqueous medium is.
(A) $\mathrm{CH}_{3} \mathrm{NH}_{2}$
(B) $\mathrm{NCCH}_{2} \mathrm{NH}_{2}$
(C) $\left(\mathrm{CH}_{3}\right)_{2} \mathrm{NH}$
(D) $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NHCH}_{3}$
Ans: (C)
Because of the electron-releasing nature of the alkyl group, it (R) pushes electrons towards nitrogen, making the unshared electron pair more available for sharing with the acid's proton. Furthermore, the +I effect of the alkyl group stabilizes the substituted ammonium ion formed from the amine by dispersing the positive charge.
As a result, alkylamines are more powerful bases than ammonia.
As a result, the basic nature of aliphatic amines should increase as the number of alkyl groups increases.
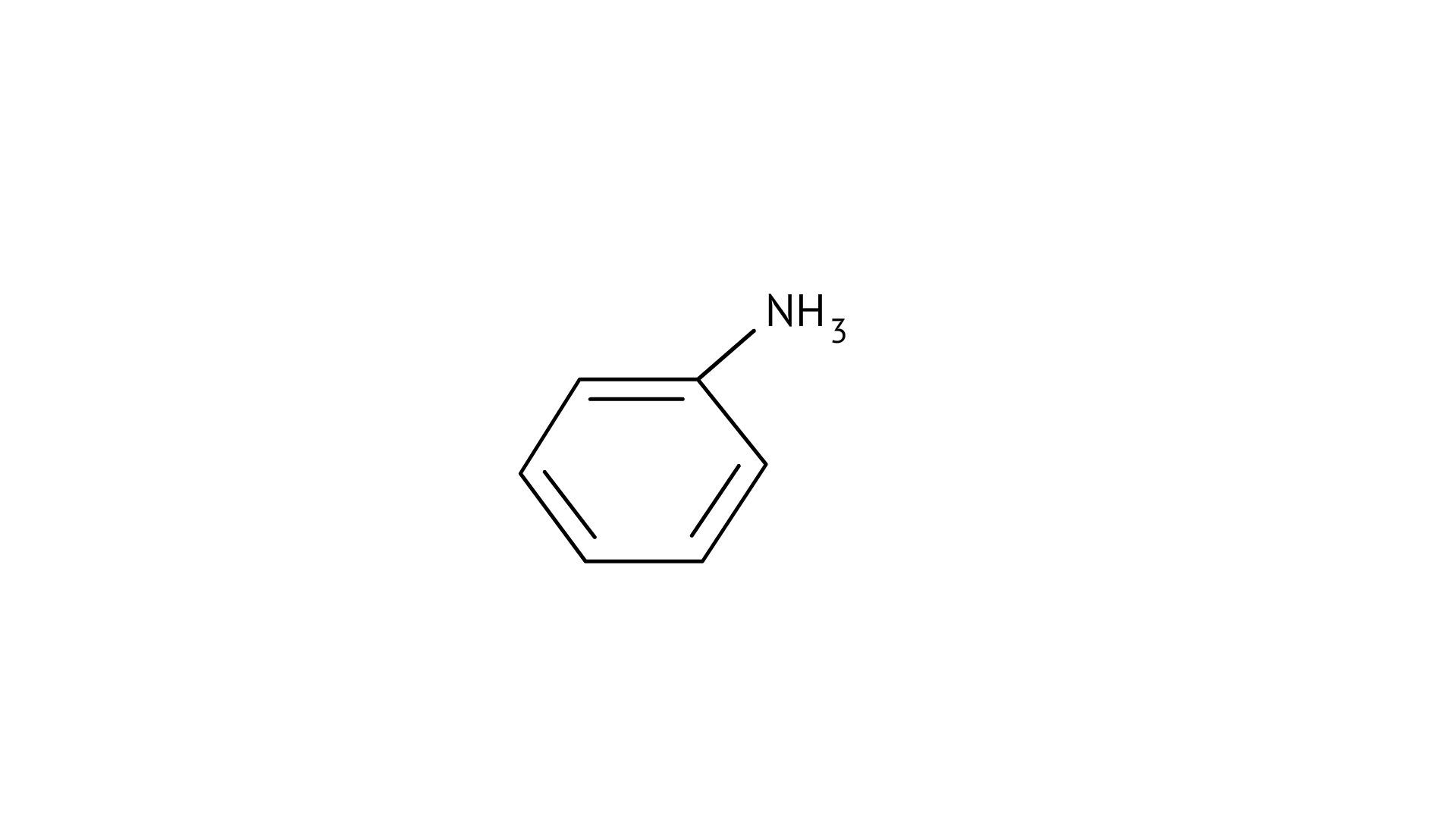
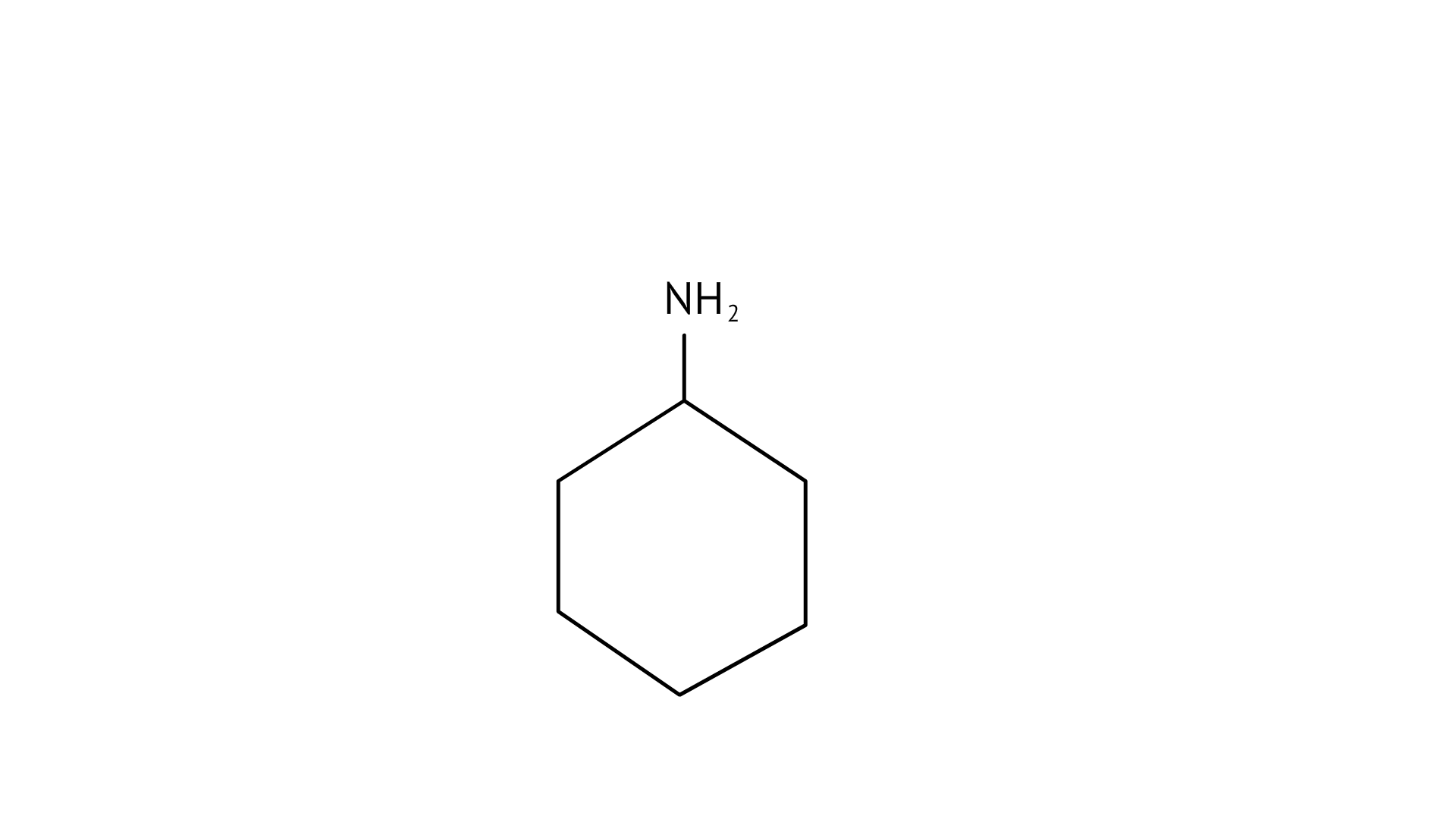
4. Which of the following is the weakest Bronsted base?
(A)
(B)
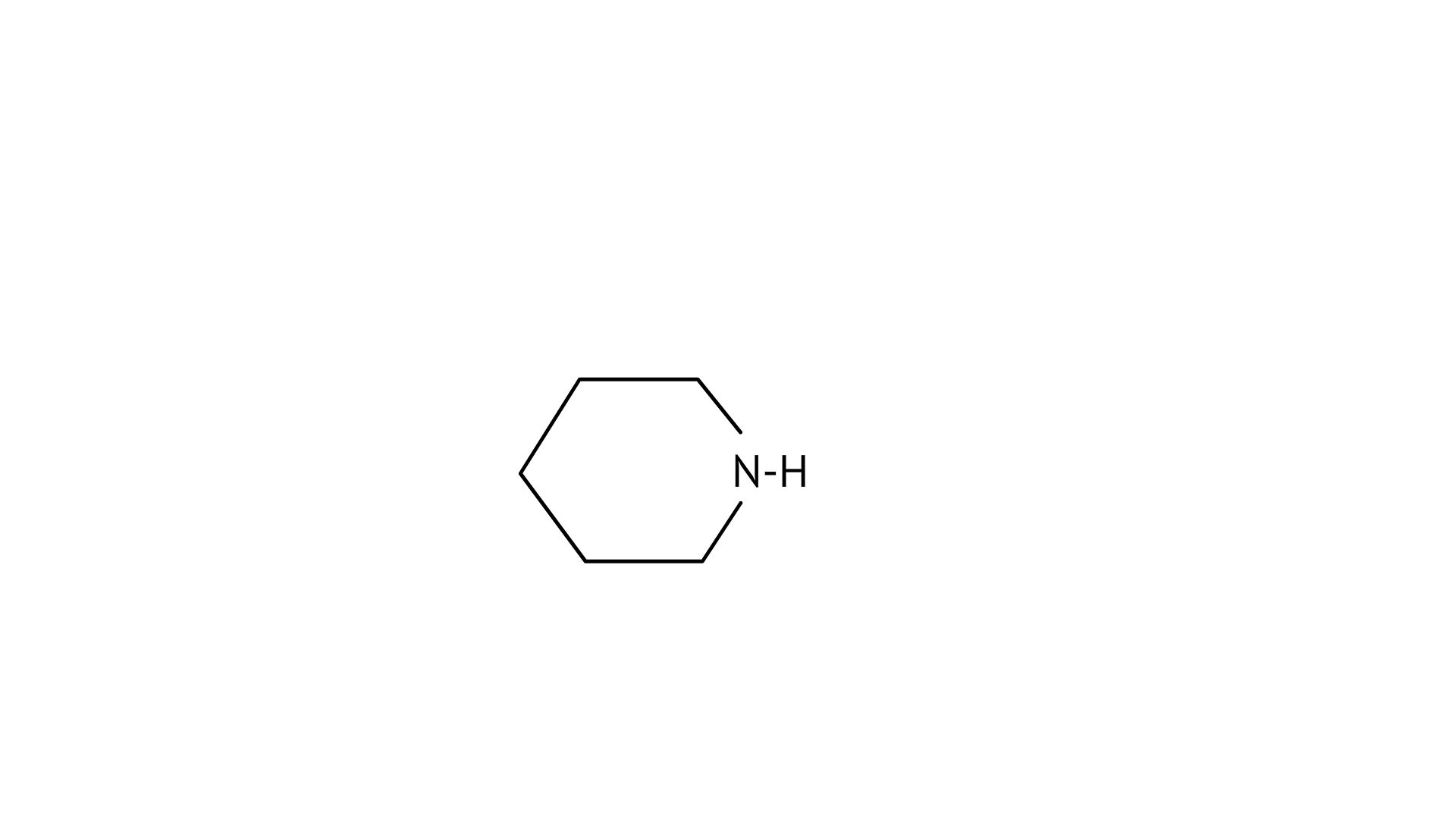
(C)
(D) $\mathrm{CH}_{3} \mathrm{NH}_{2}$
Ans: (A)
The $\mathrm{NH}_{2}$ group is directly attached to the benzene ring in aniline and other aryl amines. As a result, the unshared electron pair on the nitrogen atom is conjugated with the benzene ring, making it less available for protonation.
5. Benzylamine may be alkylated as shown in the following equation:
$\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{NH}_{2}+\mathrm{R}-\mathrm{X} \rightarrow \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{NHR}$
Which of the following alkyl halides is best suited for this reaction through $\text{S}{{\text{N}}^{\text{1}}}$ mechanism?
(A) $\mathrm{CH}_{3} \mathrm{Br}$
(B) $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{Br}$
(C) $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{Br}$
(D) $\mathrm{C}_{2} \mathrm{H}_{5} \mathrm{Br}$
Ans: (C)
SN1 reaction: A nucleophilic reaction that occurs in two steps, first is the bond-breaking step and the second is the production of the carbocation. The stability of carbocation formed in the second step determines the rate of reactivity of reactant toward $\text{S}{{\text{N}}^{\text{1}}}$ reaction. Here, $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{Br}$,
In the process of ionization, removal of bromine, a stable Benzyl carbocation is produced. Therefore, it is best suited for reaction through the $\text{S}{{\text{N}}^{\text{1}}}$ mechanism.
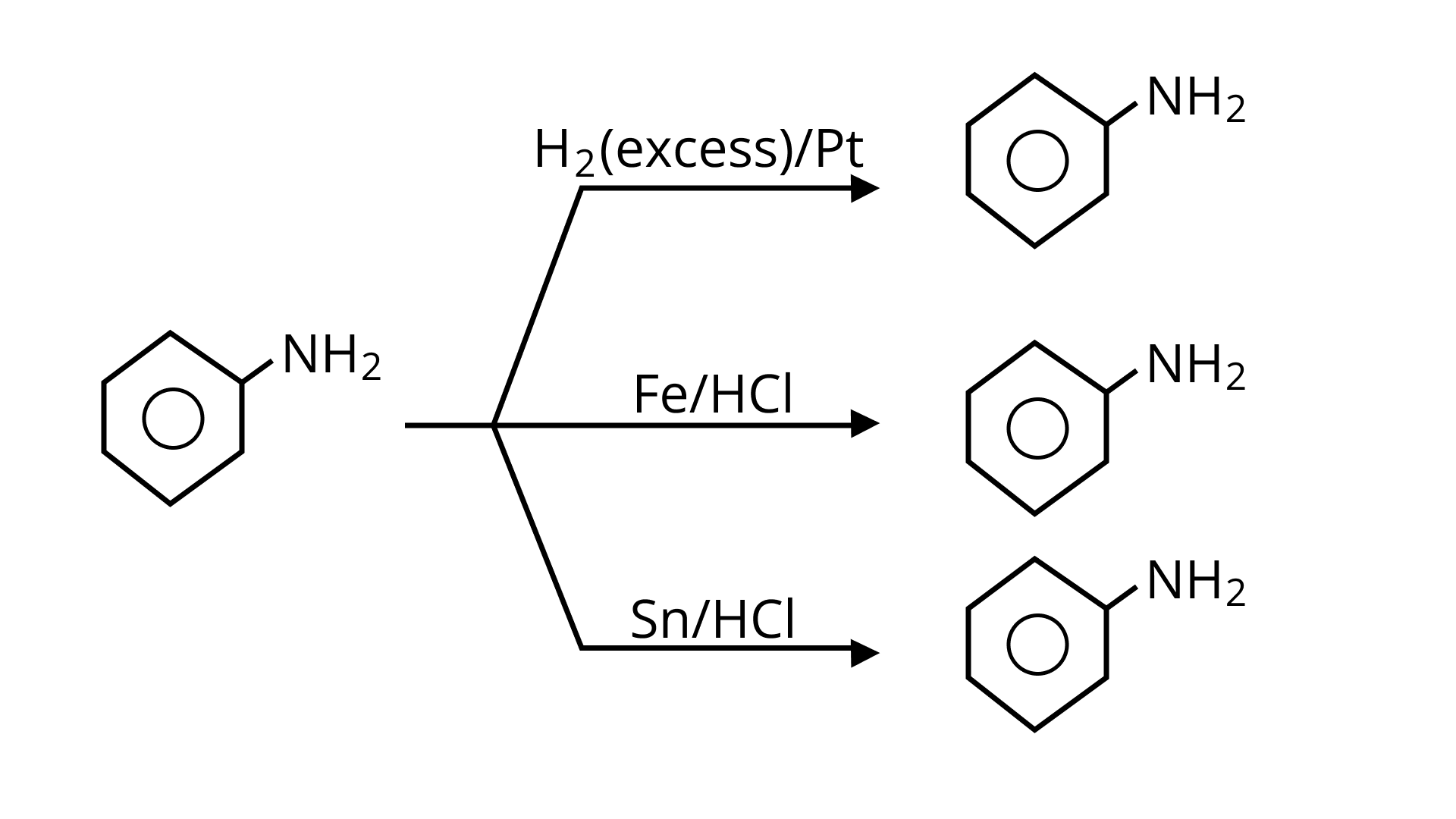
6. Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
(A) $\mathrm{H}_{2}$ (excess) $/ \mathrm{Pt}$
(B) $\mathrm{LiAlH}_{4}$ in ether
(C) $\mathrm{Fe}$ and $\mathrm{HCl}$
(D) $\mathrm{Sn}$ and $\mathrm{HCl}$
Ans: (B)
Lithium aluminum hydride in ether is a strong reducing agent that donates its $\mathrm{H}^{-}$ hydride ion to any $\mathrm{C}=\mathrm{O}$ containing a functional group. In addition to $\mathrm{LiAlH}_{4}$ to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
$2 \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NO}_{2} \stackrel{\text { LiAlH }_{4} / \text { ether }}{\longrightarrow} \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{~N}=\mathrm{N}-\mathrm{C}_{6} \mathrm{H}_{5}$
7. In order to prepare a primary amine from an alkyl halide with simultaneous addition of one $\mathrm{CH}_{2}$ group in the carbon chain, the reagent used as a source of nitrogen is
(A) sodium amide, $\mathrm{NaNH}_{2}$
(B) sodium azide, $\mathrm{NaN}_{3}$
(C) potassium cyanide, $\mathrm{KCN}$
(D) potassium phthalimide, $\mathrm{C}_{6} \mathrm{H}_{4}\left(\mathrm{CO}_{2}\right) \mathrm{N}^{-} \mathrm{K}^{+}$
Ans: (C)
Potassium cyanide, as cyanide on reduction with sodium metal in alcohol, produces amine with increased carbon atoms.
$\mathrm{R}-\mathrm{X} \stackrel{\mathrm{KCN}}{\stackrel{-\mathrm{KX}}{\longrightarrow}} \mathrm{R}-\mathrm{CN} \stackrel{\mathrm{Na} / \mathrm{C}_{2} \mathrm{H}_{5} \mathrm{OH}}{\longrightarrow} \mathrm{R}-\mathrm{CH}_{2} \mathrm{NH}_{2}$
8. The source of nitrogen in Gabriel synthesis of amines is.
(A) sodium azide, $\mathrm{NaN}_{3}$
(B) sodium nitrite, $\mathrm{NaNO}_{2}$
(C) potassium cyanide, $\mathrm{KCN}$
(D)potassium phthalimide, $\mathrm{C}_{6} \mathrm{H}_{4}(\mathrm{CO} 2) \mathrm{N}^{-} \mathrm{K}^{+}$
Ans: (D)
Gabriel synthesis reaction:
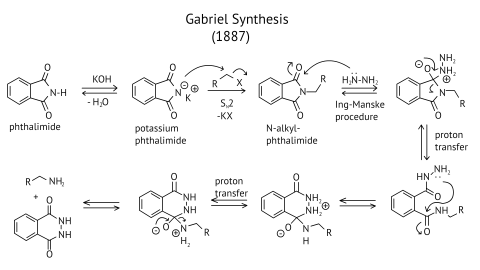
Gabriel’s synthesis is a method for producing primary amines. When phthalimide is treated with ethanolic potassium hydroxide, it forms a potassium salt of phthalimide, which when heated with an alkyl halide and then alkaline hydrolyzed yields the corresponding primary amine. This method cannot produce aromatic primary amines because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide.
9. Amongst the given set of reactants, the most appropriate for preparing $2^{\circ}$ amine is.
(A) $2^{\circ} \mathrm{R}-\mathrm{Br}+\mathrm{NH}_{3}$
(B) $2^{\circ} \mathrm{R}-\mathrm{Br}+\mathrm{NaCN}$ followed by $\mathrm{H}_{2} / \mathrm{Pt}$
(C)$1^{\circ} \mathrm{R}-\mathrm{NH}_{2}+\mathrm{RCHO}$ followed by $\mathrm{H}_{2} / \mathrm{Pt}$
(D) $1^{\circ} \mathrm{R}-\mathrm{Br}+(2 \mathrm{~mol})+$ potassium phthalimide followed by $\mathrm{H}_{3} \mathrm{O}^{+} /$ heat
Ans: ( C)
$\underset{1^{\circ} \text { Amine }}{\mathrm{R}-\mathrm{NH}_{2}}+\mathrm{O}=\mathrm{CHR} \underset{\text { Aldehyde }}{\stackrel{\text { Reductive }}{\text { amination }} \longrightarrow \mathrm{R}-\mathrm{N}=\mathrm{CHR} \frac{\mathrm{H}_{2} / \mathrm{Pt}}{\text { Reduction }} \underset{2^{\circ} \text { Amine }}{\mathrm{RNH}}-\mathrm{CH}_{2} \mathrm{R}}$
10. The best reagent for converting 2-phenylpro-panamide into-2-phenyl- propanamine is.
(A) excess $\mathrm{H}_{2}$
(B) $\mathrm{Br}_{2}$ in aqueous $\mathrm{NaOH}$
(C) iodine in the presence of red phosphorus
(D) $\mathrm{LiAlH}_{4}$ in ether
Ans: (D)
Lithium aluminium hydride in ether is a strong reducing agent that donates its hydride ion to any $\mathrm{C}=\mathrm{O}$ containing functional groups into corresponding reduced compounds. Here, the amide group is converted to an amine functional group.
$\mathrm{LiAlH}_{4}$ in ether is the best reagent for converting 2-phenylpropanamide to 2-phenylpropanamine.
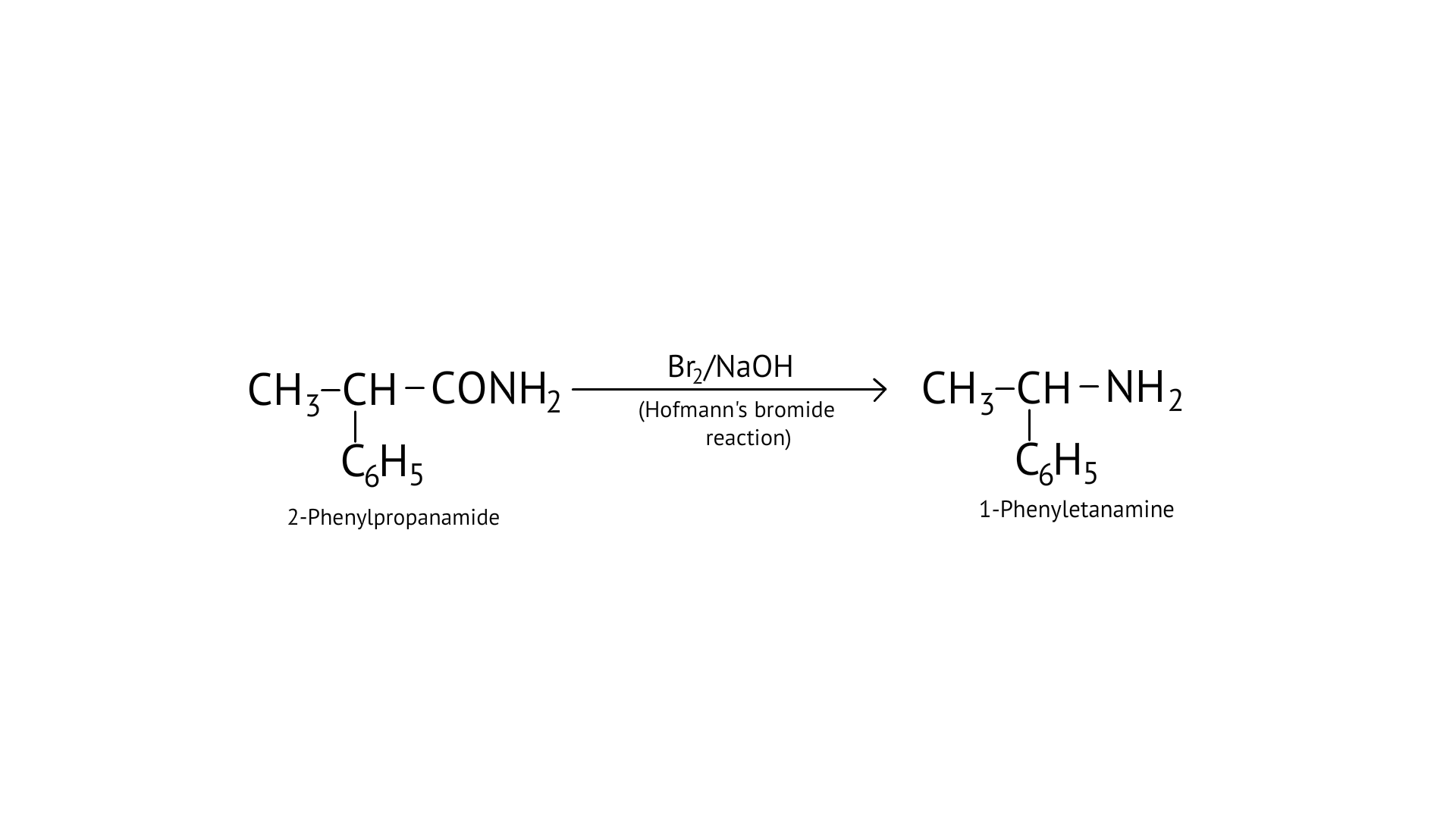
11. The best reagent for converting-2-phenylpro-panamide into 1-phenylethanamine is.
(A) excess $\mathrm{H}_{2} / \mathrm{Pt}$
(B) $\mathrm{NaOH} / \mathrm{Br}_{2}$
(C) $\mathrm{NaBH}_{4} /$ methanol
(D) $\mathrm{LiAlH}_{4} /$ ether
Ans: (B)
Hoffmann bromamide reaction- it is also called degradation reaction as in this reaction primary amide group is treated with halogen first (Br) then the halogen-substituted amide product is converted to a primary amine with the release of carbon dioxide gas.
12. Hoffmann bromamide degradation reaction is shown by.
(A) $\mathrm{ArNH}_{2}$
(B) $\mathrm{ArCONH}_{2}$
(C) $\mathrm{ArNO}_{2}$
(D) $\mathrm{ArCH}_{2} \mathrm{NH}_{2}$
Ans: (B)
Hoffmann invented a method for producing primary amines by treating an amide with bromine in an aqueous or ethanolic sodium hydroxide solution. An alkyl or aryl group migrates from the amide's carbonyl carbon to the nitrogen atom during this degradation reaction.
$\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CONH}_{2} \stackrel{\mathrm{Br}_{2}+\mathrm{NaOH}}{\longrightarrow} \mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NH}_{2}$
13. The correct increasing order of basic strength for the following compounds is
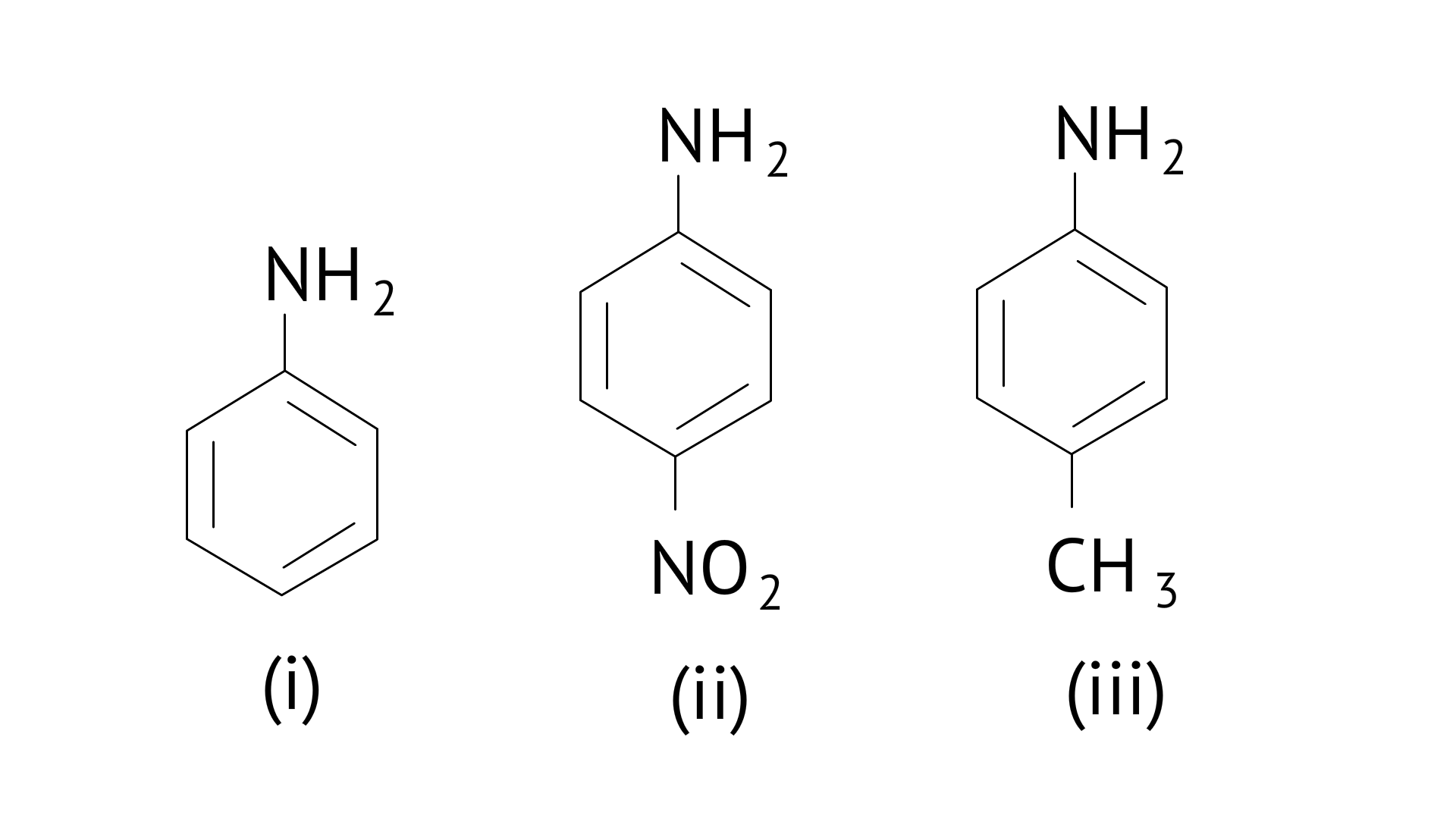
(A) (II) $<($ III $)<($ I $)$
(B) (III) $<(\mathrm{I})<(\mathrm{II})$
(C) (III) $<$ (II) $<$ (I)
(D) (II) $<(\mathrm{I})<(\mathrm{III})$
Ans: (D)
The greater the electron density towards the ring, the greater its basic strength.
The electron withdrawing group reduces basic strength, whereas the electron donating group increases basic strength.
14. Methylamine reacts with $\mathrm{HNO}_{3}$ to form
(A) $\mathrm{CH}_{3}-\mathrm{O}-\mathrm{N}=\mathrm{O}$
(B) $\mathrm{CH}_{3}-\mathrm{O}-\mathrm{CH}_{3}$
(C) $\mathrm{CH}_{2} \mathrm{OH}$
(D) $\mathrm{CH}_{3} \mathrm{CHO}$
Ans: (C)
Methyl amine reacts with $\mathrm{HNO}_{3}$ form methanol with release of nitrogen gas and water as side products.
$\mathrm{CH}_{3} \mathrm{NH}_{2}+\mathrm{HNO}_{3} \stackrel{0^{\circ}-5^{\circ} \mathrm{C}}{\rightarrow} \mathrm{CH}_{3} \mathrm{OH}+\mathrm{N}_{2}+\mathrm{H}_{2} \mathrm{O}$
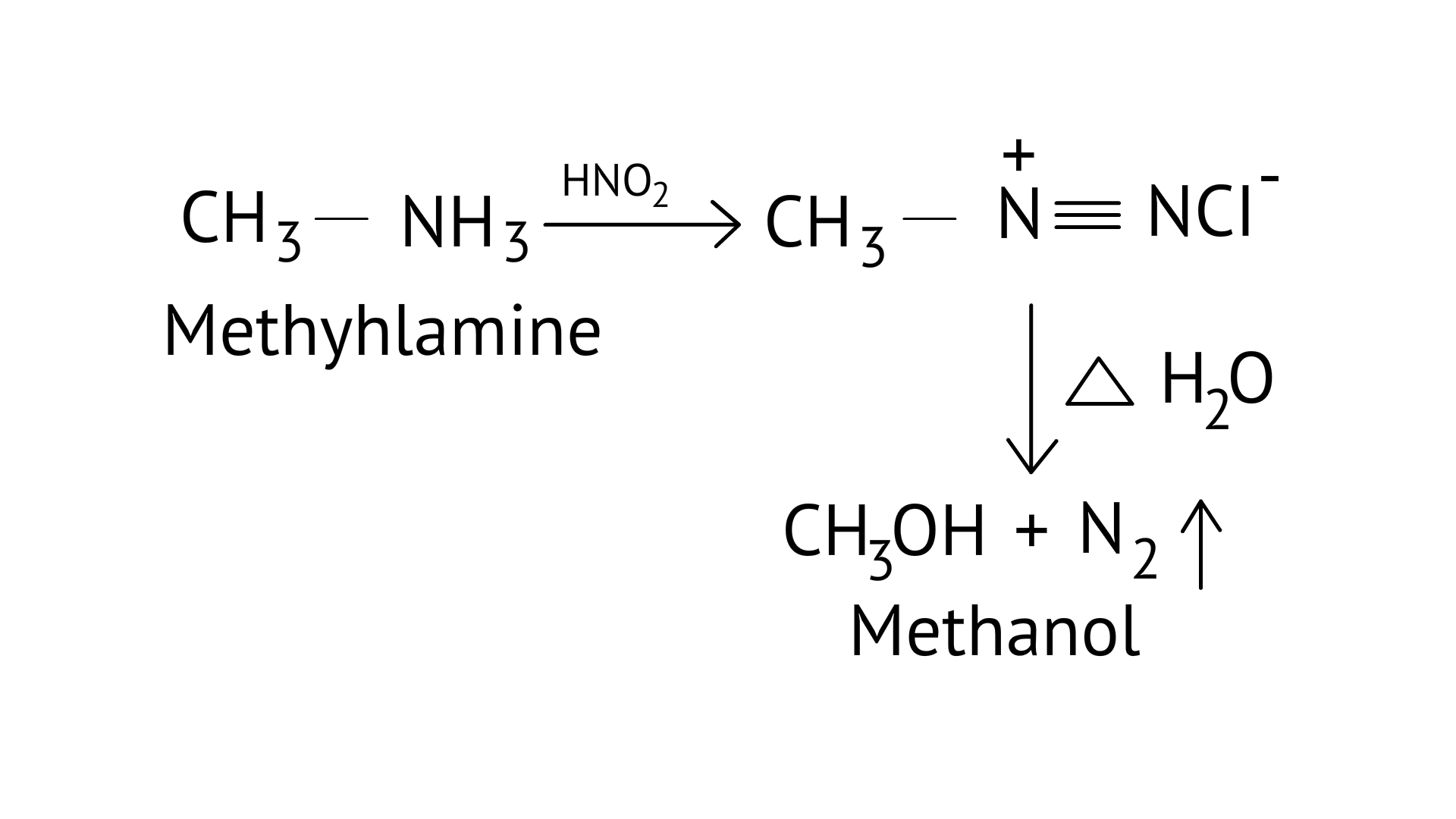
15. The gas evolved when methylamine reacts with nitrous acid is .
(A) $\mathrm{NH}_{3}$
(B) $\mathrm{N}_{2}$
(C) $\mathrm{H}_{2}$
(D) $\mathrm{C}_{2} \mathrm{H} 6$
Ans: (B)
Primary aliphatic amines react with nitrous acid to form aliphatic diazonium salts, which are unstable and release nitrogen gas and alcohol quantitatively.
16. In the nitration of benzene using a mixture of conc. $\mathrm{H}_{2} \mathrm{SO}_{4}$ and conc. $\mathrm{HNO}_{3}$, the species which initiates the reaction is.
(A) $\mathrm{NO}_{2}$
(B) $\mathrm{NO}^{+}$
(C) $\mathrm{NO}_{2}^{+}$
(D) $\mathrm{NH}_{2}$
Ans: (C)
In the process of nitration of benzene, firstly $\mathrm{H}_{2} \mathrm{SO}_{4}$ dissociates into $\mathrm{H}^{+}$and $\mathrm{HSO}_{4}{ }^{-}$. The proton produced further reacts with $\mathrm{HNO}_{3}$ to give a positively charged complex that is unstable and dissociates into nitronium ions and water as products. The nitronium ion is an electron-deficient species i.e. electrophile that attacks the electron cloud of the benzene ring.
$\mathrm{H}_{2} \mathrm{SO}_{4} \text { (conc.) } \longrightarrow \mathrm{H}^{+}+\mathrm{HSO}_{4}^{-}$
$\mathrm{H}^{+}+\mathrm{HNO}_{3} \longrightarrow \mathrm{H}_{2} \mathrm{NO}_{3}^{+}$
$\mathrm{H}_{2} \mathrm{NO}_{3}^{+} \longrightarrow \mathrm{NO}_{2}^{+}+\mathrm{H}_{2} \mathrm{O}$
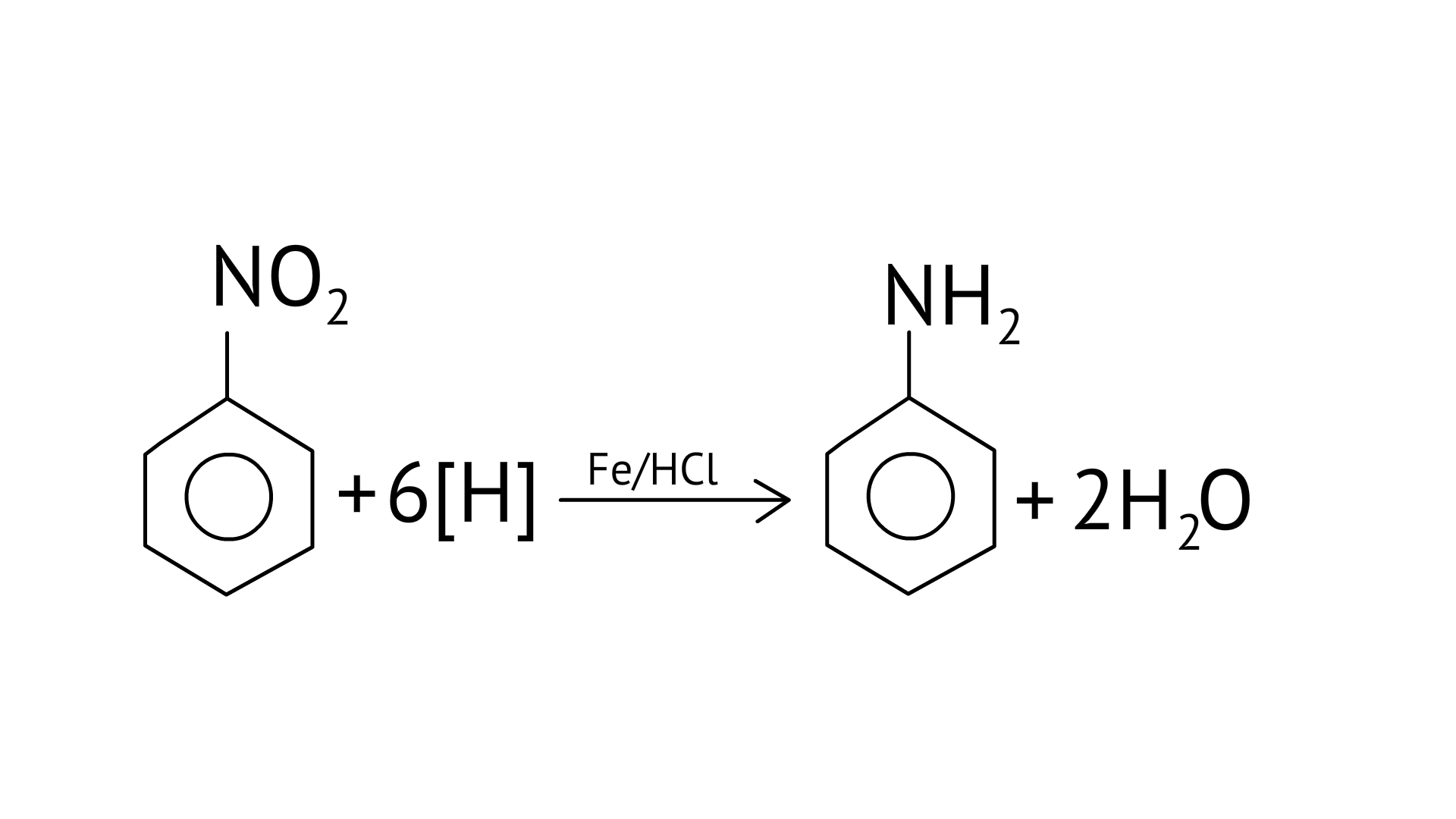
17. Reduction of aromatic nitro compounds using Fe and HCl gives.
(A) aromatic oxime
(B) aromatic hydrocarbon
(C) aromatic primary amine
(D) aromatic amide
Ans: (C)
Using active metals like iron in acidic conditions can be efficiently used for the reduction of nitro compounds.
Under the acidic conditions, the intermediate compounds that may form are readily reduced to primary amine and pure product of primary amine is obtained.
18. The most reactive amine towards dilute hydrochloric acid is
(A) $\mathrm{CH}_{3}-\mathrm{NH}_{2}$
(B)
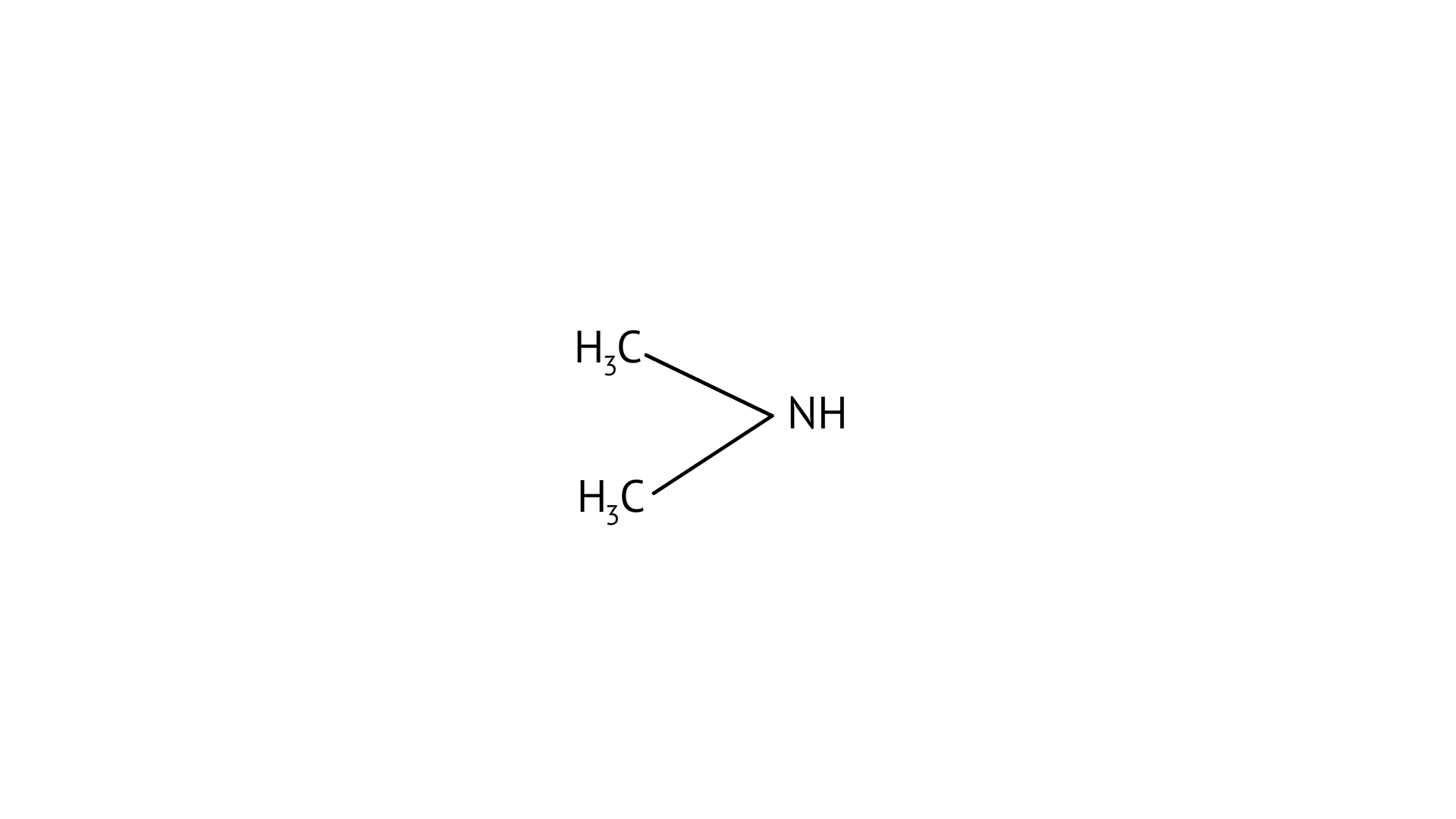
(C)
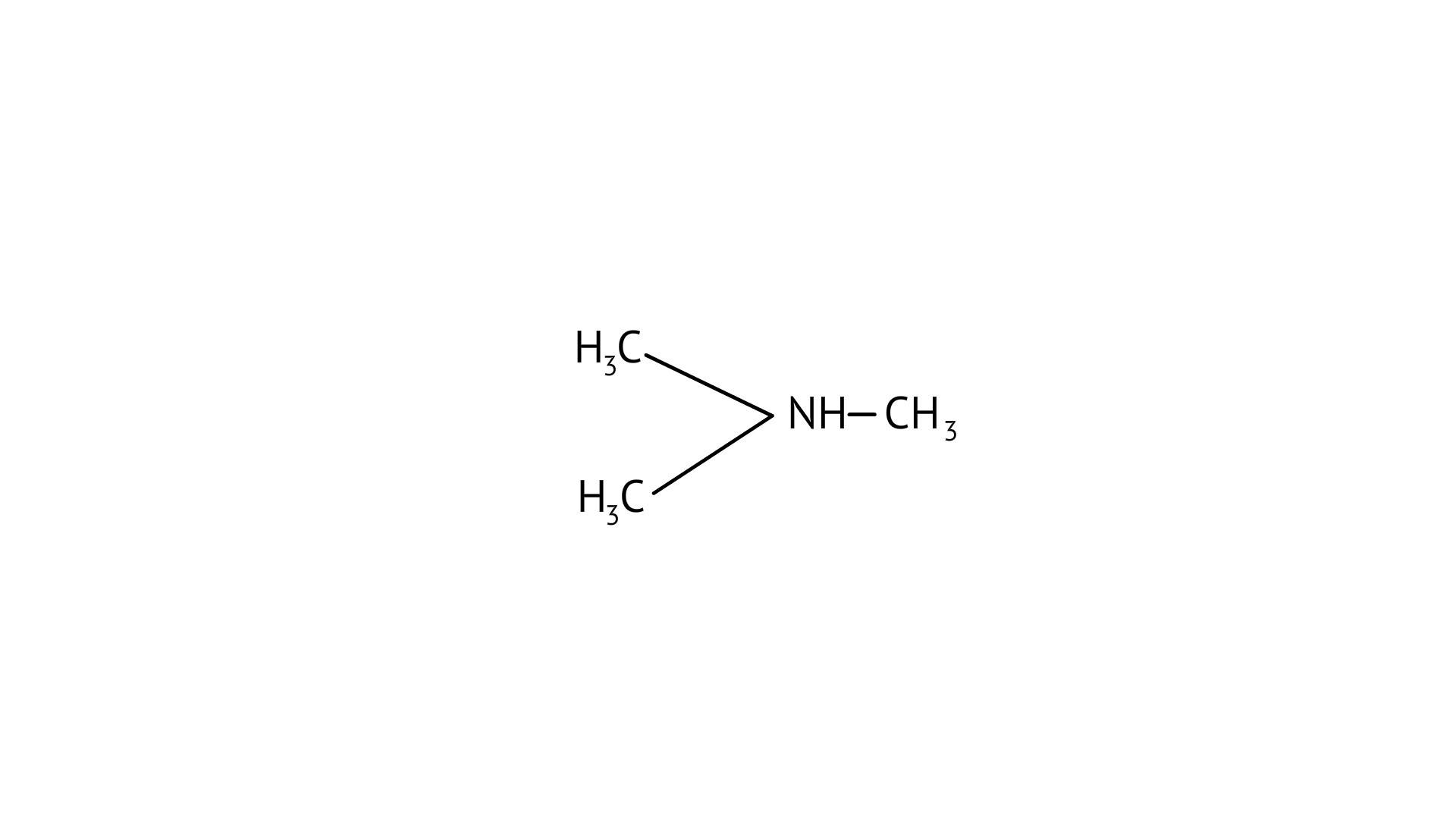
(D)
Ans: (B)
Greater will be the basic character, greater will be its reactivity towards dilute hydrochloric acid. As, (B), secondary amine, it will be the most basic among all and hence the most reactive amine towards acid.
19. Acid anhydrides on reaction with primary amines give.
(A) amide
(B) imide
(C) secondary amine
(D) imine
Ans: (A)
On Reaction of acid anhydrides with primary amines, amides are formed as the primary product along with carboxylic acid.
$(\mathrm{RCO})_{2} \mathrm{O}+\mathrm{RNH}_{2} \rightarrow \mathrm{RCONH}_{2}+\mathrm{RCOOH}$
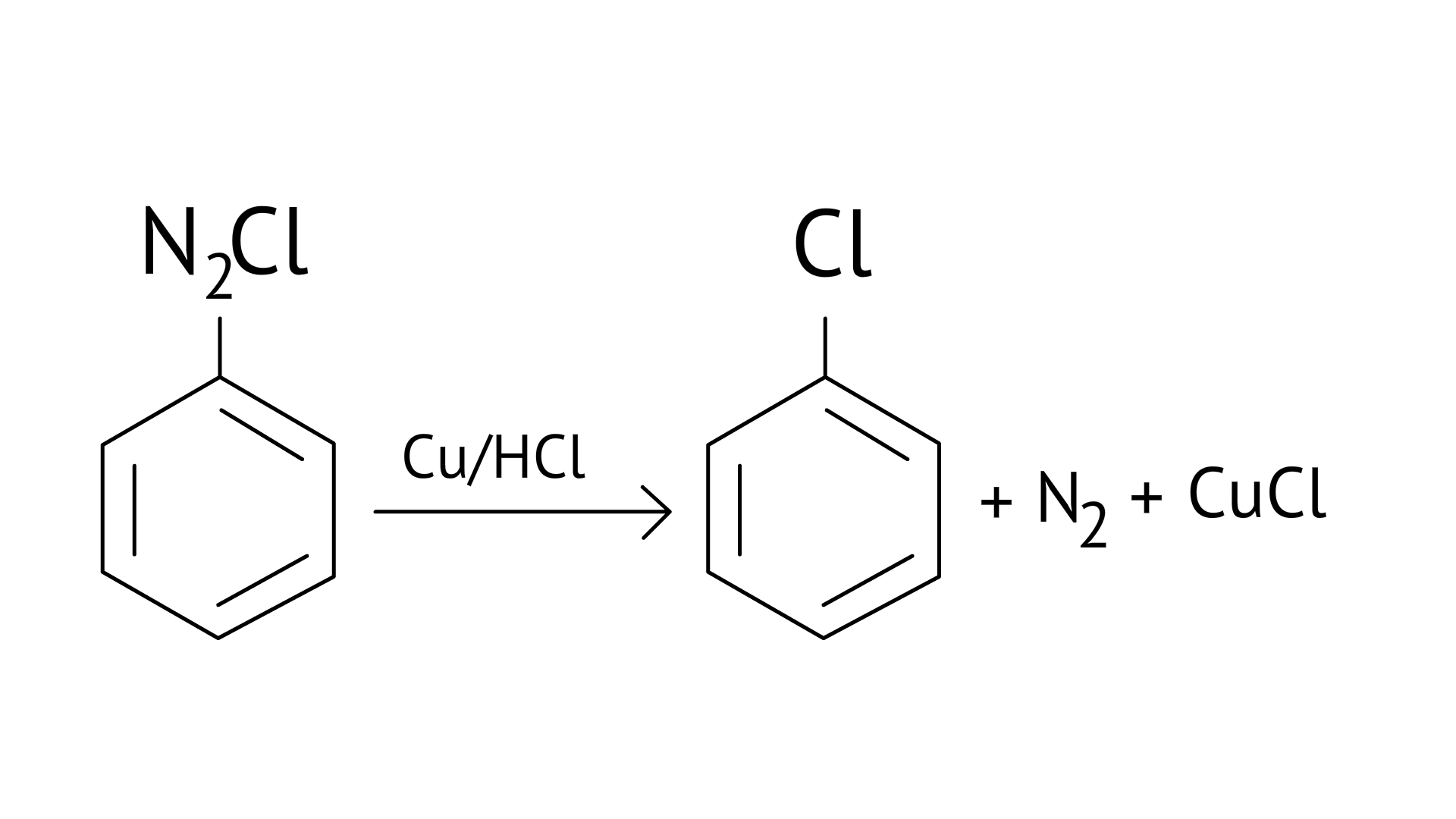
20. The reaction as $\mathrm{Ar} \mathrm{N}_{2} \mathrm{Cl}^{-} \stackrel{\mathrm{Cu} / \mathrm{HCl}}{\longrightarrow} \mathrm{ArCl}+\mathrm{N}_{2}+\mathrm{CuCl}$ is names
(A) Sandmeyer reaction
(B) Gatterman reaction
(C) Claisen reaction
(D) Carbylamine reaction
Ans: (B)
Diazonium salts react with copper powder and halogen acid to form aryl halide.
Gattermann reaction is a Sandmeyer reaction variant in which Cu powder replaces CuCl.
This substitution produces aryl halide more easily and under milder conditions than the Sandmeyer reaction.
21. Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is
(A) Hoffmann bromamide reaction
(B) Gabriel phthalimide reaction
(C) Sandmeyer reaction
(D) reaction with $\mathrm{NH}_{3}$
Ans: (B)
The reaction of transformation of primary alkyl halides to primary amines using potassium phthalimide is the Gabriel phthalimide reaction.
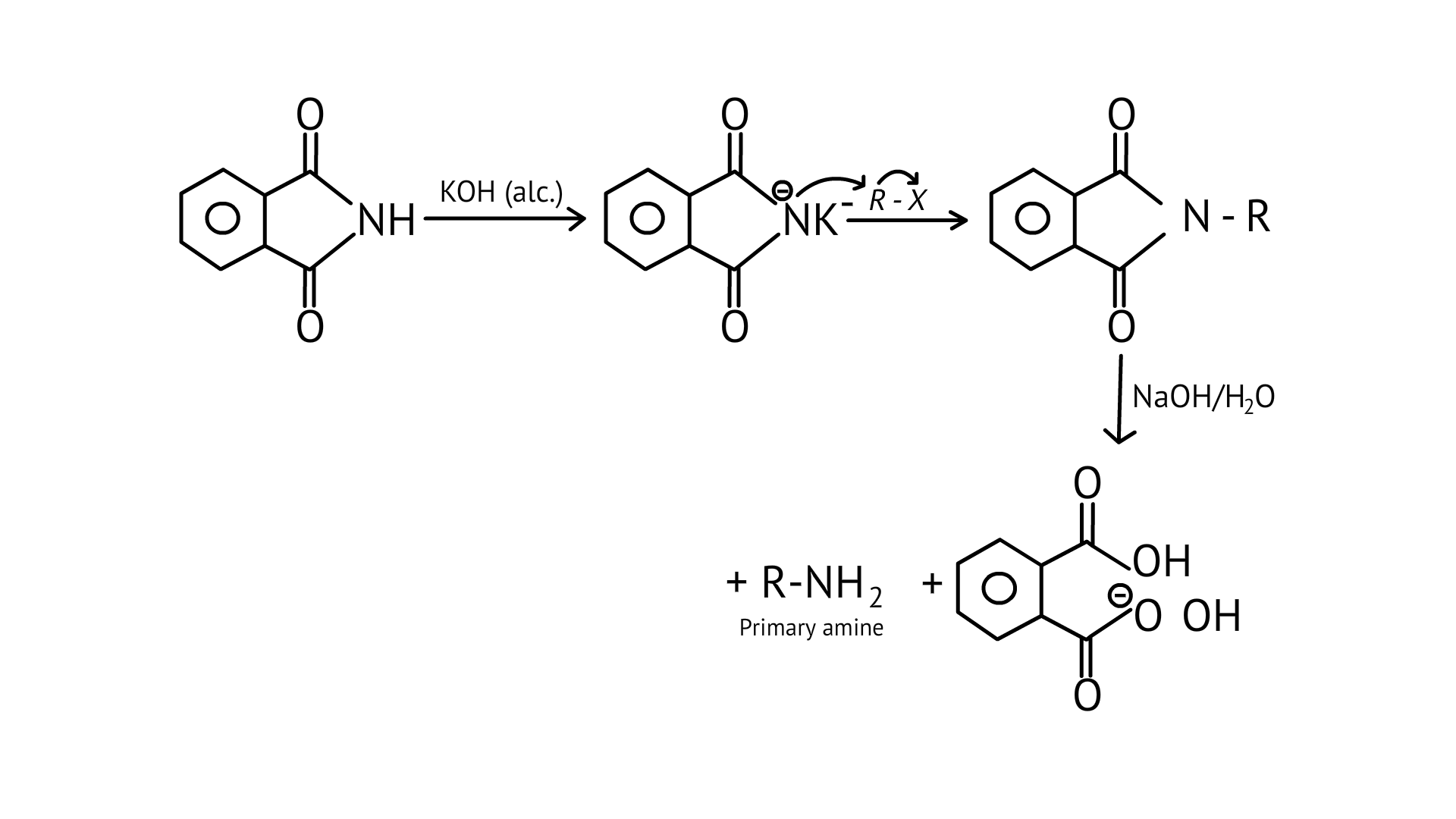
22. Which of the following compounds will not undergo azo coupling reaction with benzene diazonium chloride?
(A) Aniline
(B) Phenol
(C) Anisole
(D) Nitrobenzene
Ans: (D)
Weak electrophile such as Diazonium cation readily reacts with electron-rich compounds which are having electron-rich compounds such as hydroxyl group, amino group. They don’t react with electron-withdrawing groups like nitro groups. Therefore, nitrobenzene will not undergo an azo coupling reaction with benzene diazonium chloride.
23. Which of the following compounds is the weakest Bronsted base?
(A)
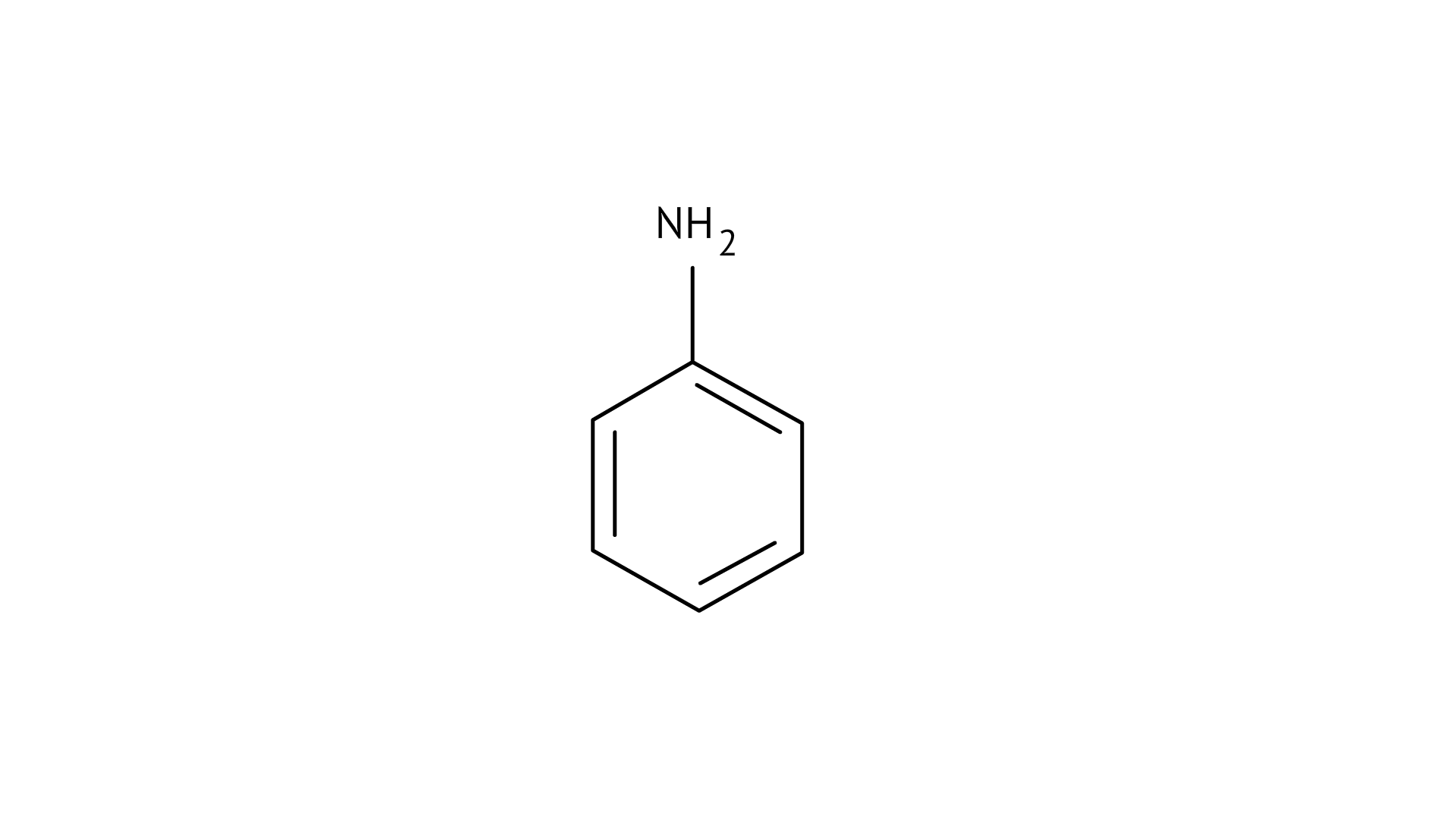
(B)
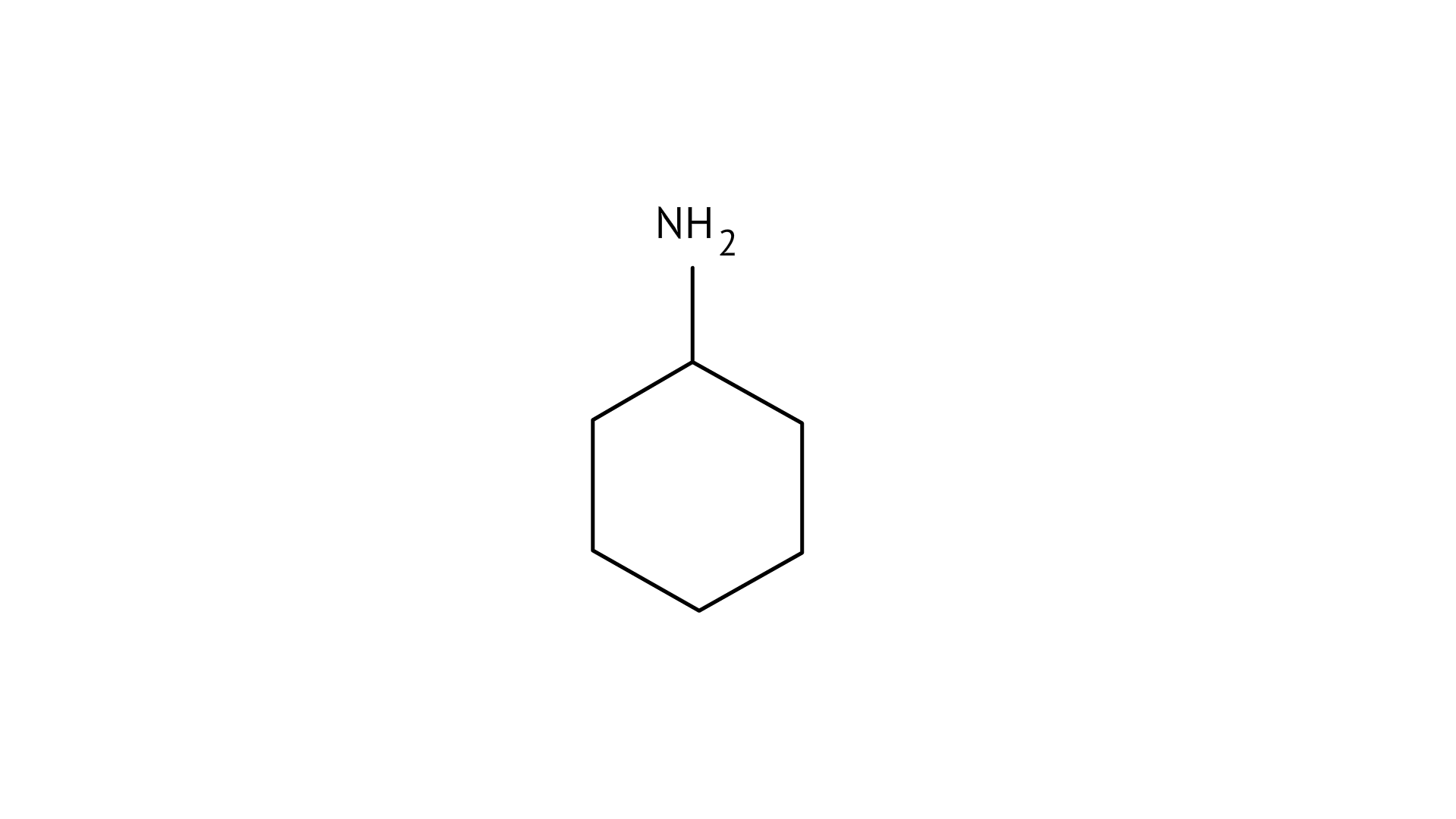
(C)
(D)
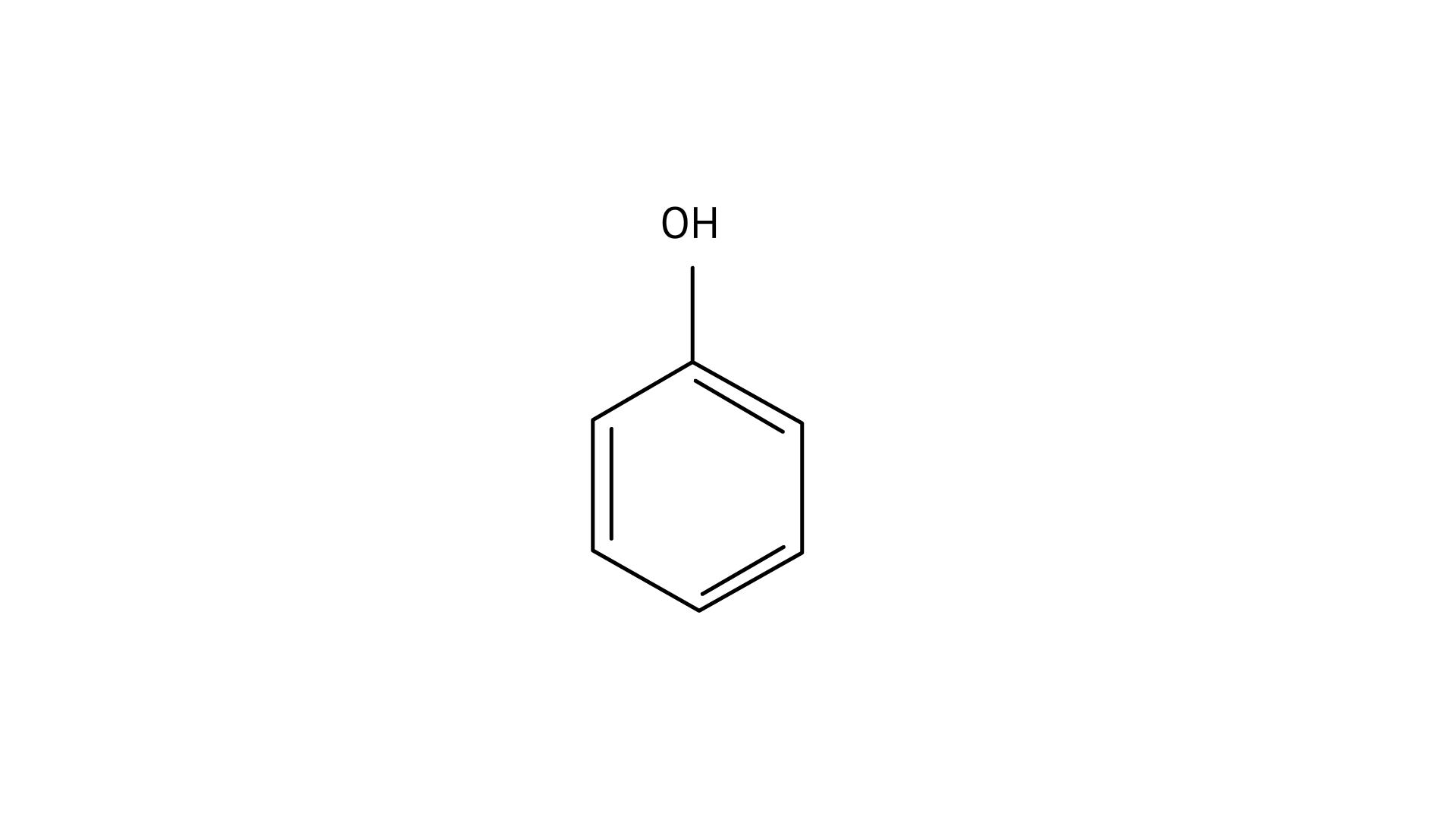
Ans: (D)
Since phenol is the strongest acid among the four options listed above, it is the weakest Brönsted base. The stronger the acid, the weaker the conjugate base.
Amines have a strong tendency to accept electrons thus they are a strong bronsted base while phenol is the strongest acid among all, therefore as per the relation of conjugative strong acids and weak bases, phenol is the weakest base.
24. Among the following amines’ the strongest Bronsted base is
(A)
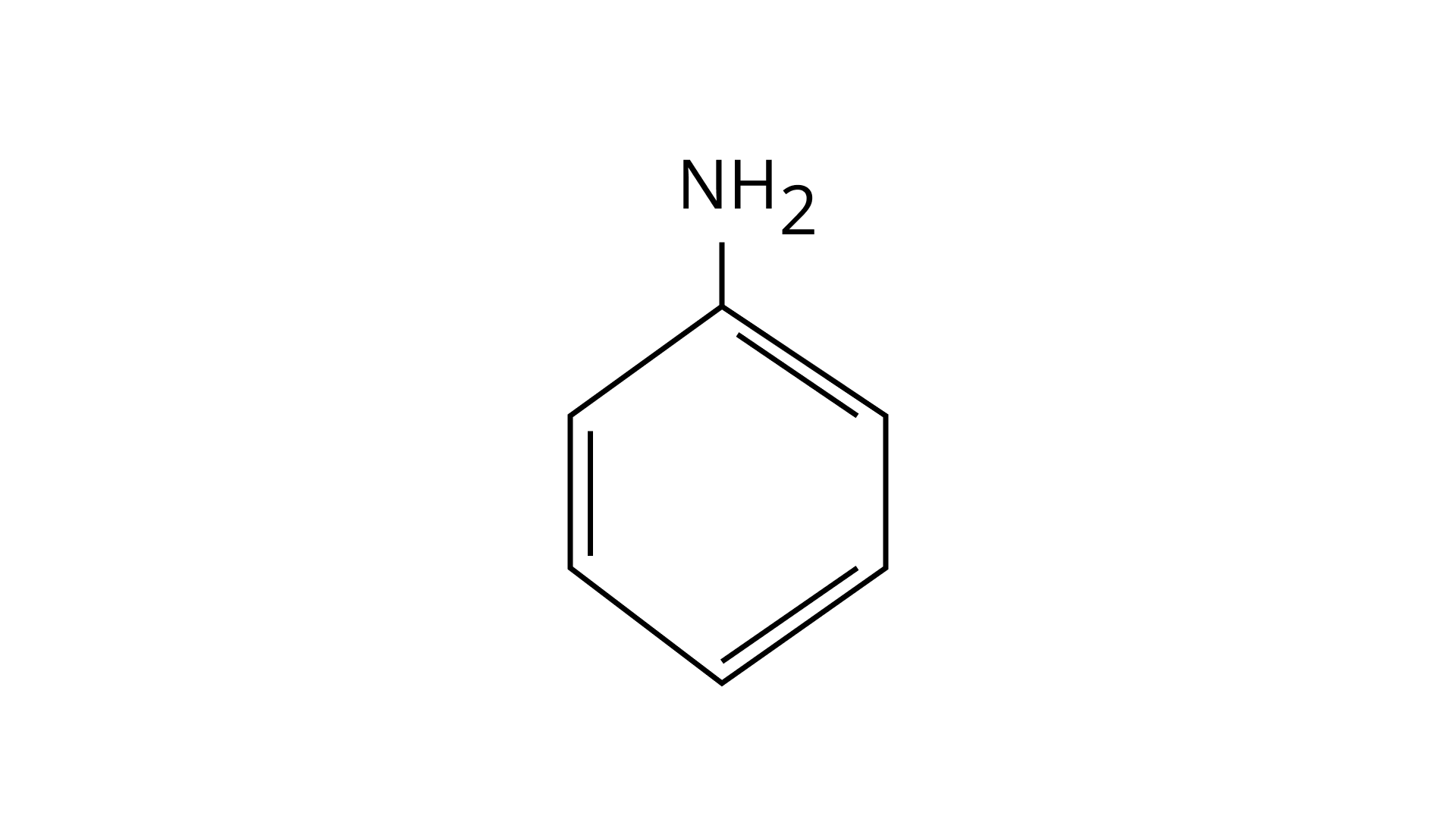
(B) $\mathrm{NH}_{3}$
(C)
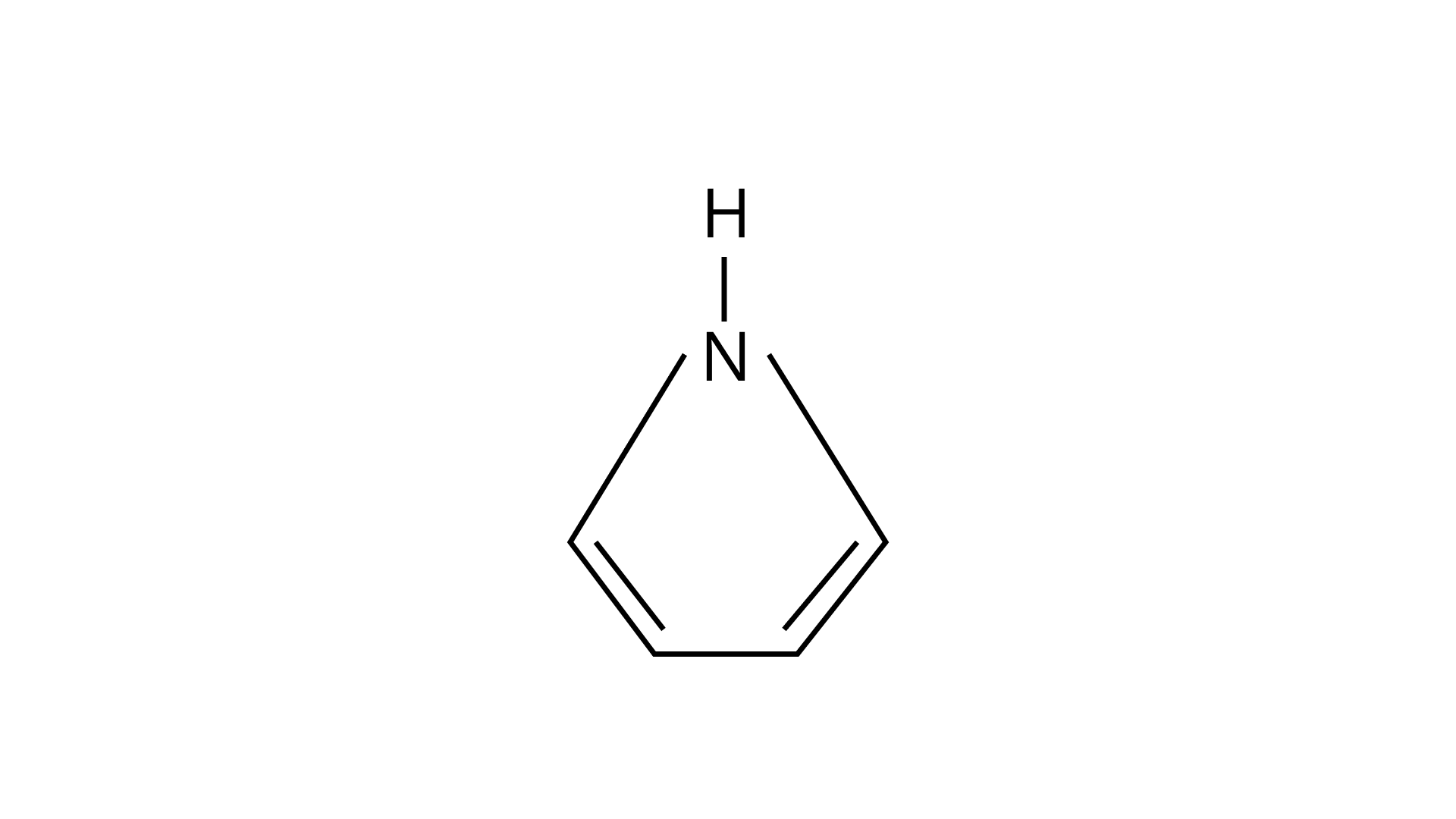
(D)
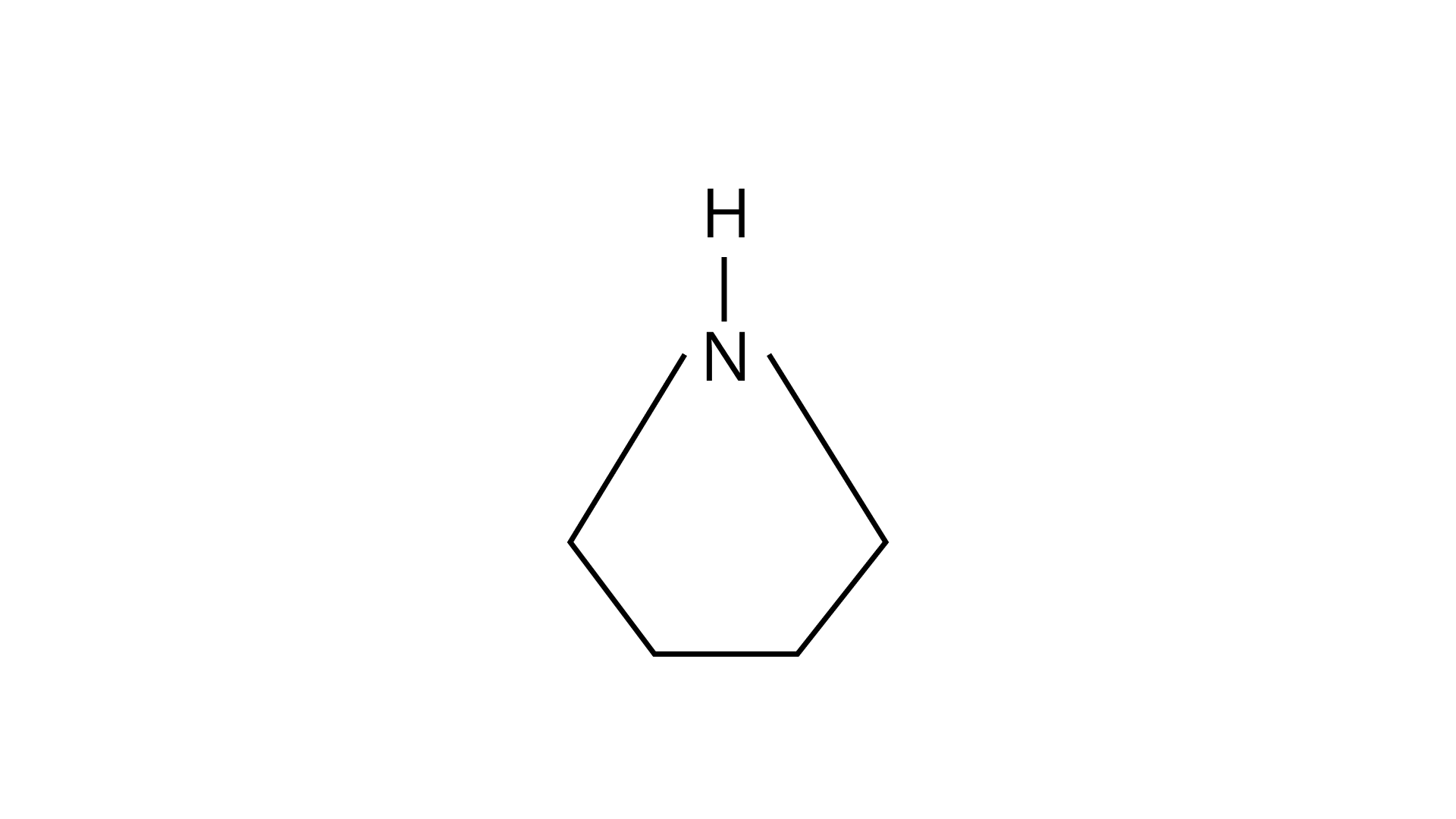
Ans: (D)
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance, and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
25. The correct decreasing order of basic strength of the following species is
$\mathrm{H}_{2} \mathrm{O}, \mathrm{NH}_{3}, \mathrm{OH}^{-}, \mathrm{NH}_{2}^{-}$
(A) $\mathrm{NH}_{2}^{-}>\mathrm{OH}^{*}>\mathrm{NH}_{3}>\mathrm{H}_{2} \mathrm{O}$
(B) $\mathrm{OH}^{-}>\mathrm{NH}_{2}^{-}>\mathrm{H}_{2} \mathrm{O}>\mathrm{NH}_{3}$
(C) $\mathrm{NH}_{3}>\mathrm{H}_{2} \mathrm{O}>\mathrm{NH}_{2}^{-}>\mathrm{OH}^{-}$
(D) $\mathrm{H}_{2} \mathrm{O}>\mathrm{NH}_{3}>\mathrm{OH}^{-}>\mathrm{NH}_{2}^{-}$
Ans: (A)
The basic strength of an atom is determined by its electron-donating capacity; in this case, the amide is the most basic due to the presence of a negative charge and two lone pairs of electrons on the nitrogen atom.
26. Which of the following should be most volatile?
(i) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{NH}_{2}$
(ii) $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{~N}$
(iii)
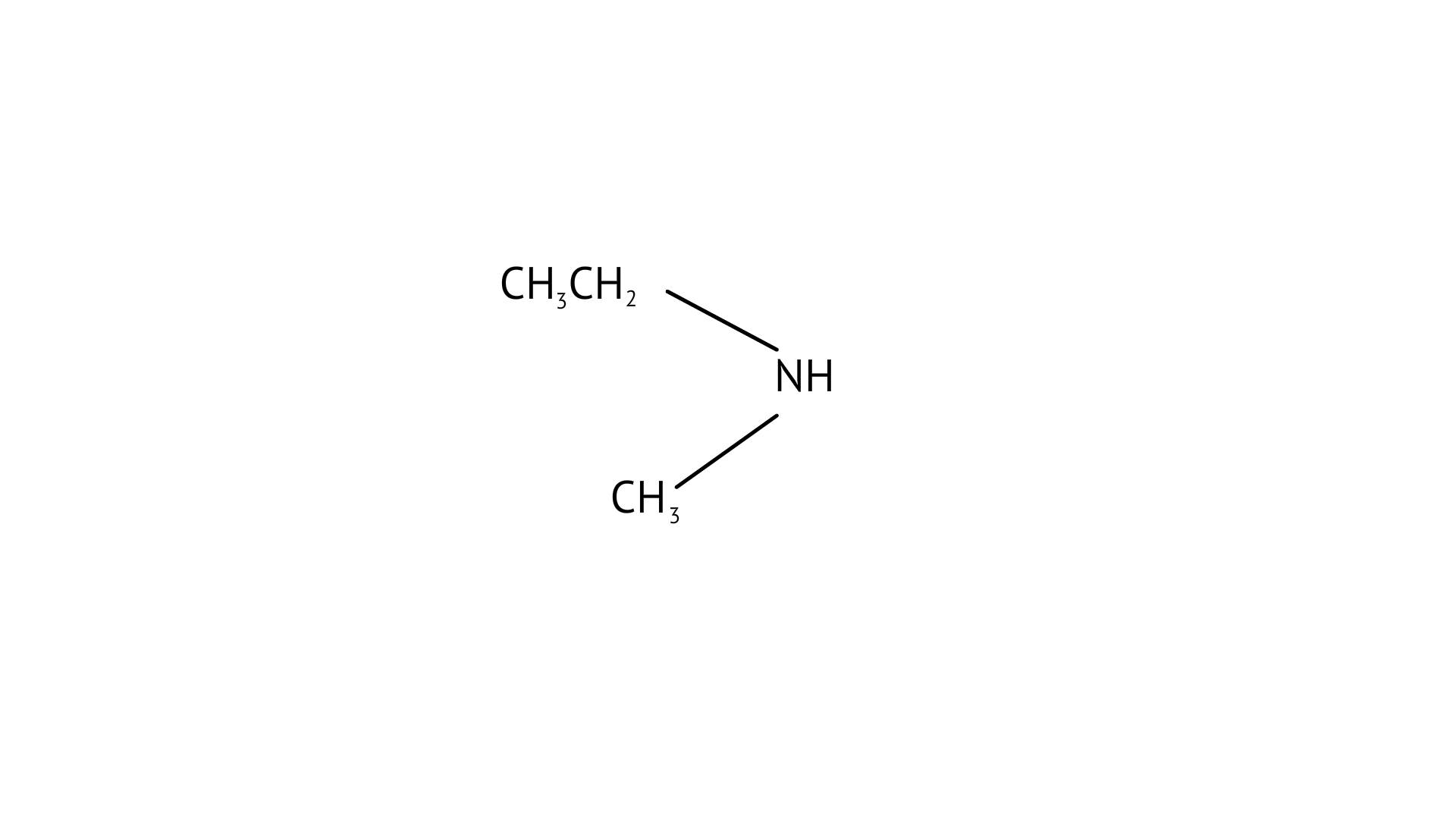
(iv) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{3}$
(A) II
(B) IV
(C) I
(D) III
Ans: (B)
Among primary, secondary, and tertiary amines, tertiary amines are the most volatile compounds as they don’t have any strong intermolecular H-bonding between N-H like the primary and secondary amines have.
So due to weak dipole-dipole interactions, (B) will be the most volatile.
27. Which of the following methods of preparation of amines will not give the same number of carbon atoms in the chain of amines as in the reactant?
(A) Reaction of nitrile with $\mathrm{LiAlH}_{4}$
(B) Reaction of amide with $\mathrm{LiAlH}_{4}$ followed by treatment with water.
(C) Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis
(D) Treatment of amide with bromine in aqueous solution of sodium hydroxide.
Ans: (D)
Only treatment of amide with $\mathrm{Br}_{2}$ in aqueous $\mathrm{NaOH}$ solution yields an amine with fewer carbon atoms than the reactant, whereas $\mathrm{RCONH}_{2} \mathrm{Br}_{2} / \mathrm{NaOH} \mathrm{RNH}_{2}$ all other reactions yield an amine with the same number of carbon atoms as the reactant.
Multiple Choice Questions (Type- II)
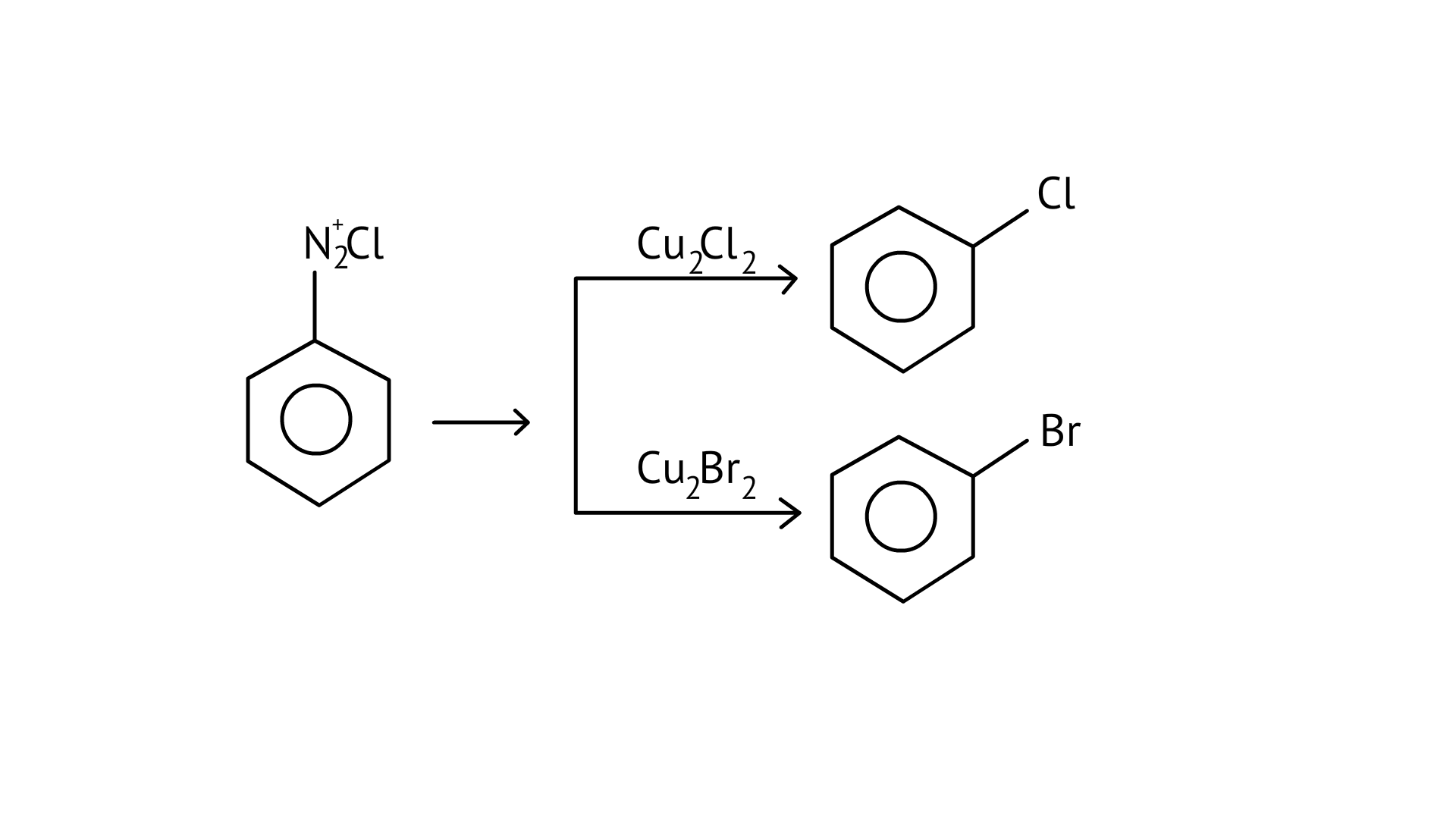
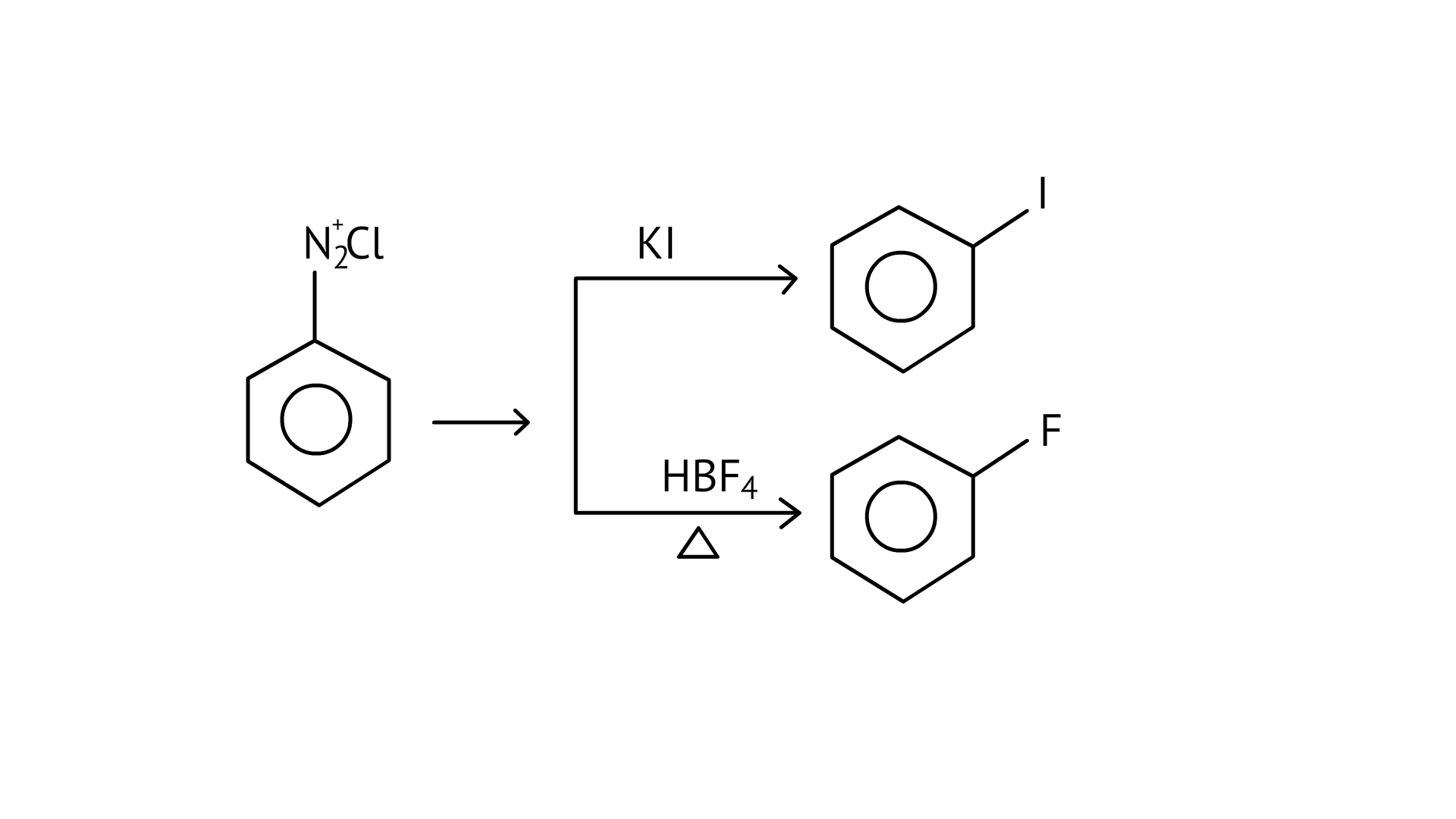
28. Which of the following cannot be prepared by Sandmeyer’s reaction?
(A) Chlorobenzene
(B) Bromobenzene
(C) Iodobenzene
(D) Fluorobenzene
Ans: (C and D)
Chlorobenzene and bromobenzene are prepared by the sandmeyer’s reaction.
While fluorobenzene and iodobenzene are prepared by simple heating of diazonium salt with aqueous KI Solution.
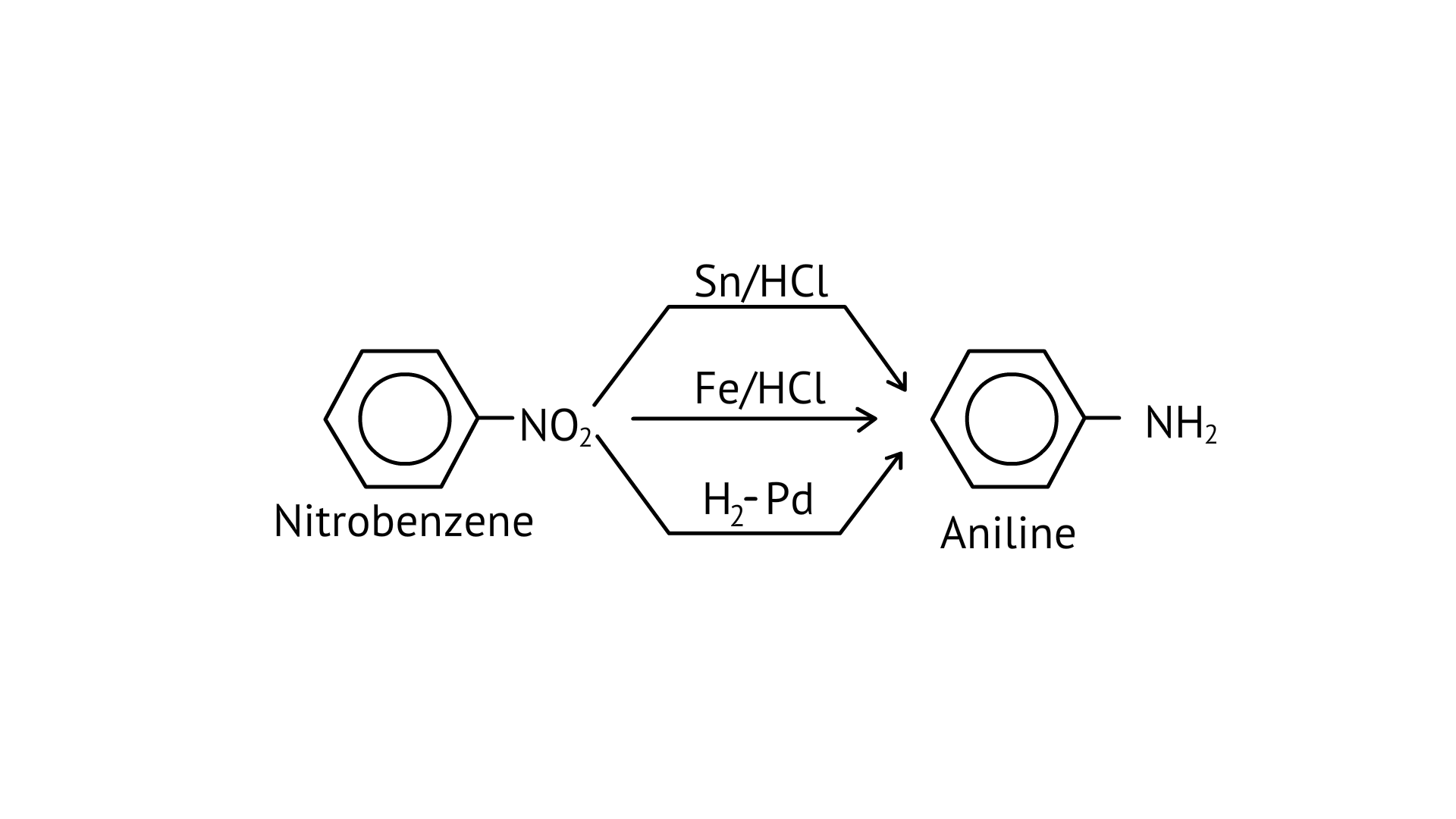
29. Reduction of nitrobenzene by which of the following reagents give aniline?
(A) $\mathrm{Sn} / \mathrm{HCl}$
(B) $\mathrm{Fe} / \mathrm{HCl}$
(C) $\mathrm{H}_{2}-\mathrm{Pd}$
(D) $\mathrm{Sn} / \mathrm{NH}_{4} \mathrm{OH}$
Ans: (A, B & C)
Nitro compounds are reduced to amines by passing hydrogen gas through an acidic medium containing finely divided nickel, palladium, or platinum, as well as by reduction with metals in an acidic medium.
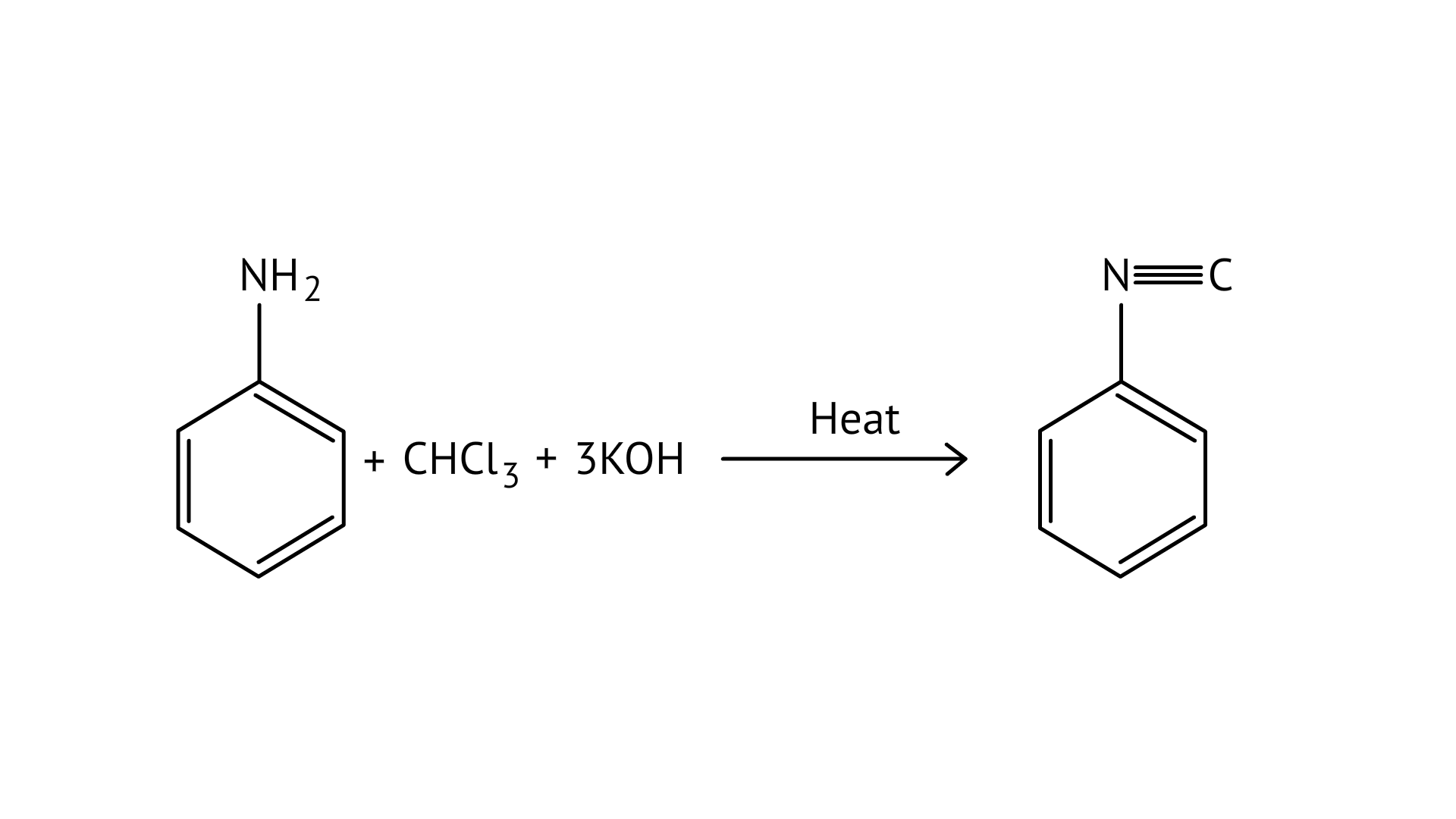
30. Which of the following species are involved in the carbylamine test?
(A) R-NC
(B) $\mathrm{CHCl}_{3}$
(C) $\mathrm{COCl}_{2}$
(D) $\mathrm{NaNO}_{2}+\mathrm{HCl}$
Ans: (A and B)
When aliphatic and aromatic primary amines are heated with chloroform and ethanolic potassium hydroxide, they form isocyanides or carbylamines, which have a foul odor. This reaction does not occur in secondary or tertiary amines.
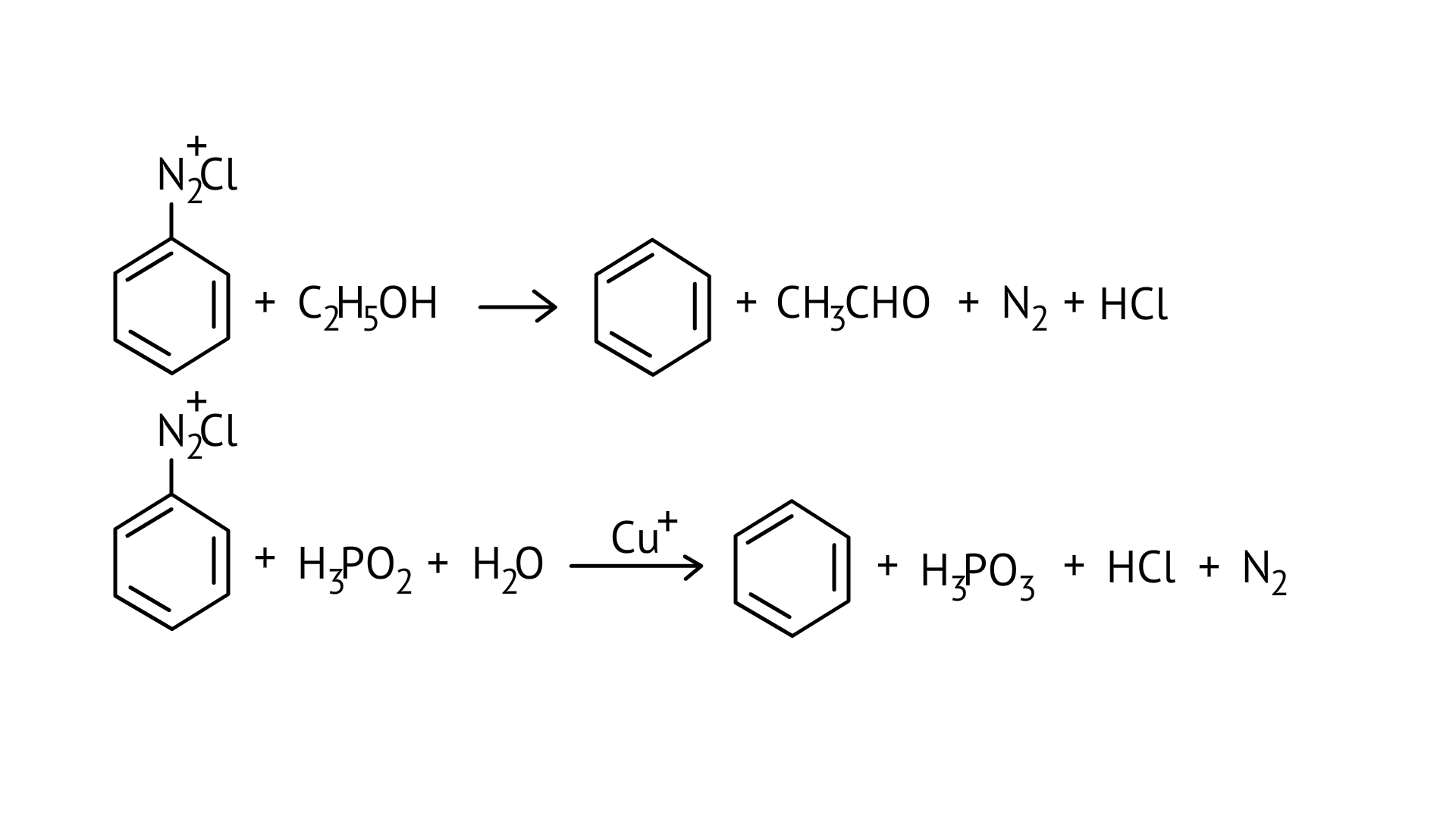
31. The reagents that can be used to convert benzene diazonium chloride to benzene are
(A) $\mathrm{SnCl}_{2} / \mathrm{HCl}$
(B) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{OH}$
(C) $\mathrm{H}_{3} \mathrm{PO}_{2}$
(D) $\mathrm{LiAlH}_{4}$
Ans: (B and C)
Certain mild reducing agents like hypophosphorous acid (phosphinic acid) or ethanol reduce diazonium salts to arenes and themselves get oxidised to phosphorous acid and ethanol, respectively.
32. The Product of the following reaction is.
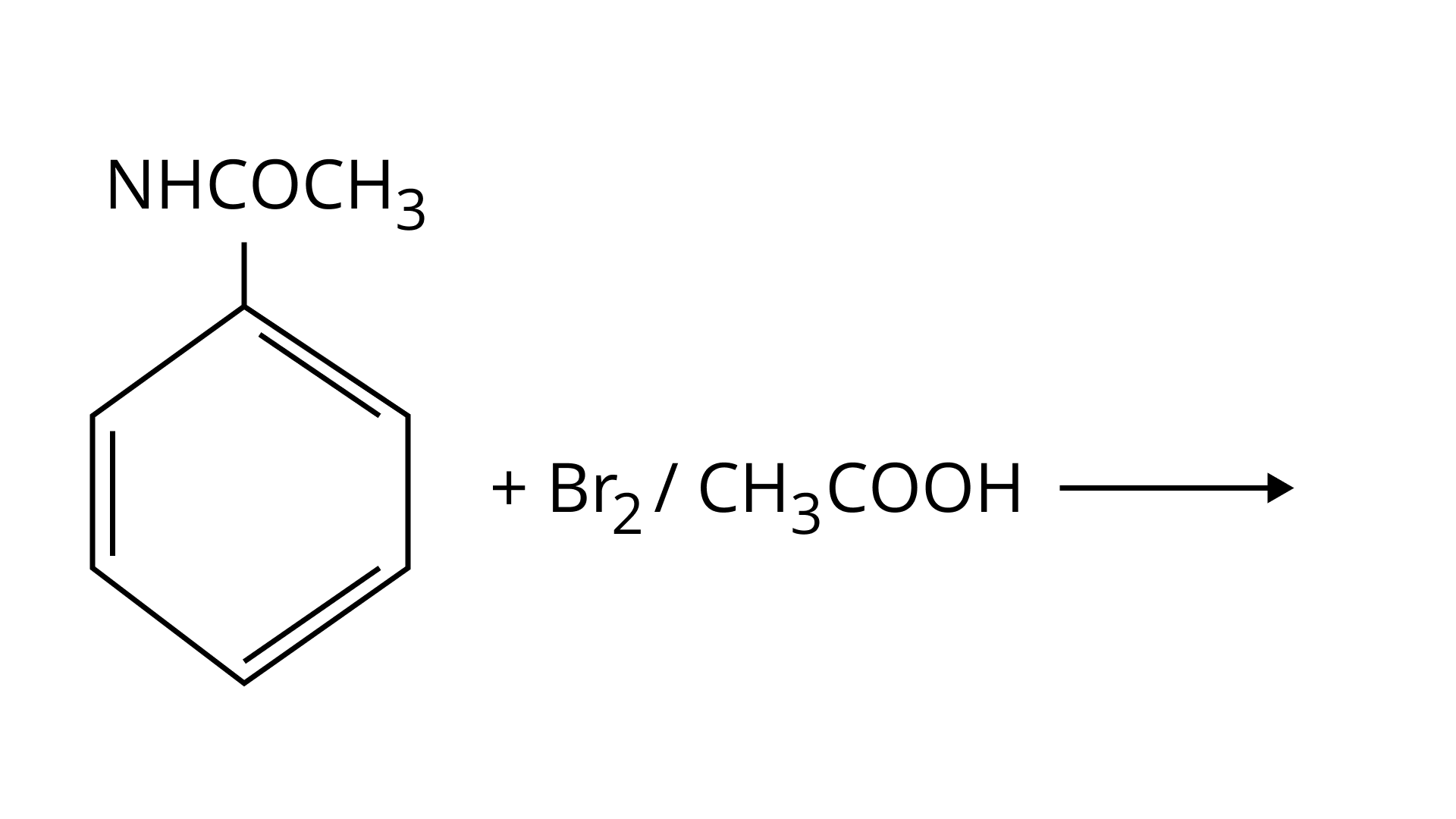
(A)
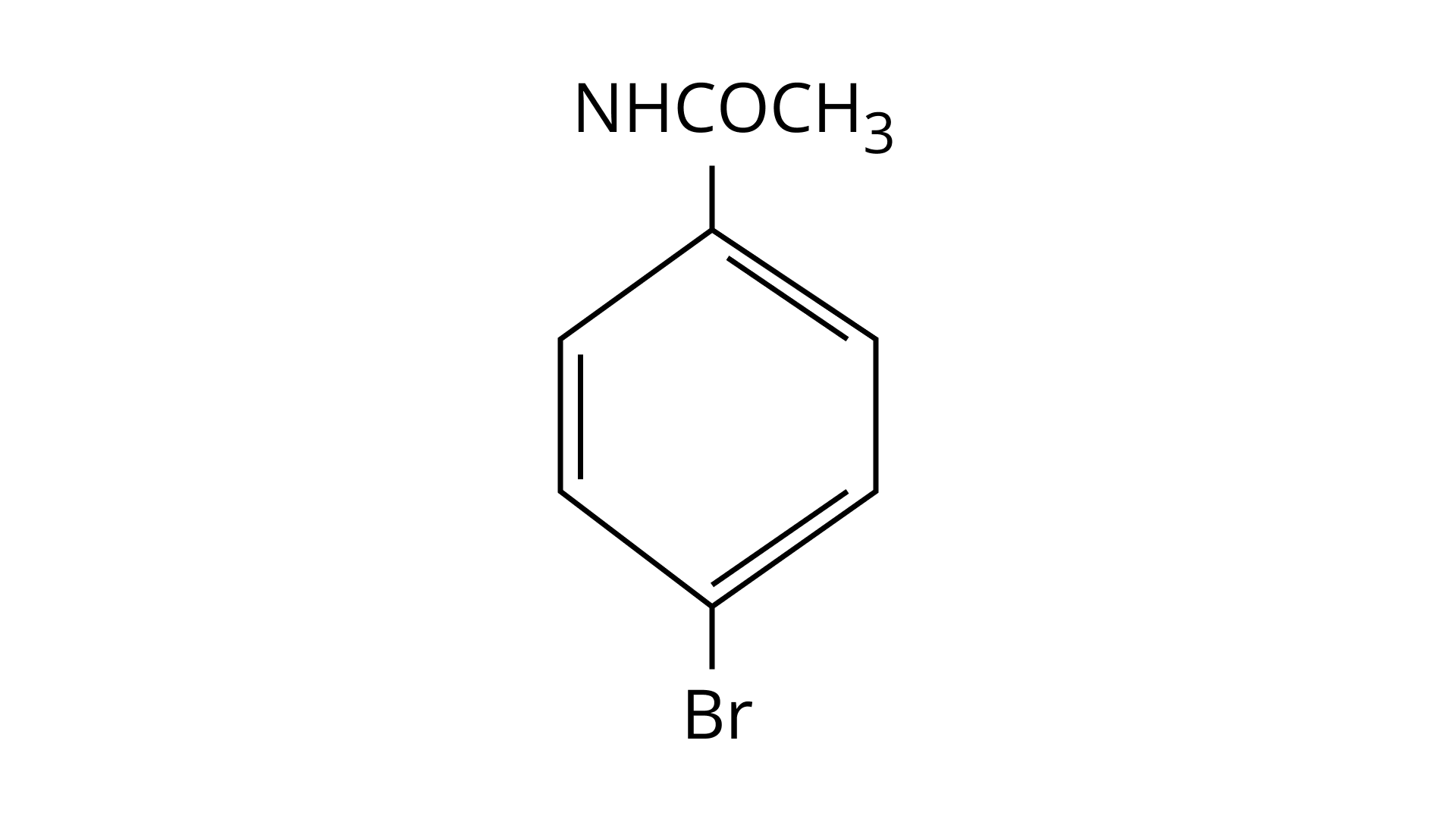
(B)
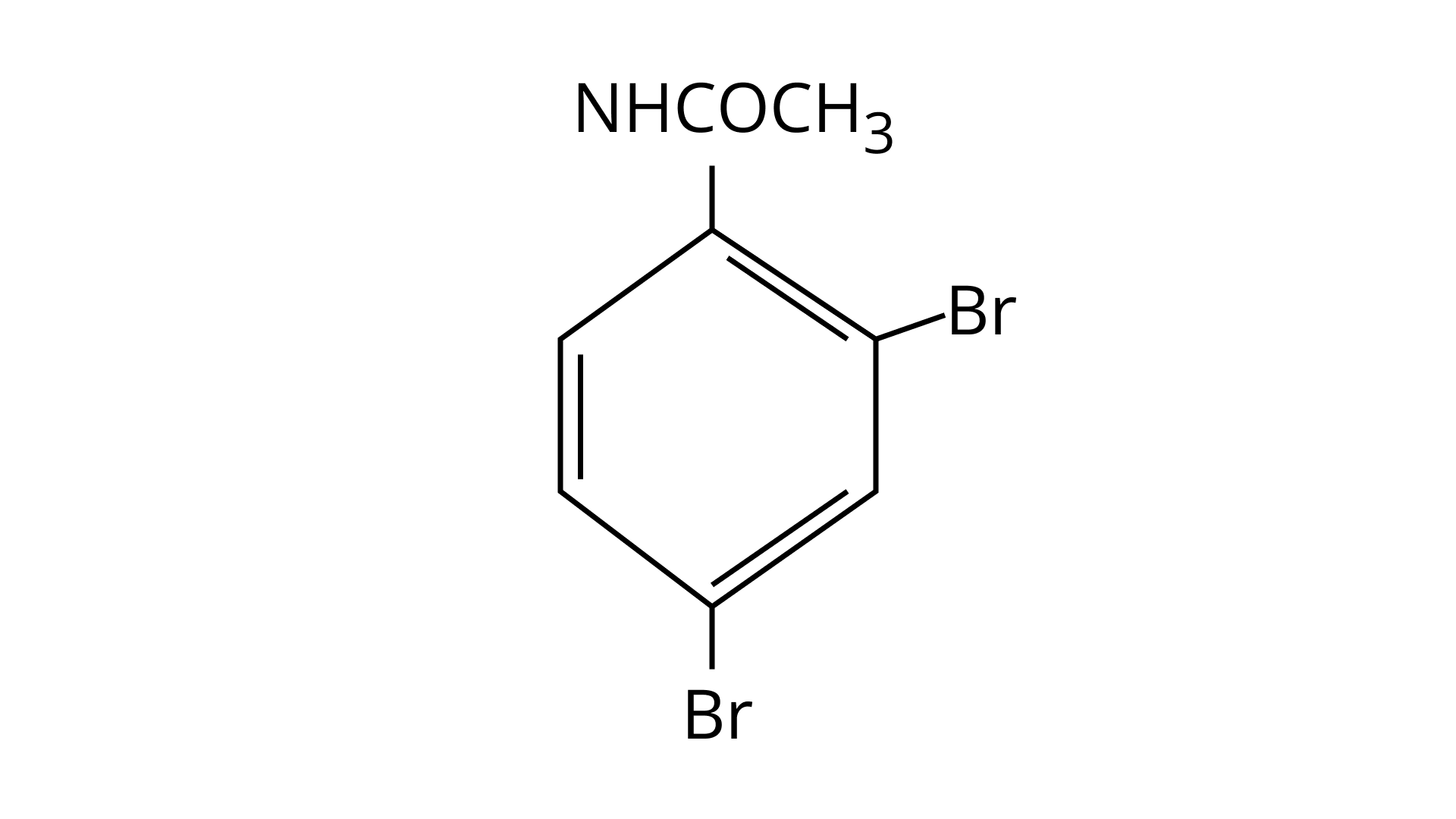
(C)
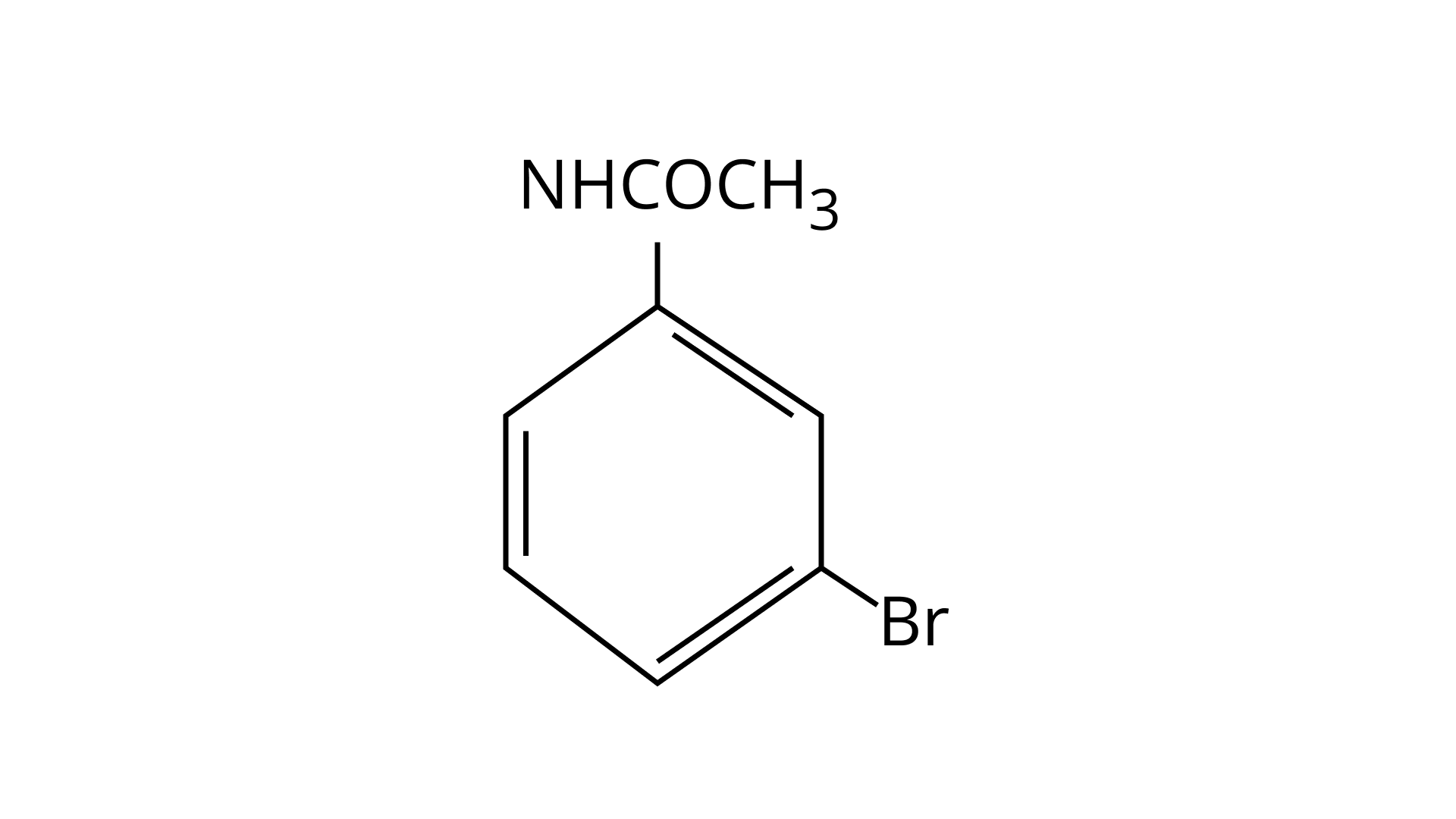
(D)
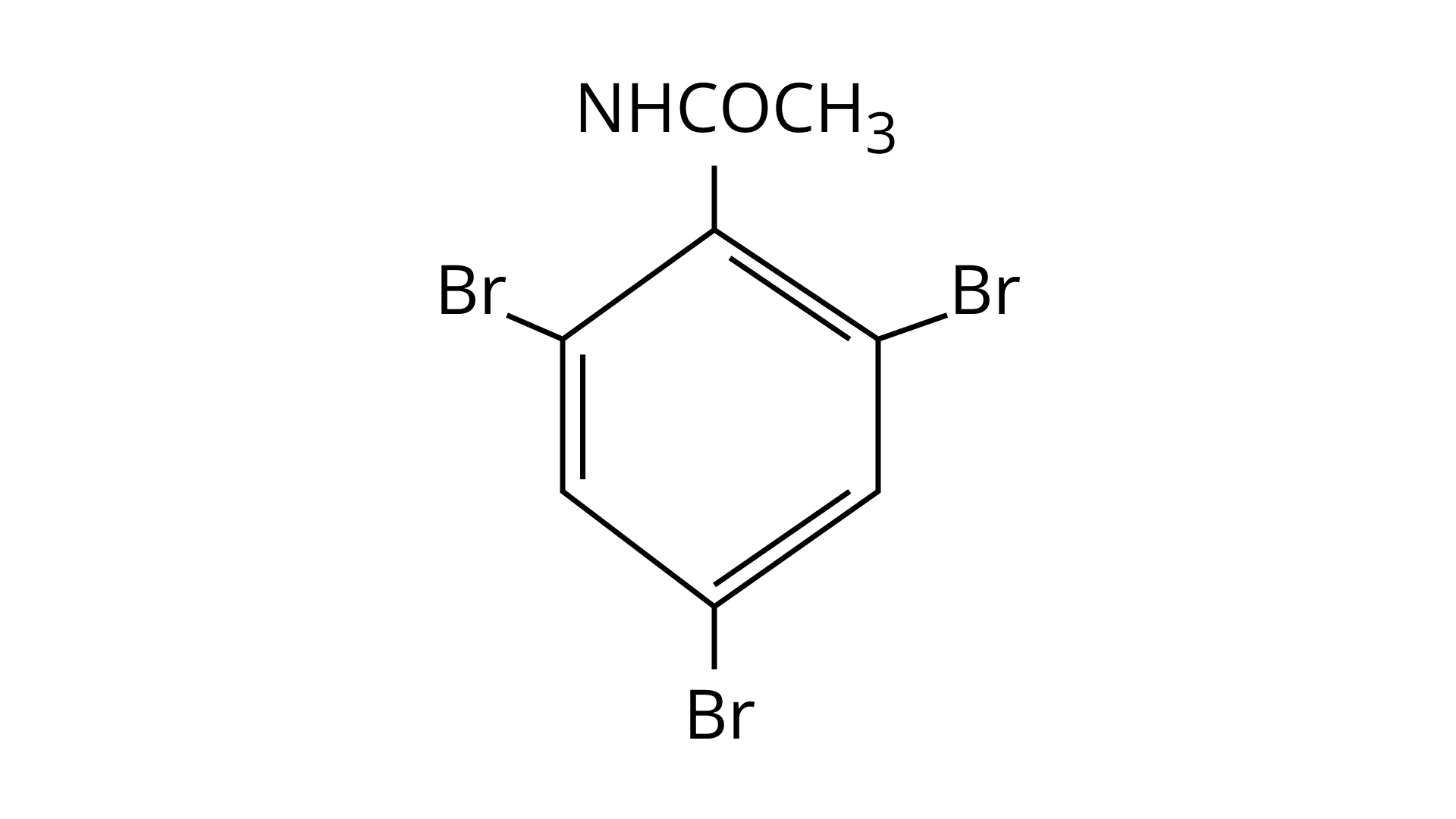
Ans: (A and B)
$\mathrm{NHCOCH}_{3}$ is an ortho- para directing group so ortho-para products are formed.
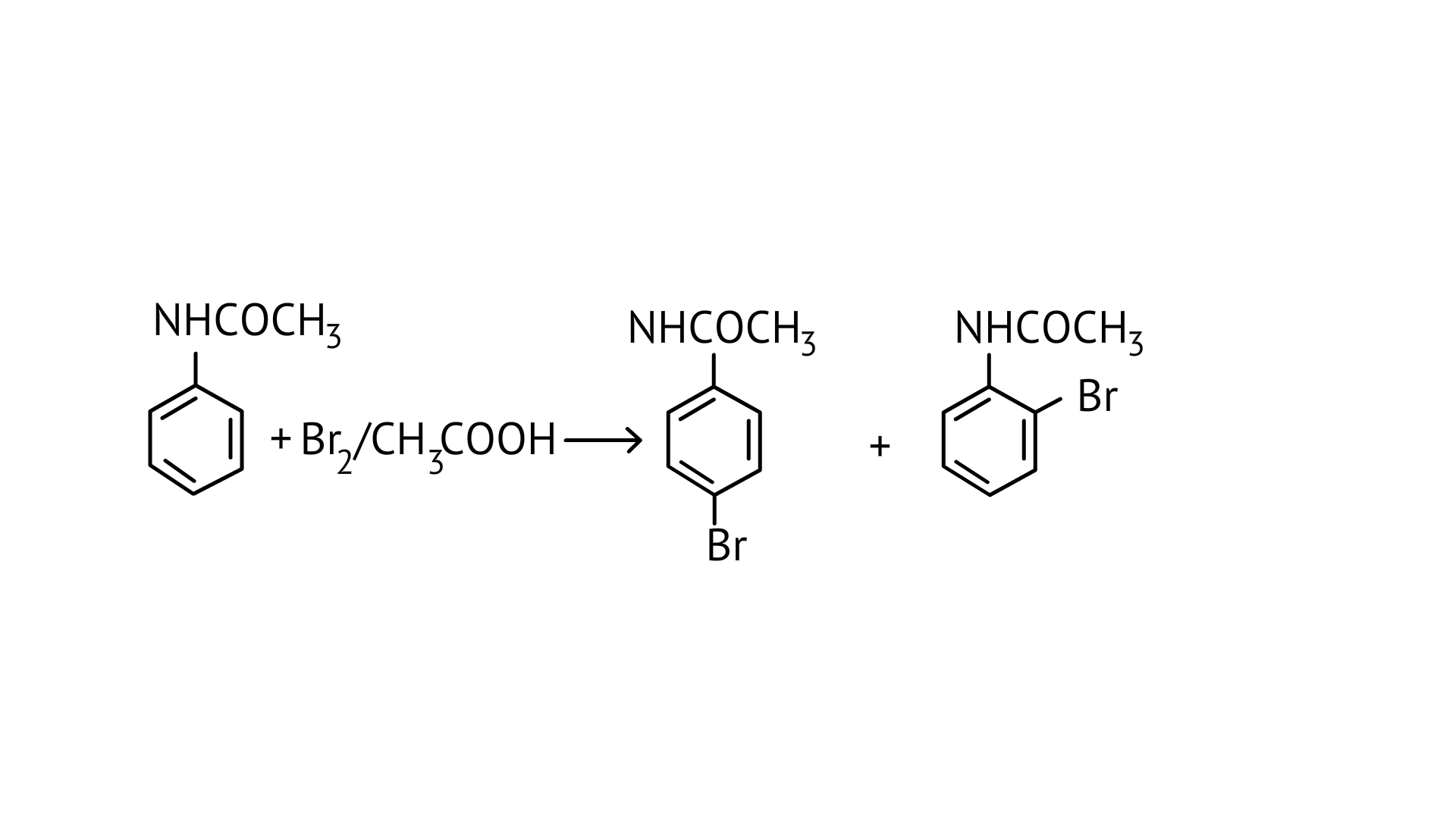
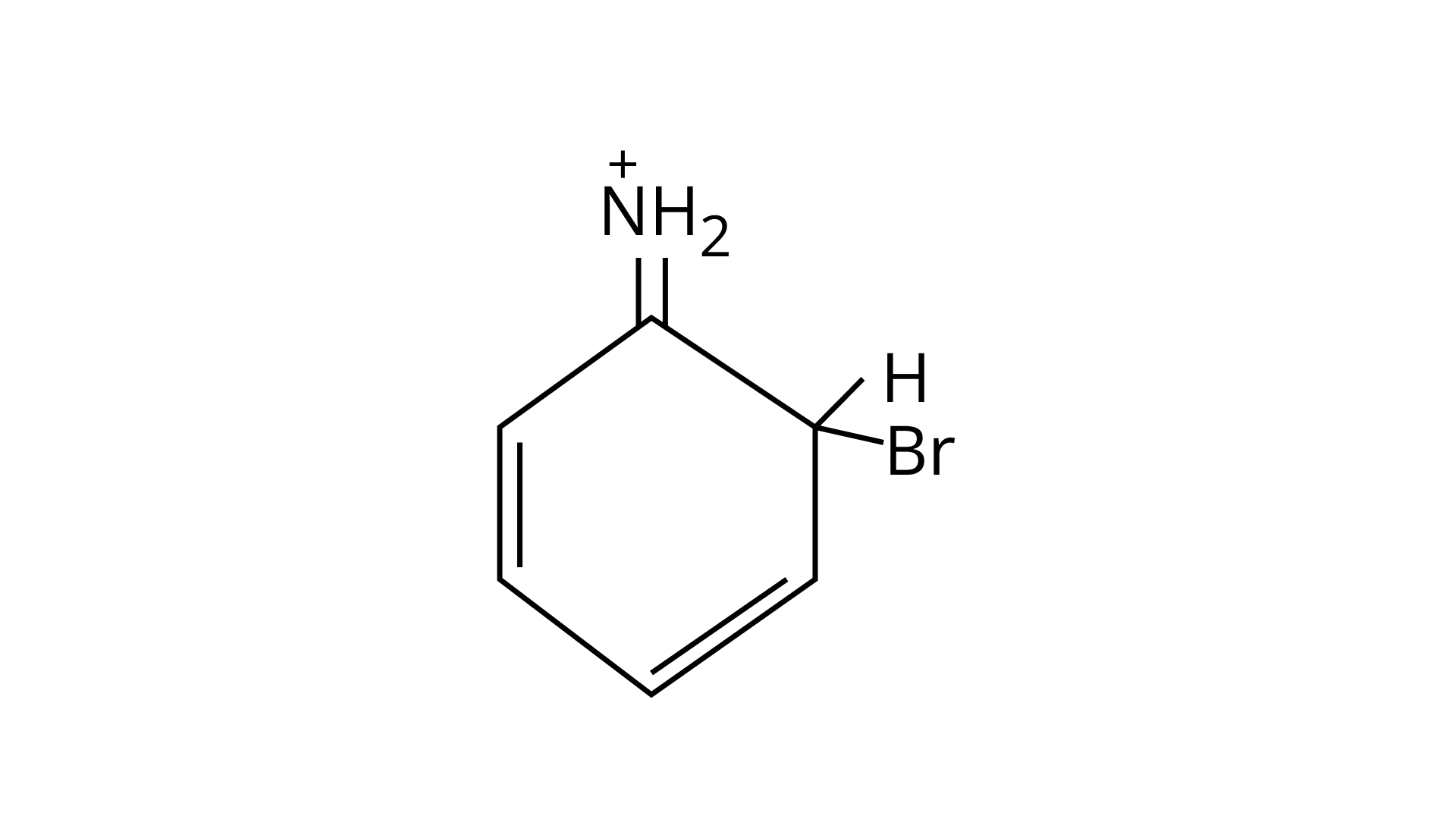
33. Arenium ion involved in the bromination of aniline is.
(A)
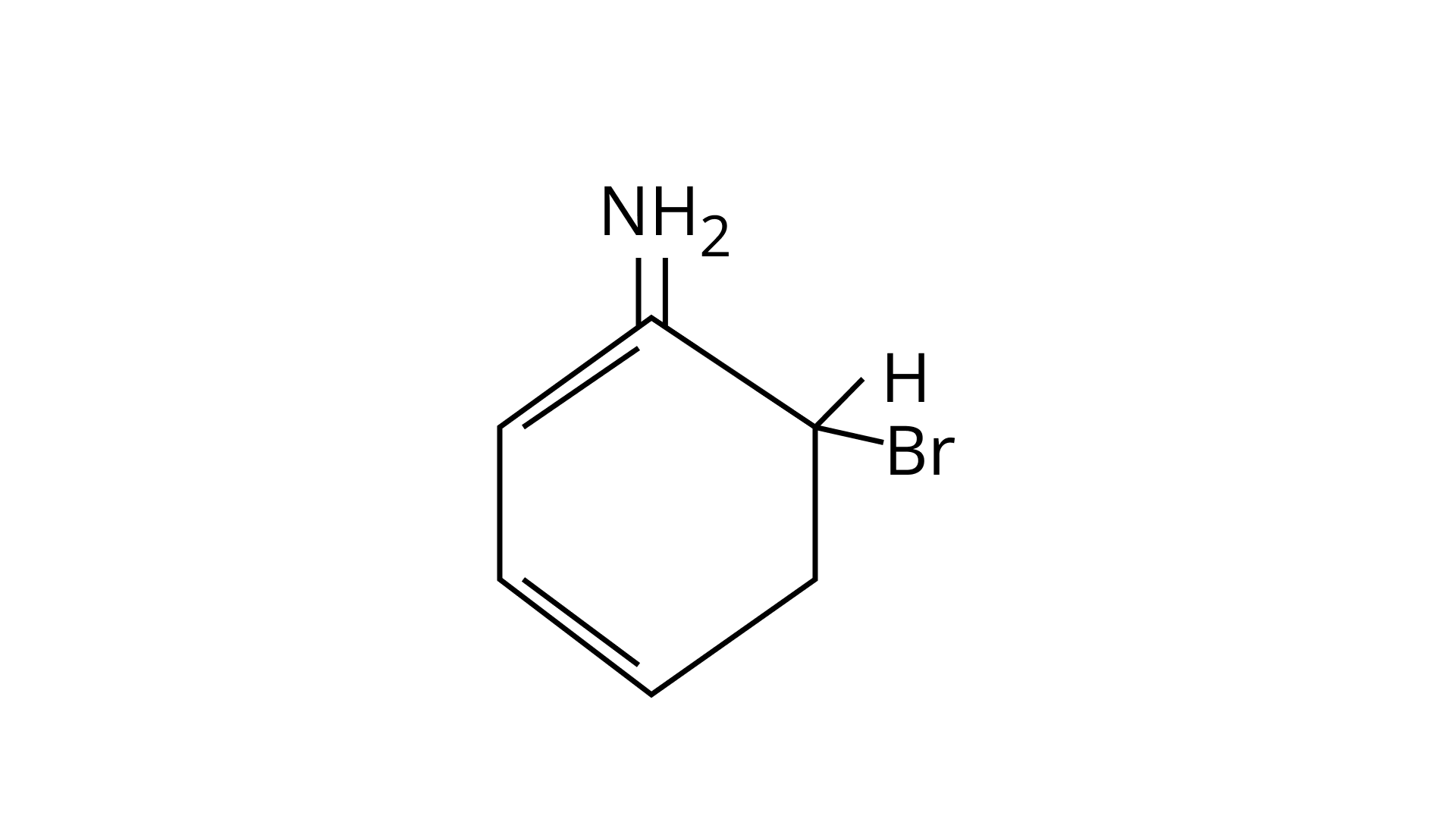
(B)
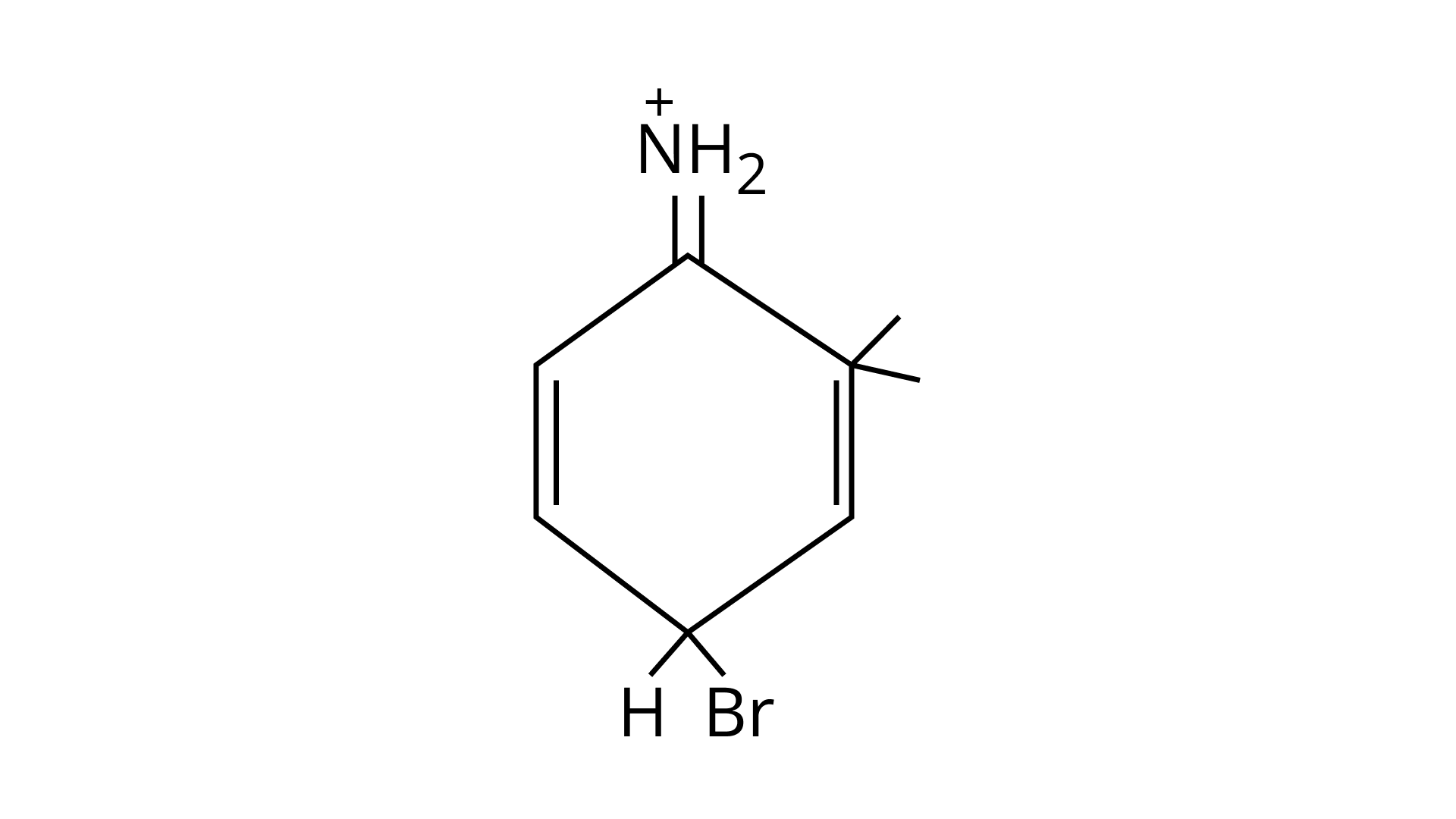
(C)
(D)
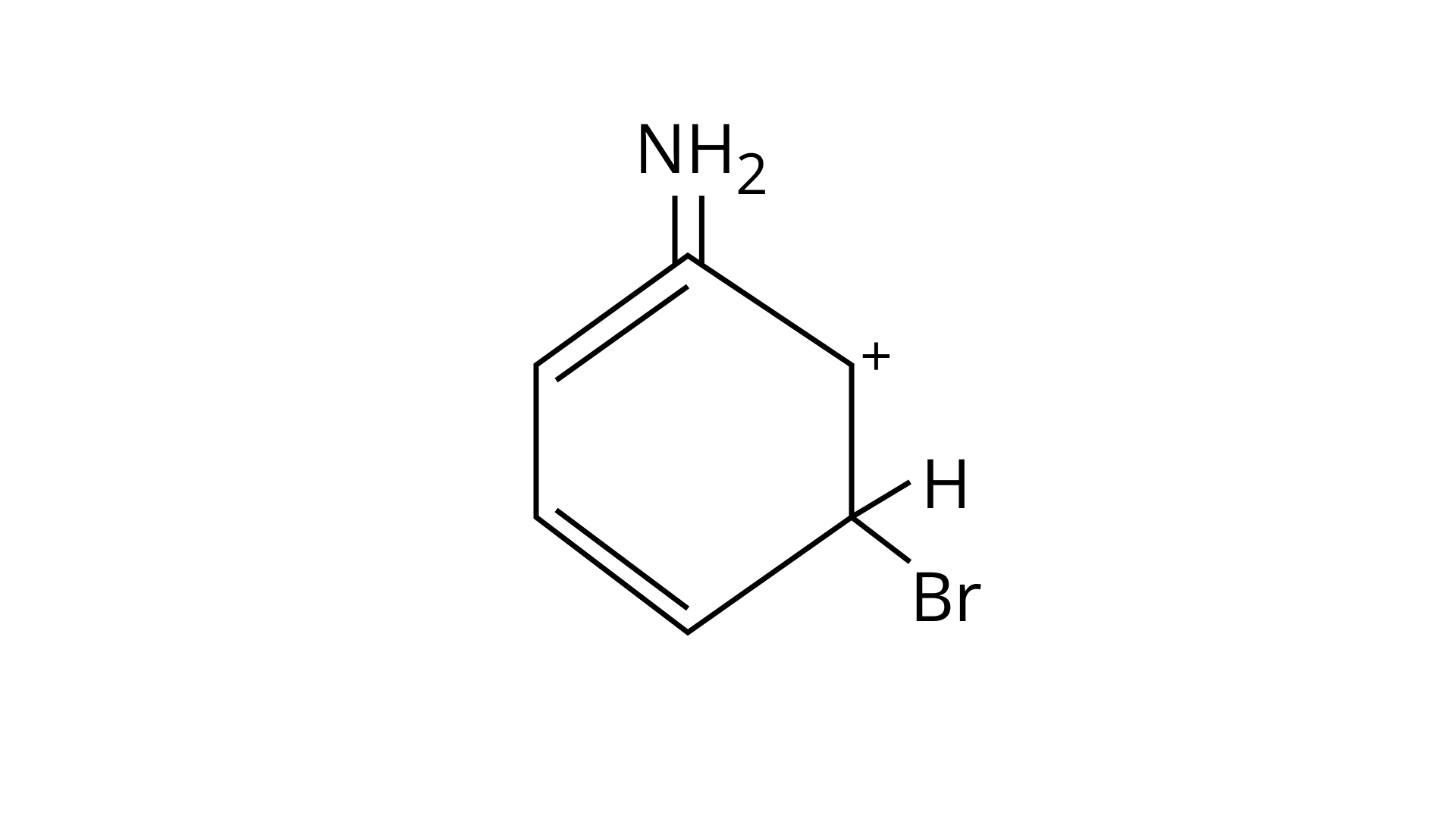
Ans: (A, B & C)
As the $\mathrm{NH}_{2}$ group is Ortho para directing group, so $\mathrm{Br}^{+}$attacks only at the o and p positions thus the products formed corresponds to the option A, B and C.
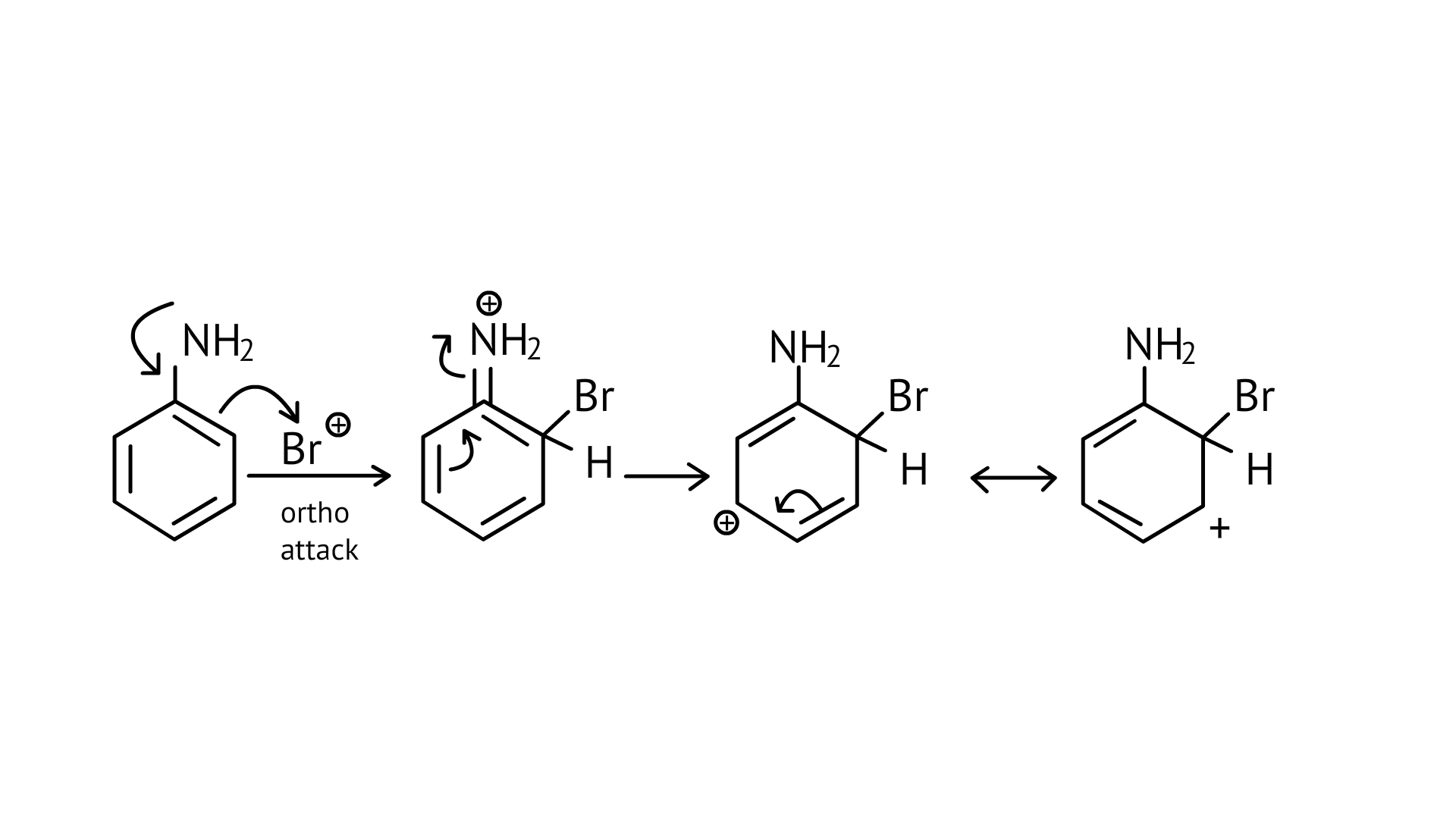
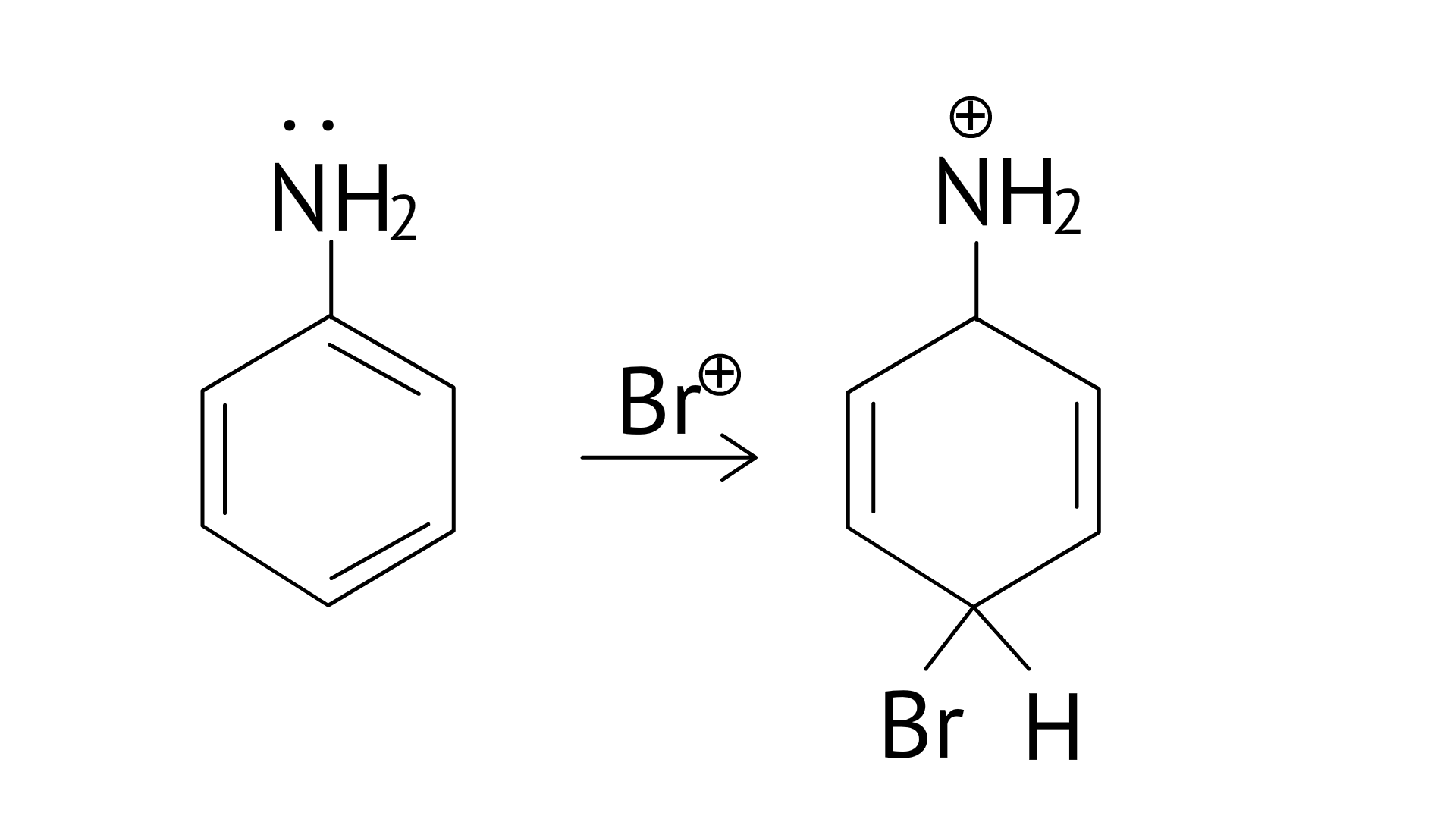
34. Which of the following amines can be prepared by Gabriel synthesis?
(A) Isobutyl amine
(B) 2-Phenylethylamine
(C) N-Methylbenzylamine
(D) Aniline
Ans: (A and B)
Gabriel synthesis is a method for producing primary amines. When phthalimide is treated with ethanolic potassium hydroxide, it forms a potassium salt of phthalimide, which when heated with an alkyl halide and then alkaline hydrolyzed yields the corresponding primary amine.
35. Which of the following reactions are correct?
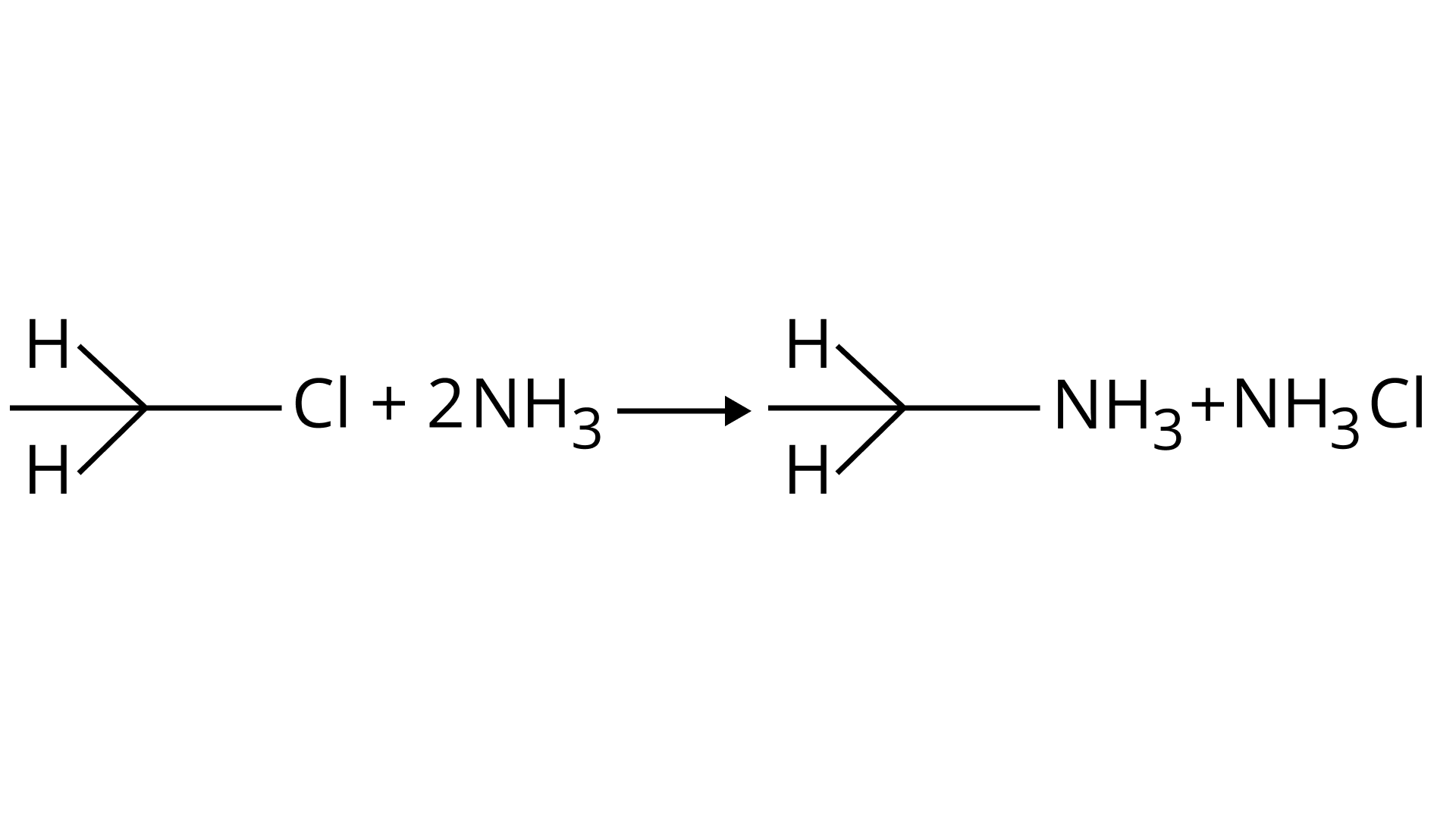
(A)
(B)
(C)
(D)
Ans: (A and C)
(a) Primary alkyl halides react with ammonia to give primary amines. Correct reaction.
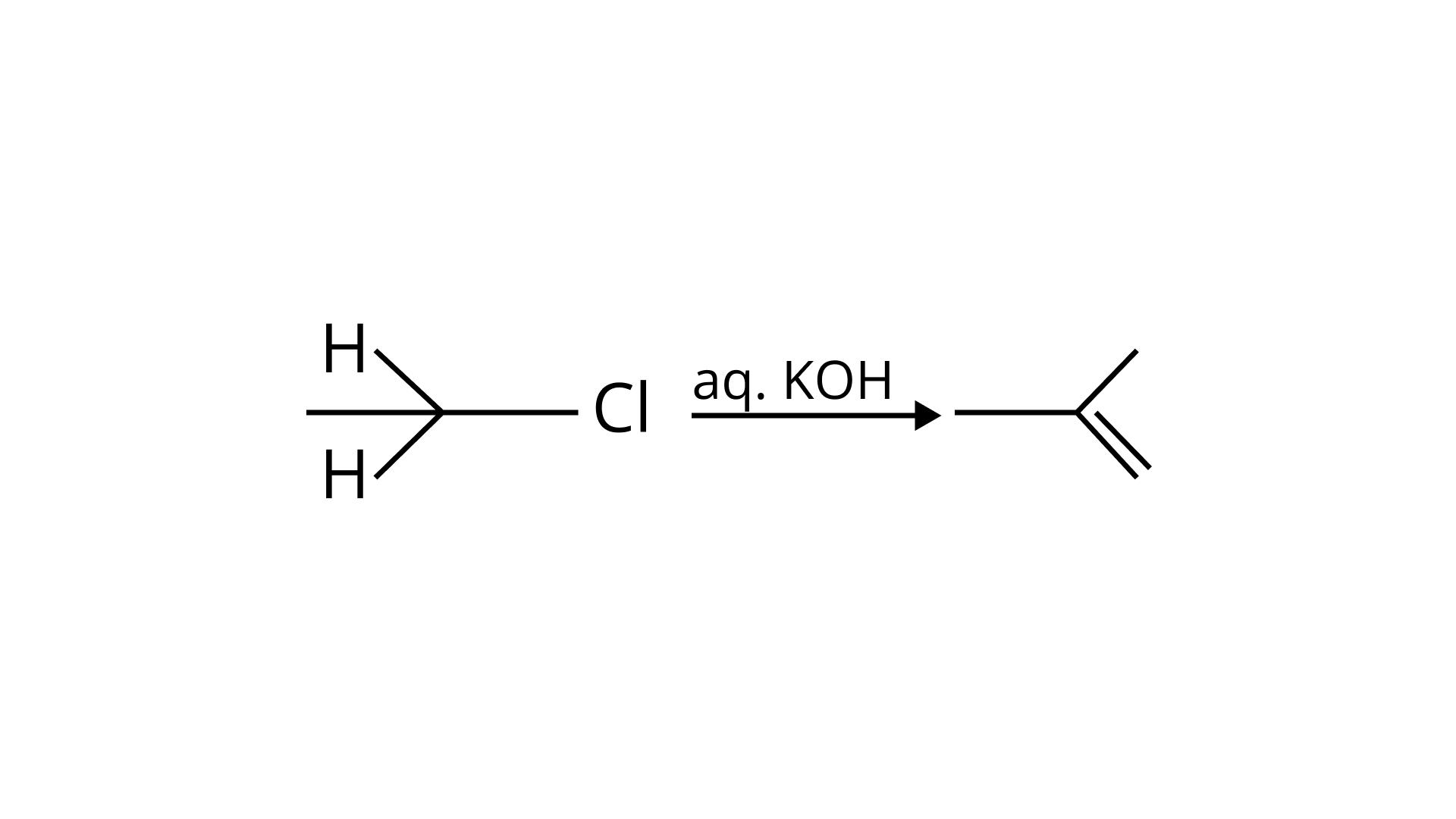
(B) Elimination process doesn’t occur with aq. KOH, hydrolysis process takes place. (B) is incorrect.
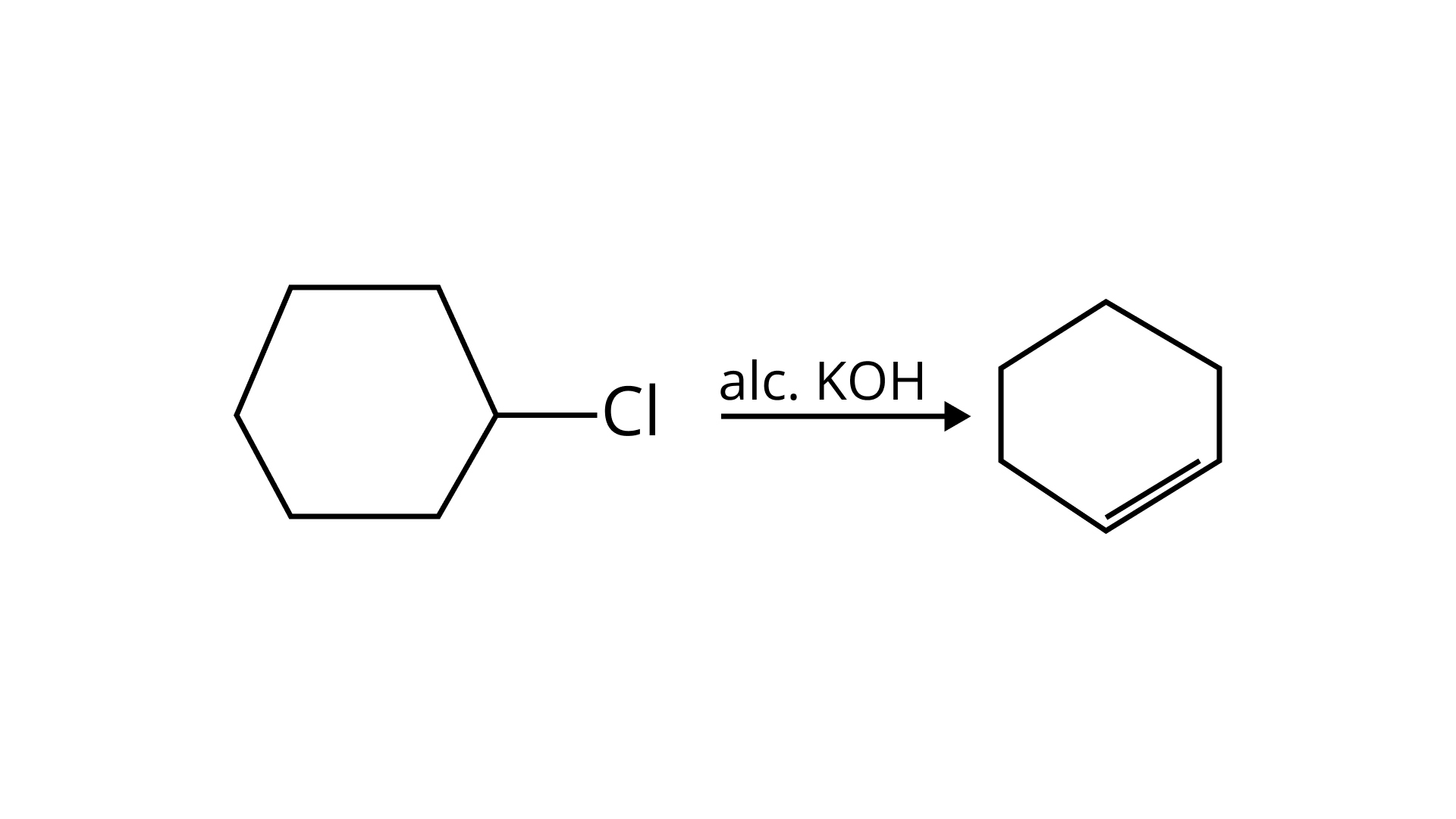
(c) Dehydrohalogenation I.e. elimination of HCl occurs which produces alkene as the product takes place with alc. KOH . (c) is correct.
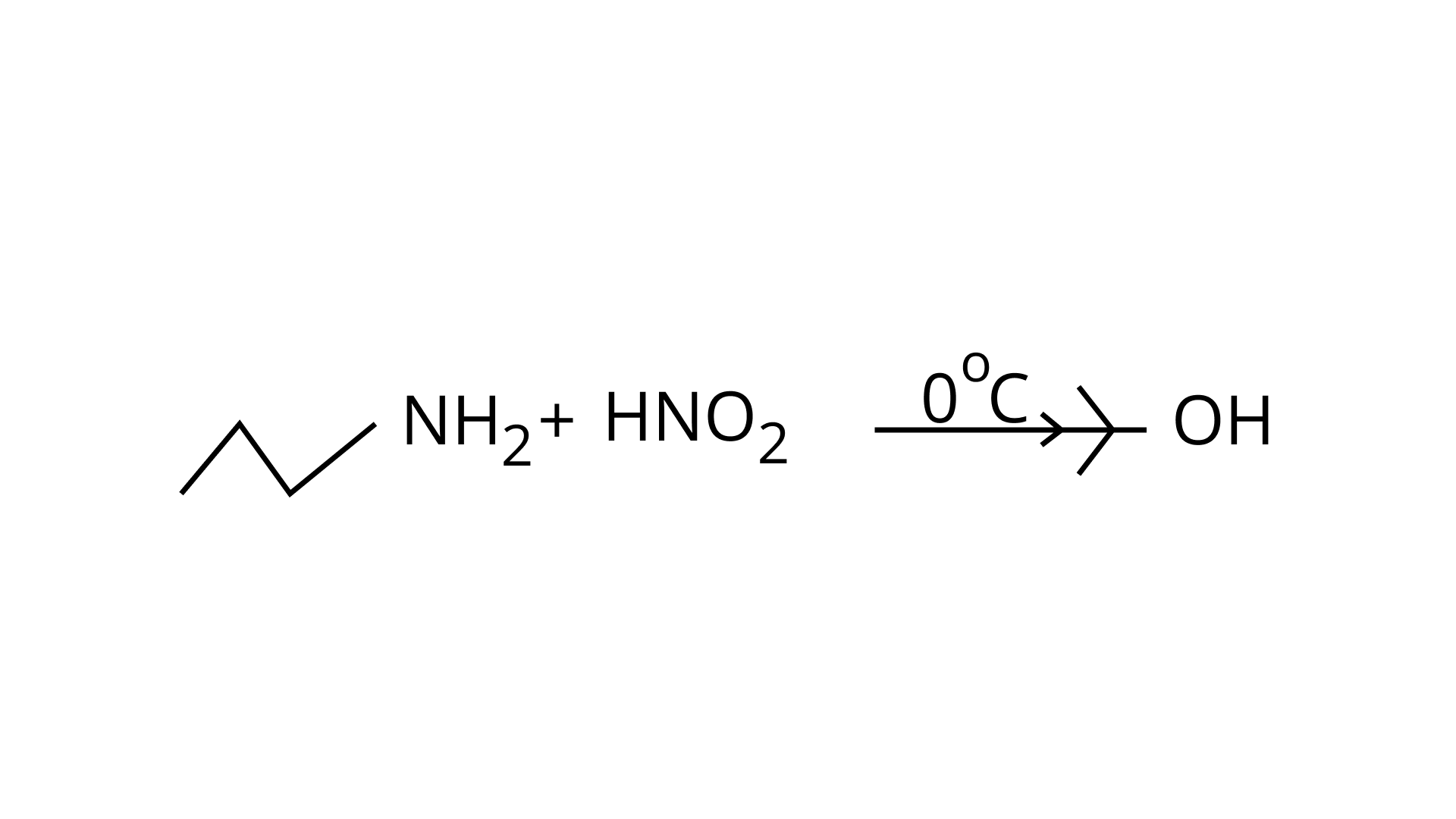
(d) In this reaction, aliphatic primary amines, on treatment with nitrous acid, produce primary alcohol as a product.
Therefore (D) is incorrect.
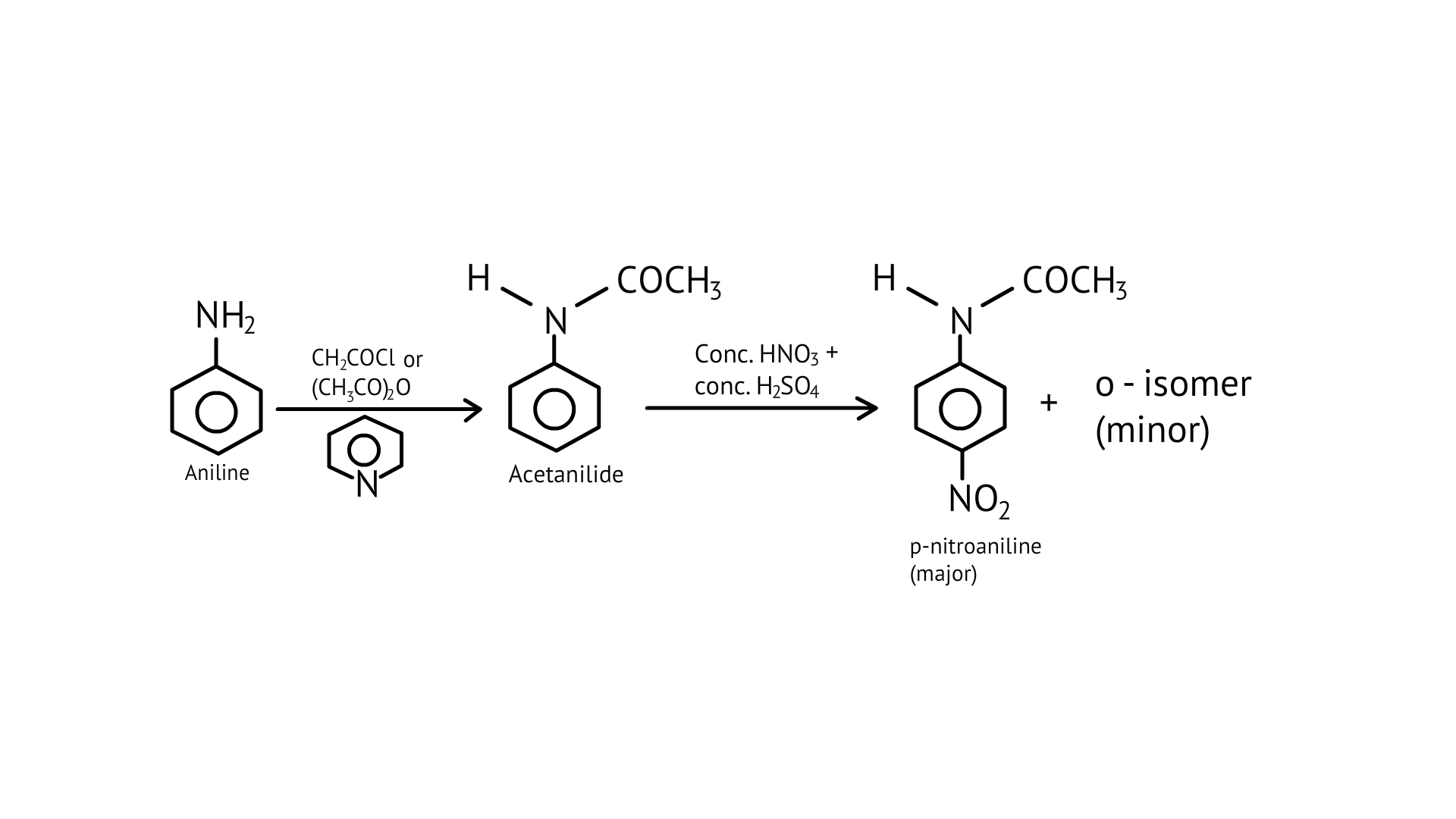
36. Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(A) Acetyl chloride/pyridine followed by reaction with conc. $\mathrm{H}_{2} \mathrm{SO}_{4}+$ conc. $\mathrm{HNO}_{3}$
(B) Acetic anhydride/pyridine followed by conc. $\mathrm{H}_{2} \mathrm{SO}_{4}+$ conc. $\mathrm{HNO}_{3}$
(C) Dil. $\mathrm{HCl}$ followed by reaction with conc. $\mathrm{H}_{2} \mathrm{SO}_{4}+$ conc. $\mathrm{HNO}_{3}$
(D) Reaction with conc. $\mathrm{H}_{2} \mathrm{SO}_{4}+$ conc. $\mathrm{HNO}_{3}$
Ans: (A and B)
In addition to the nitro derivatives, direct nitration of aniline produces tarry oxidation products. Furthermore, aniline is protonated in the strongly acidic medium to form the anilinium ion, which is meta directing. As a result, in addition to the ortho and para derivatives, a significant amount of meta derivative is formed.
However, by protecting the $-\mathrm{NH}_{2}$ group with an acetylation reaction with acetic anhydride, the nitration reaction can be controlled and the p-nitro derivative obtained as the main product.
37. Which of the following reactions belong to electrophilic aromatic substitution?
(A) Bromination of acetanilide
(B) Coupling reaction of aryldiazonium salts
(C) Diazotization of aniline
(D) Acylation of aniline
Ans: (A and B)
Benzene diazonium chloride reacts with phenol to form p-hydroxy azobenzene by coupling the phenol molecule at its para position with the diazonium salt. This is referred to as a coupling reaction. This is an illustration of an electrophilic substitution reaction.
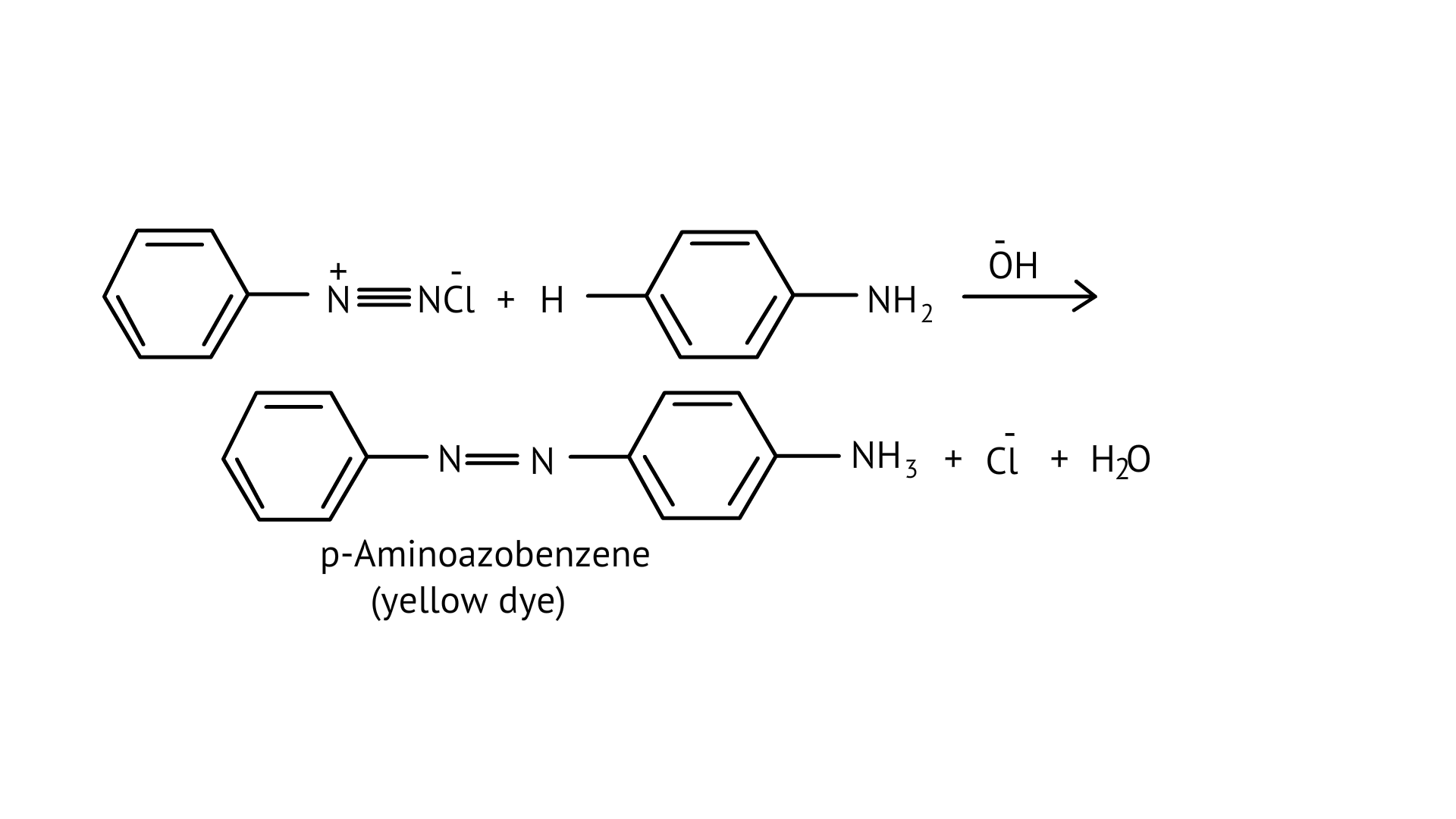
At room temperature, aniline reacts with bromine water to form a white precipitate of 2,4,6-tribromoaniline.
By acetylation with acetic anhydride to protect the $-\mathrm{NH}_{2}$ group, then carrying out the desired substitution followed by hydrolysis of the substituted amide to the substituted amine.
SHORT ANSWER TYPE:
38. Which is the role of $\mathrm{HNO}_{3}$ in the nitrating mixture used for nitration of benzene?
Ans:
In the nitrating mixture of conc. $\mathrm{H}_{2} \mathrm{SO}_{4}+$ conc. $\mathrm{HNO}_{3}, \mathrm{HNO}_{3}$ act as base to generate the nitronium ion which is an initiation species for the nitration reaction.
$\mathrm{HNO}_{3}+\mathrm{H}_{2} \mathrm{SO}_{4} \longrightarrow \underset{\text { Electrophile }}{\mathrm{NO}_{2}^{+}}+\underset{4}{\mathrm{HSO}_{4}^{-}}+\mathrm{H}_{2} \mathrm{O}$
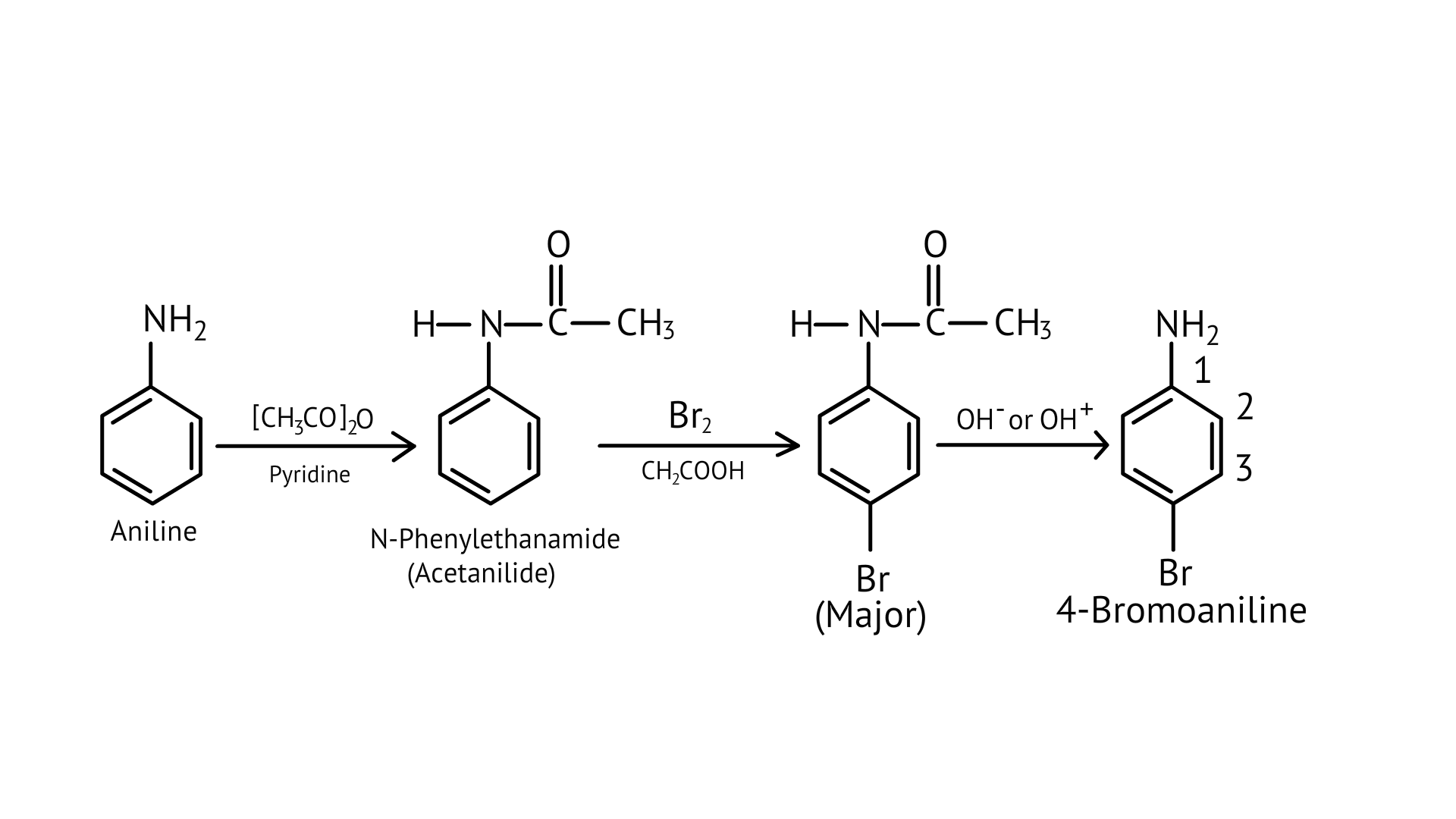
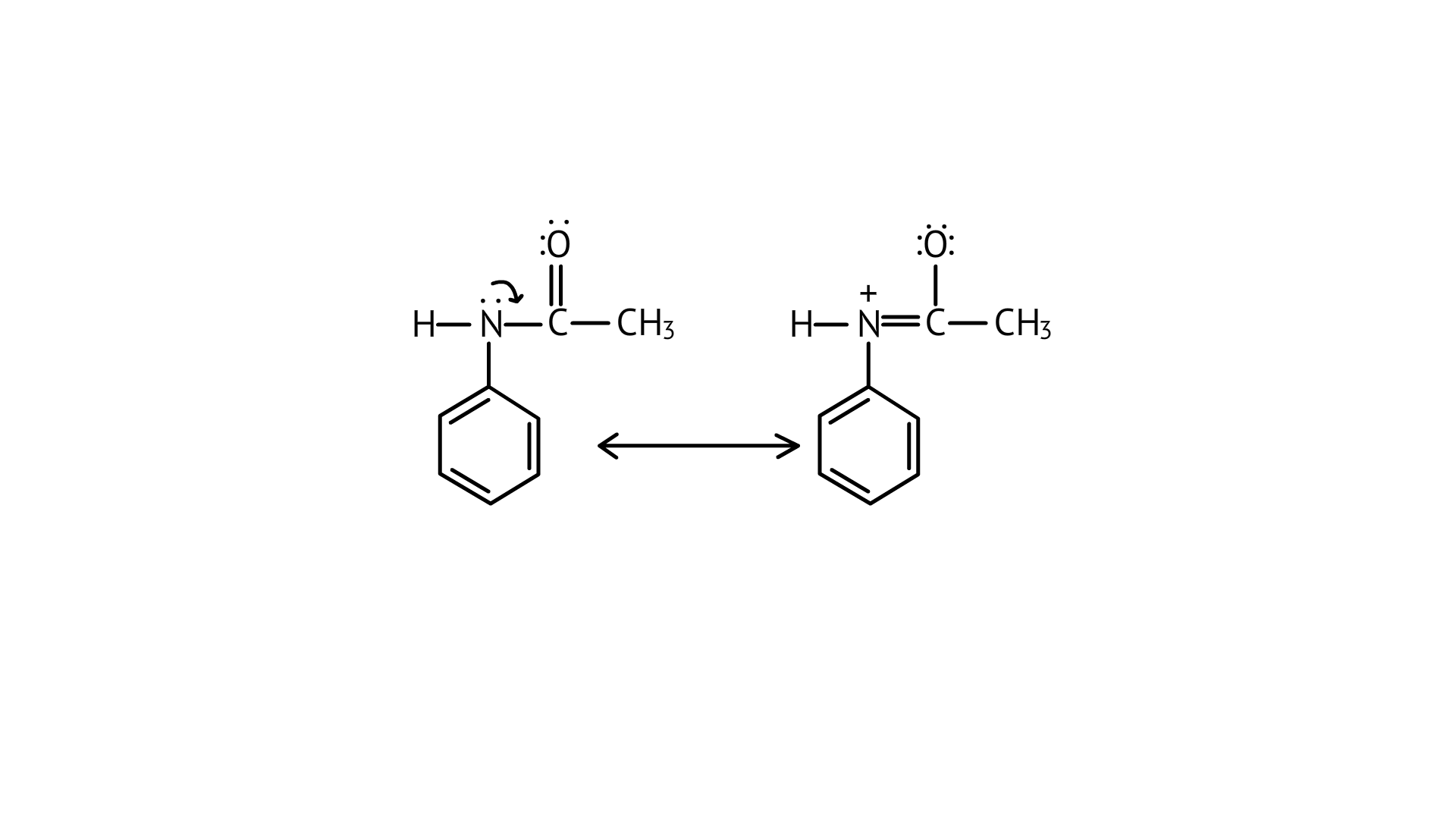
39. Why is $-\mathrm{NH}_{2}$ group of aniline acetylated before carrying out nitration?
Ans:
As $-\mathrm{NH}_{2}$ is a strong activating group, the aniline will readily undergo electrophilic substitution reaction, and it is difficult to cease reaction at the mono substitution stage. So, direct acetylation of aniline is not possible as it undergoes oxidation.
Therefore, the activating group $-\mathrm{NH}_{2}$ is protected by an acetylation process and then followed by the nitration reaction to obtain a monosubstituted nitrated product.
The acetylated complex formed utilises the lone pair of nitrogen and is less available for donation, this helps to carry out the nitration reaction easily.
40. What is the product when $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{NH}_{2}$ reacts with $\mathrm{HNO}_{2}$ ?
Ans: When Benzyl amine is treated with nitrous acid, firstly BDC is formed which is unstable and decomposes to Benzyl alcohol.
$\underset{\text { Benzyl amine }}{\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{NH}_{2}}+\mathrm{HONO} \stackrel{\mathrm{HCl}}{\longrightarrow} \underset{\text { Benzyl alcohol }}{\longrightarrow} \underset{6}{\mathrm{H}_{5} \mathrm{CH}_{2} \mathrm{OH}}+\mathrm{N}_{2}+\mathrm{H}_{2} \mathrm{O}$
41. What is the best reagent to convert nitrile to primary amine?
Ans: On treatment of nitriles with strong reducing agents such as lithium aluminium hydride in ether or sodium metal in alcohol, primary amines are obtained.
$\underset{\text { Alkyl nitrile }}{R-\cdots} \mathrm{CN}+4[\mathrm{H}] \frac{\mathrm{Na} / \mathrm{C}_{2} \mathrm{H}_{5} \mathrm{OH}}{\text { or } \mathrm{Li} \mathrm{Ah}_{4} / \mathrm{ether}} \longrightarrow R-\mathrm{Cl}_{2} \mathrm{NH}_{2}$
42. Give the structure of ‘A’ in the following reaction.
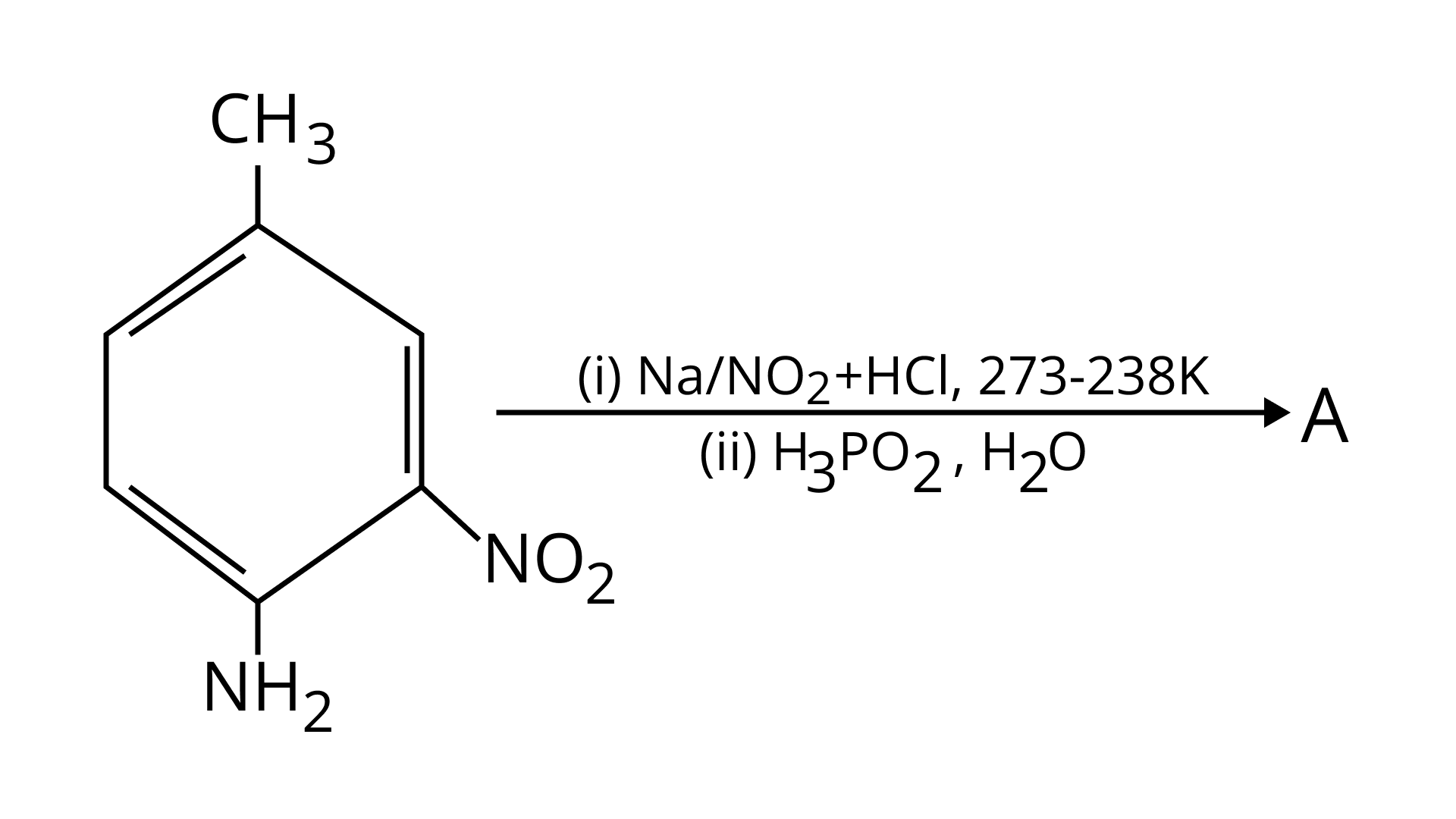
Ans:
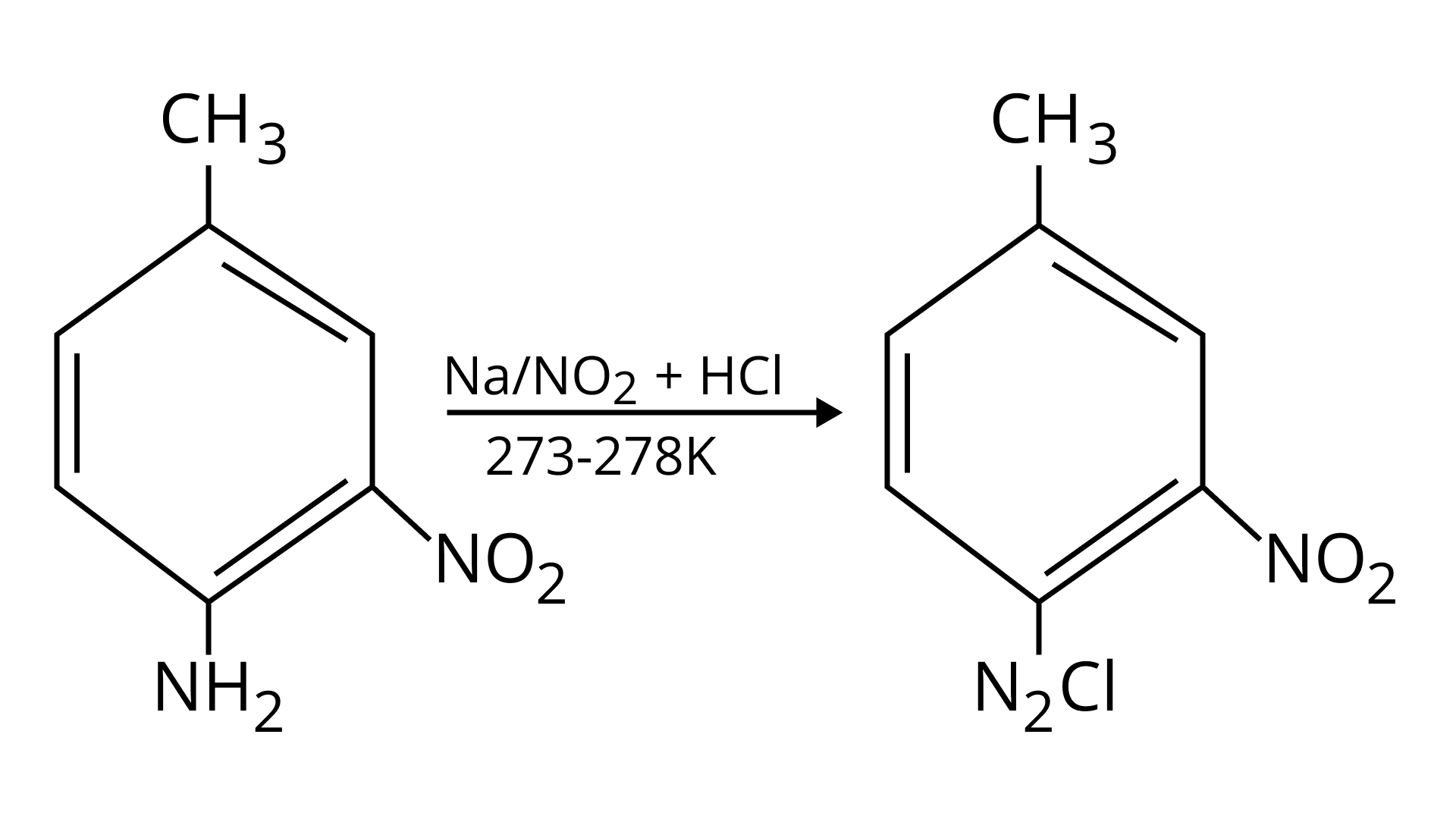
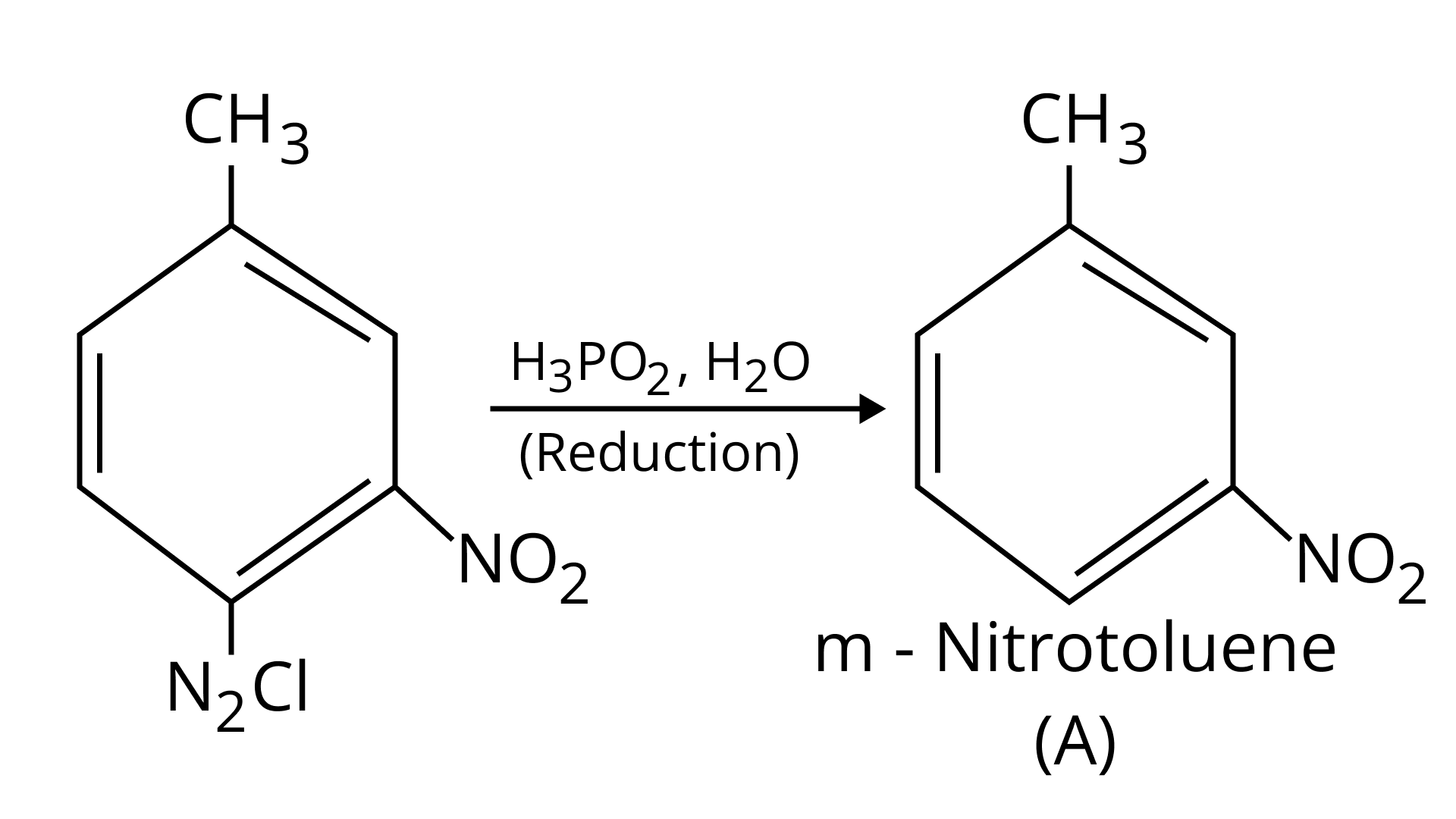
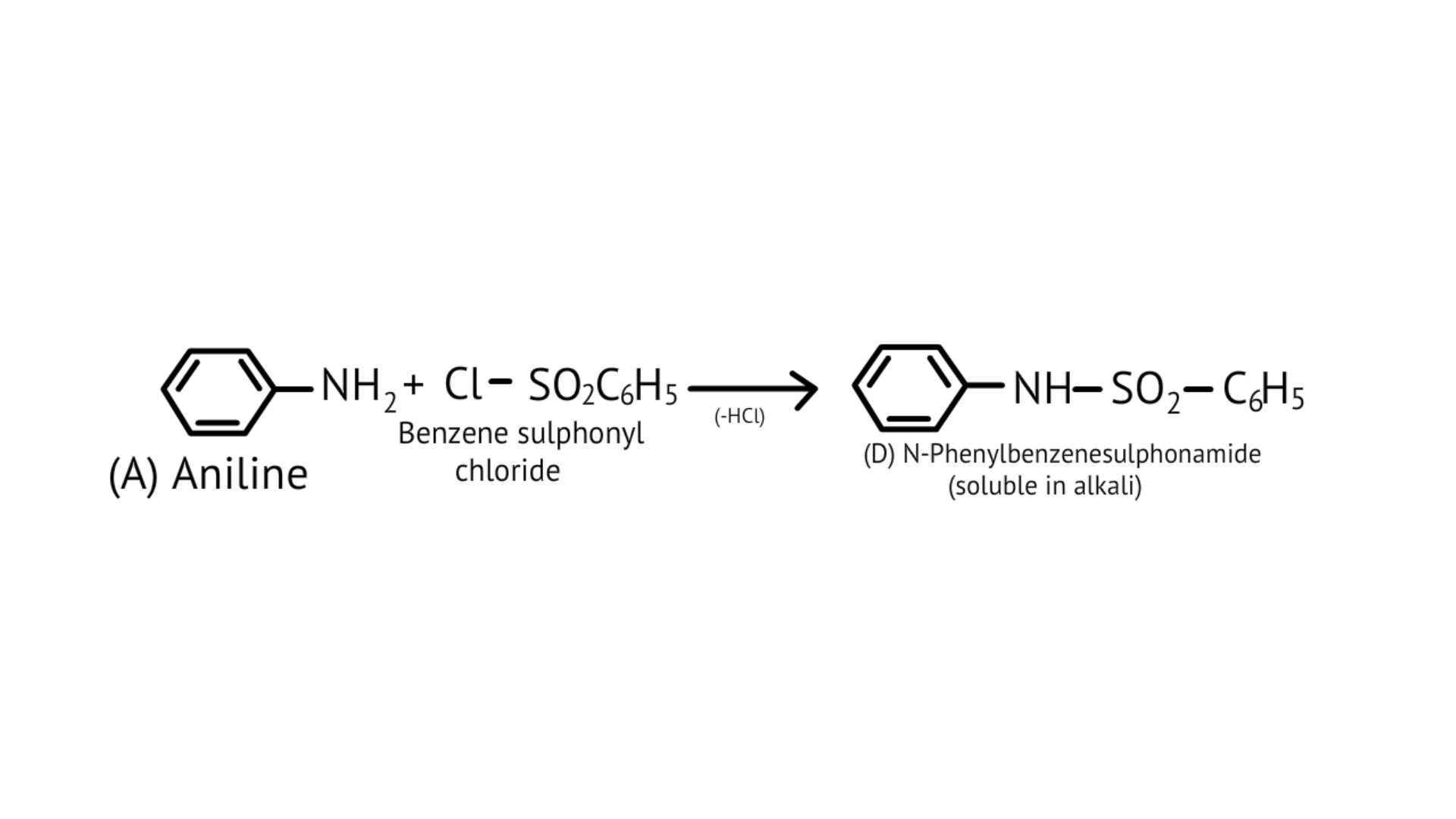
43. What is Hinsberg reagent?
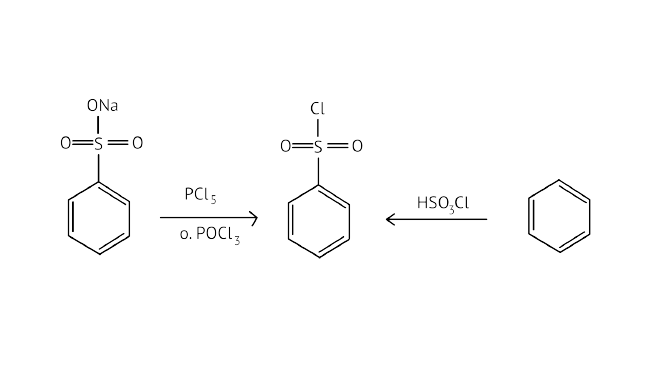
Ans: Hinsberg's reagent is also known as benzenesulphonyl chloride ($\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{SO}_{2} \mathrm{Cl}$). When it reacts with primary and secondary amines, it produces sulphonamides. Allowing secondary and tertiary amines to react with Hinsberg's reagent allows them to be distinguished (benzenesulphonyl chloride $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{SO}_{2} \mathrm{Cl}$). Secondary amines react with Hinsberg's reagent to form an alkali-insoluble product. N, N-diethylamine, for example, reacts with Hinsberg's reagent to form N,N-diethylbenzenesulphonamide, which is insoluble in alkalis. Tertiary amines, on the other hand, are unaffected by Hinsberg's reagent.
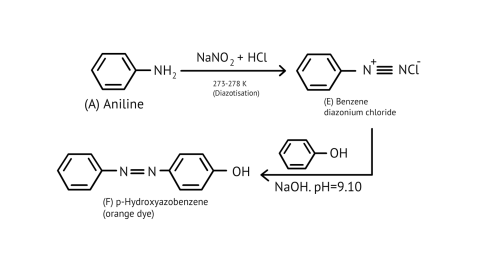
44. Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Ans: The reaction of aniline with nitrous acid at 273-278 K produces benzene diazonium chloride. The reaction of sodium nitrite with hydrochloric acid produces nitrous acid in the reaction mixture. Diazotisation is the process of converting primary aromatic amines into diazonium salts. Because of its instability, the diazonium salt is generally not stored or used immediately after preparation.
The crystalline solid benzene diazonium chloride is colorless.
It is easily soluble in water and stable at room temperature, but it reacts with water when warmed.
In the dry state, it decomposes easily.
45. Why does acetylation of $-\mathrm{NH}_{2}$ group of aniline reduce its activating effect?
Ans: As $-\mathrm{NH}_{2}$ is a strong activating group, the aniline will readily undergo electrophilic substitution reaction, and it is difficult to cease reaction at the mono substitution stage.
Therefore, the activating group $-\mathrm{NH}_{2}$ is protected by an acetylation process.
The acetylated complex formed utilizes the lone pair of nitrogen and are less available for donation, this helps to carry out the nitration reaction easily.
46. Explain why is $\mathrm{MeNH}_{2}$ stronger base than $\mathrm{MeOH}$?
Ans: As the electronegativity of oxygen is more than the electronegativity of a nitrogen atom, the $\mathrm{O}-\mathrm{H}$ bond is more polar than the $\mathrm{N}-\mathrm{H}$ bond, therefore $\mathrm{MeOH}$ is stronger acid than $\mathrm{MeNH}_{2}$ or $\mathrm{MeNH}_{2} 2$ is stronger base than $\mathrm{MeOH}$.
47. What is the role of pyridine in the acylation reaction of amines?
Ans: Amides are the byproducts of the acylation reaction. The reaction is carried out in the presence of a stronger base than the amine, such as pyridine, which removes the formed HCl and shifts the equilibrium to the right.
48. Under what reaction conditions (acidic/basic), the coupling reaction of aryl diazonium chloride with aniline is carried out?
Ans: The azo products have an extended conjugate system with both aromatic rings linked by the –N=N- bond. These compounds are frequently colored and used as dyes. Benzene diazonium chloride reacts with phenol to form p-hydroxy azobenzene by coupling the phenol molecule in its para position with the diazonium salt. This is referred to as a coupling reaction.
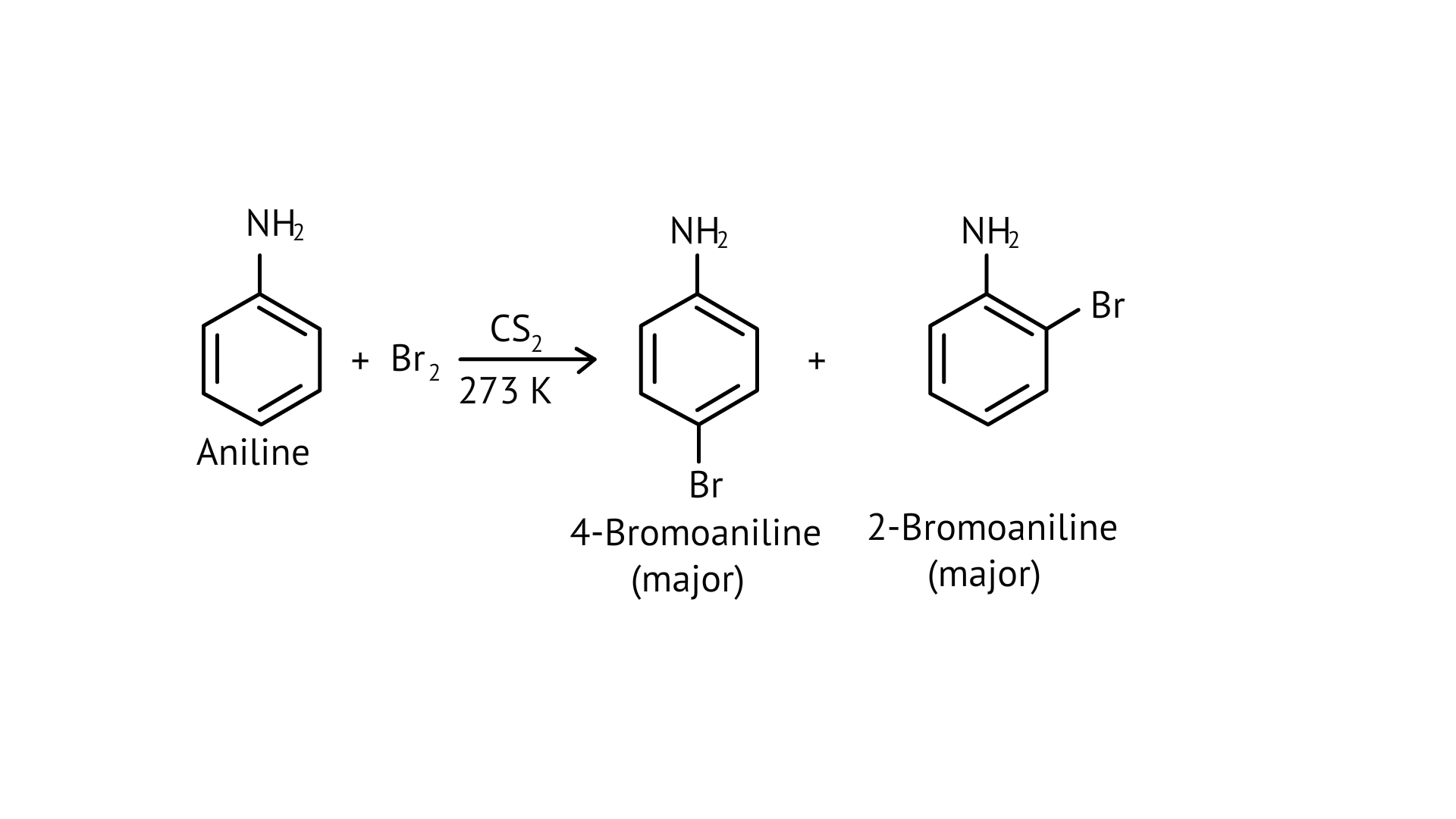
49. Predict the product of reaction of aniline with bromine in non-polar solvent such as $\mathrm{CS}_{2}$.
Ans: $\mathrm{CS}_{2}$ is a non-polar solvent which decreases the activating effect of $-\mathrm{NH}_{2}$. As a result, mono substitution occurs only at o- and p- positions giving a mixture of 2-bromoaniline (minor) and 4-bromoaniline (major) as products.
50. Arrange the following compounds in increasing order of dipole moment:
$\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{3}, \mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{NH}_{2}, \mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{OH}$.
Ans: $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{3}<\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{NH}_{2}<\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{OH}$
As oxygen is more electronegative than nitrogen therefore, the O-H bond is more polar than the N-H bond. So ethanol has more dipole moments than ethylamine. Propane is non- polar in nature hence, had the least among all.
51. What is the structure and IUPAC name of the compound, allyl amine?
Ans: Prop-2-en-1-amine
$\mathrm{CH}_{2}=\mathrm{CH}-\mathrm{CH}_{2} \mathrm{NH}_{2}$
Question 52. Write down the IUPAC name of
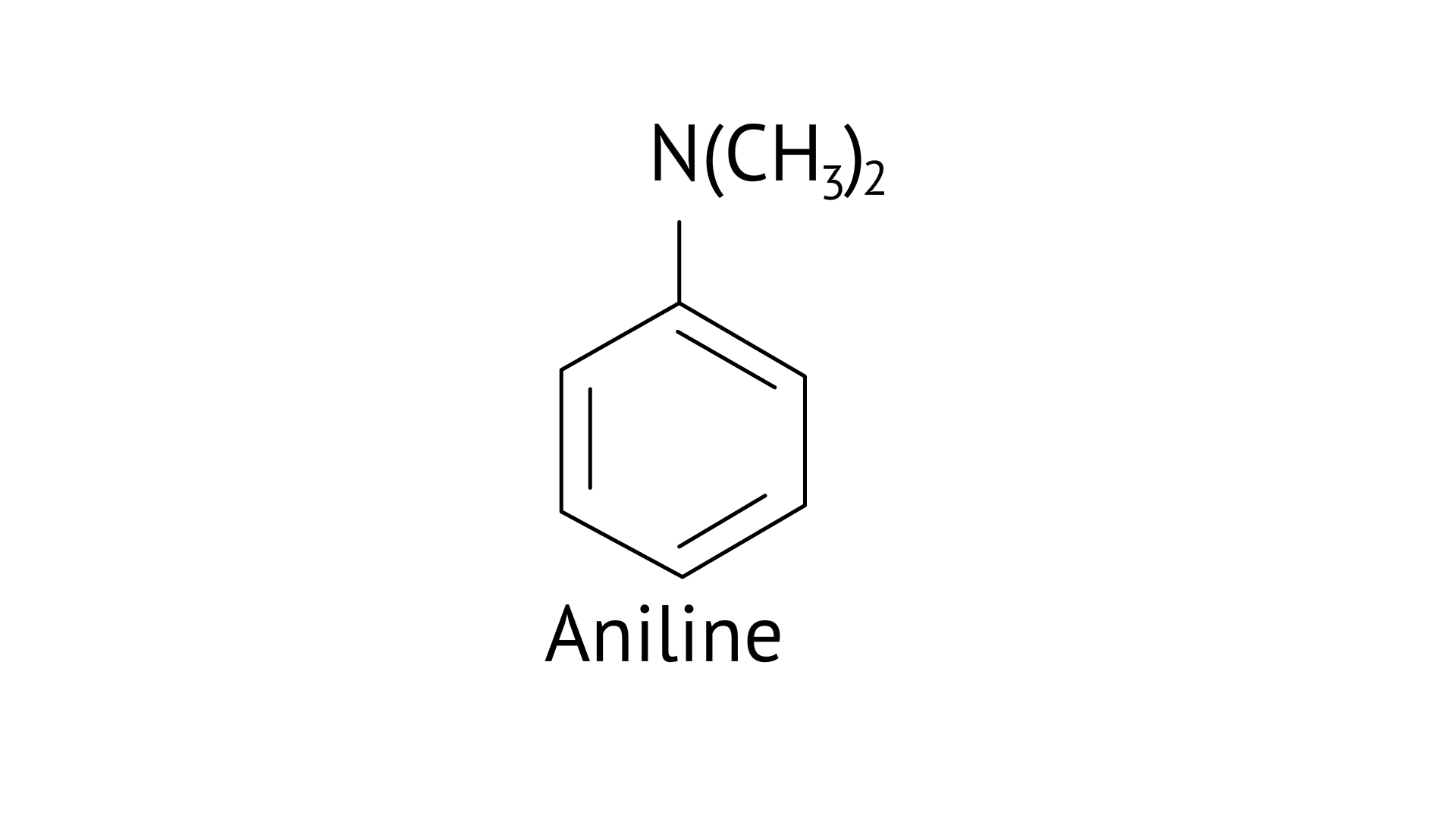
Ans: N, N-dimethylbenzenanmine
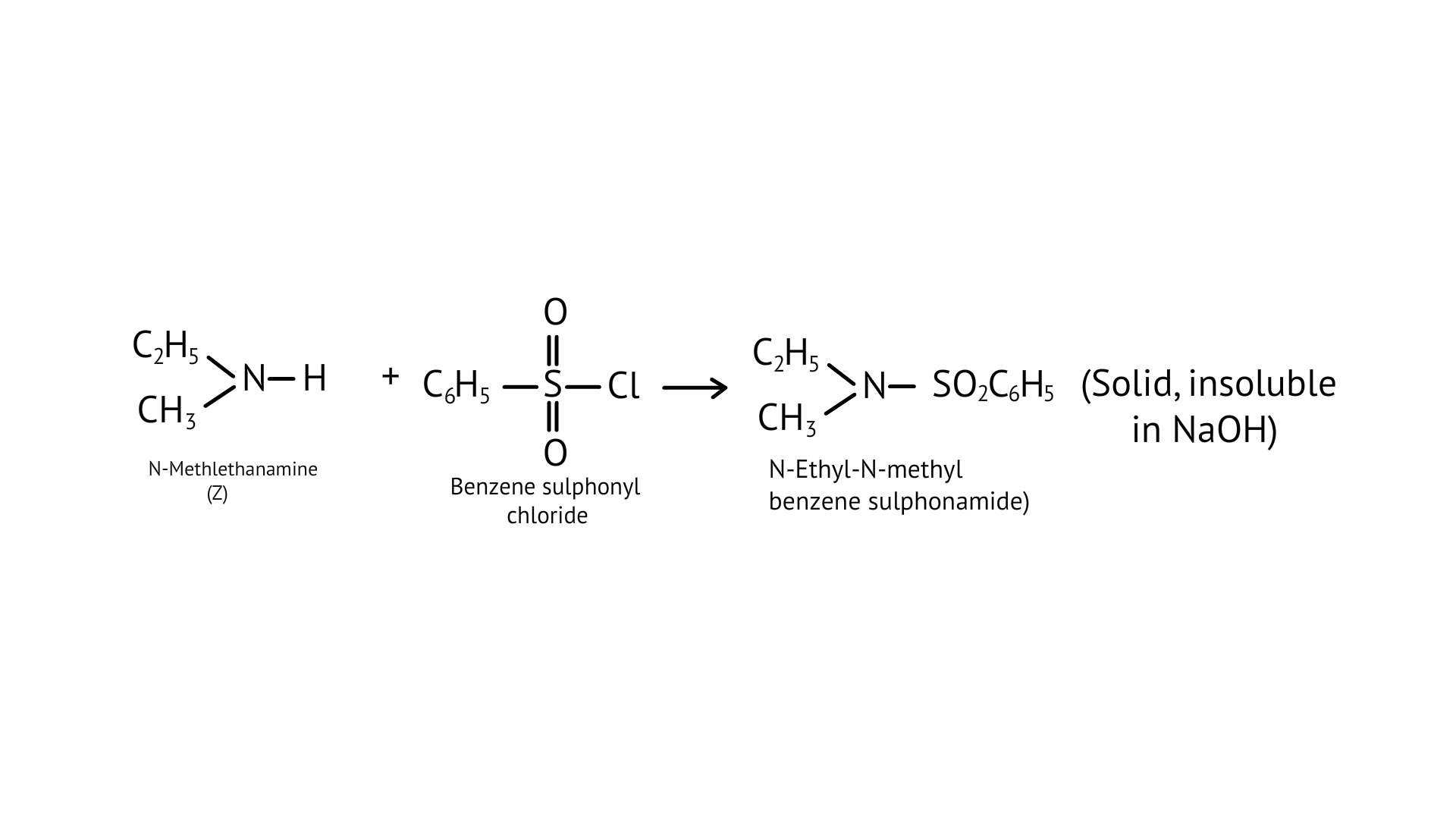
53. A compound $Z$ with molecular formula $C_{3} H_{9} N$ reacts with $C_{6} H_{5} S O_{2} C l$ to give a solid, insoluble in alkali, identify Z.
Ans: $Z$ is an aliphatic amine that produces a solid that is insoluble in base. This means that the reaction with $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{SO}_{2} \mathrm{Cl}$ must produce a product with no replaceable hydrogen attached to nitrogen.
To put it another way, the amine must be a secondary amine.
Z stands for ethylmethylamine.
54. A primary amine, $\mathrm{RNH}_{2}$ can be reacted with $\mathrm{CH}_{3}-\mathrm{X}$ to get secondary amine, $\mathrm{R}-\mathrm{NHCH}_{3}$ but the only disadvantage is that $3^{\circ}$ amine and quaternary ammonium salts are also obtained as side products. Can you suggest a method where $\mathrm{RNH}_{2}$ forms only $2^{\circ}$ amine?
Ans: Carbylamine reaction is shown by primary amines only, primary amines react with chloroform in the presence of alcoholic KOH to form isocyanides which upon catalytic reduction gives secondary amines as the final product.
$\underset{1^{\circ} \mathrm{Amine}}{R-\mathrm{NH}_{2}}+\mathrm{CHCl}_{3}+3 \mathrm{KOH} \rightarrow \underset{\text { Alkyl isocyanide }}{\longrightarrow} \frac{\mathrm{HC}_{2} / \mathrm{Pd}}{(\text { Reduction) })} \underset{2^{\circ} \text { Amine }}{\mathrm{N}}-\mathrm{NH}-\mathrm{CH}_{3}$
55. Complete the following reaction.
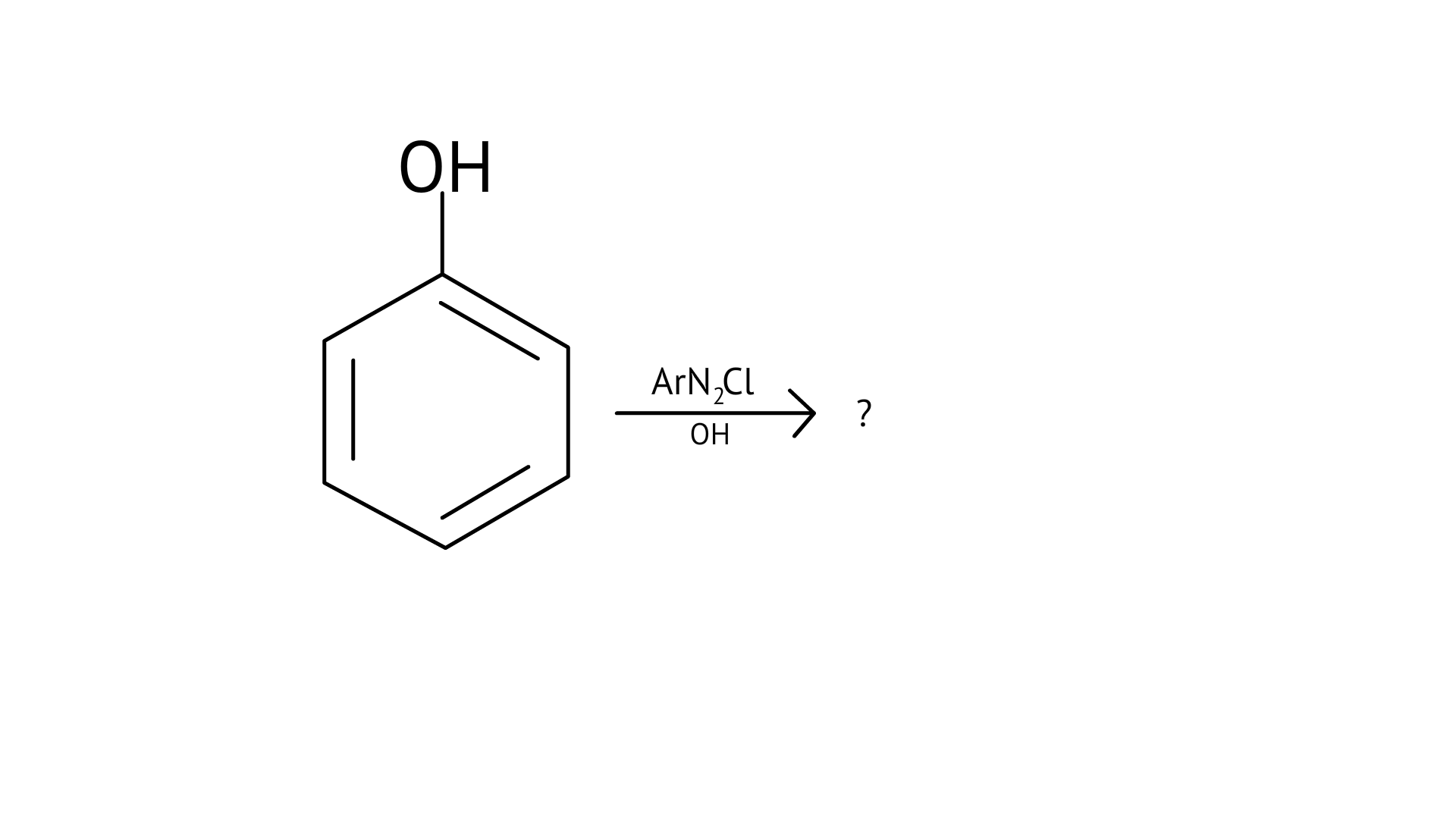
Ans:
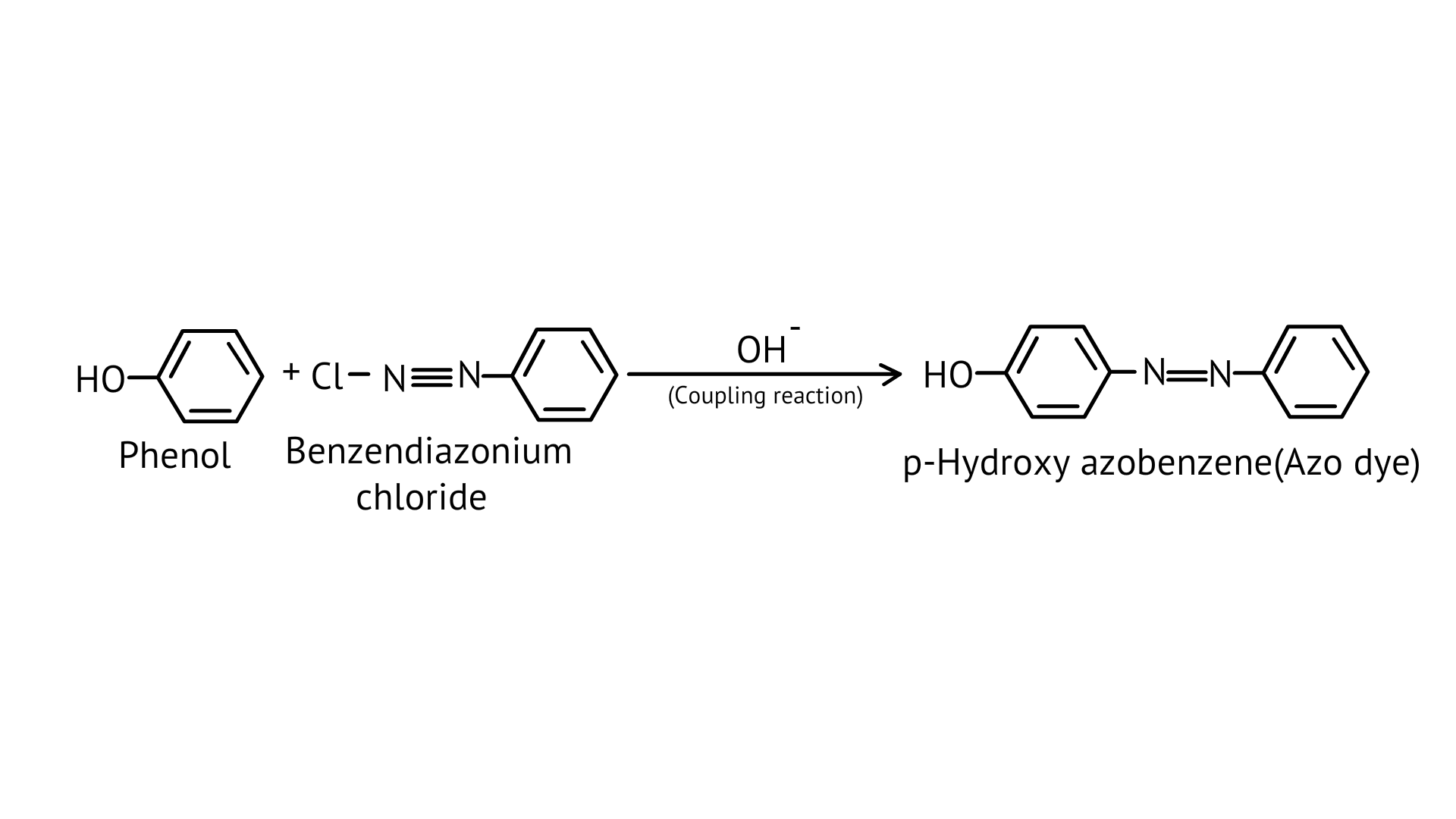
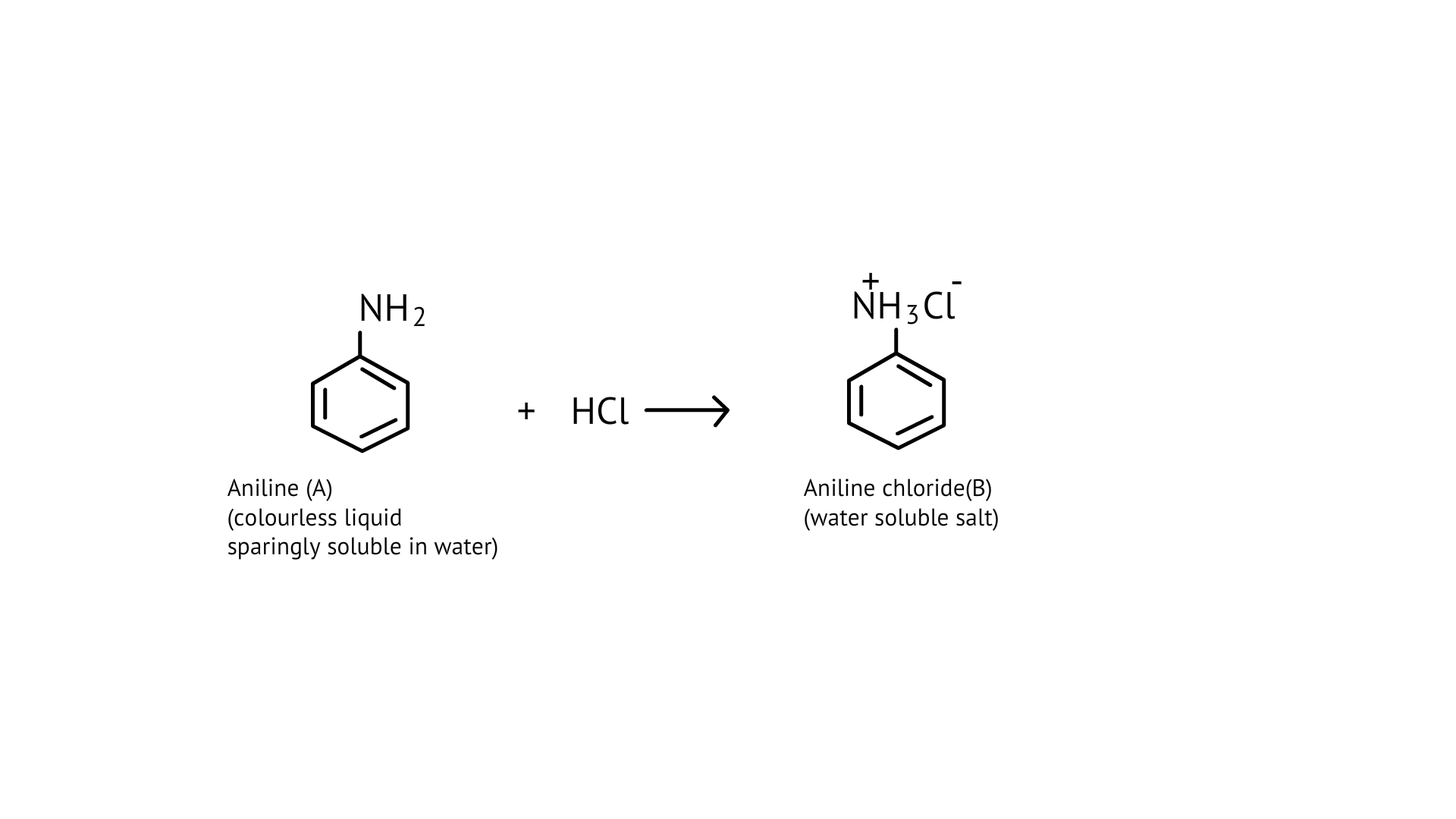
56. Why is aniline soluble in aqueous HCl?
Ans: On reaction with hydrochloric acid, aniline forms anilinium chloride ion which is water soluble. Therefore, the aniline solubility in aqueous HCl solution.
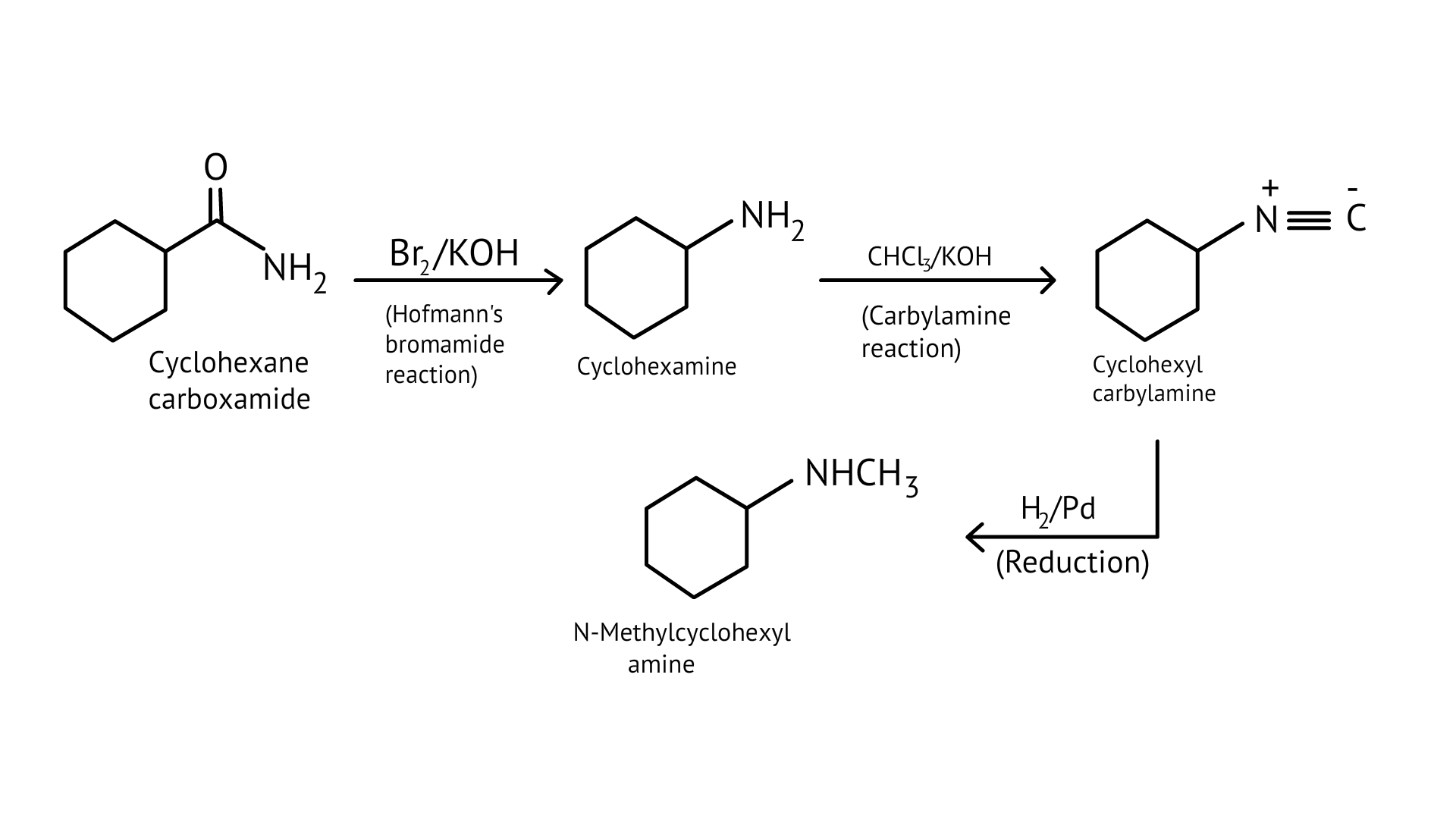
57. Suggest a route by which the following conversion can be accomplished.
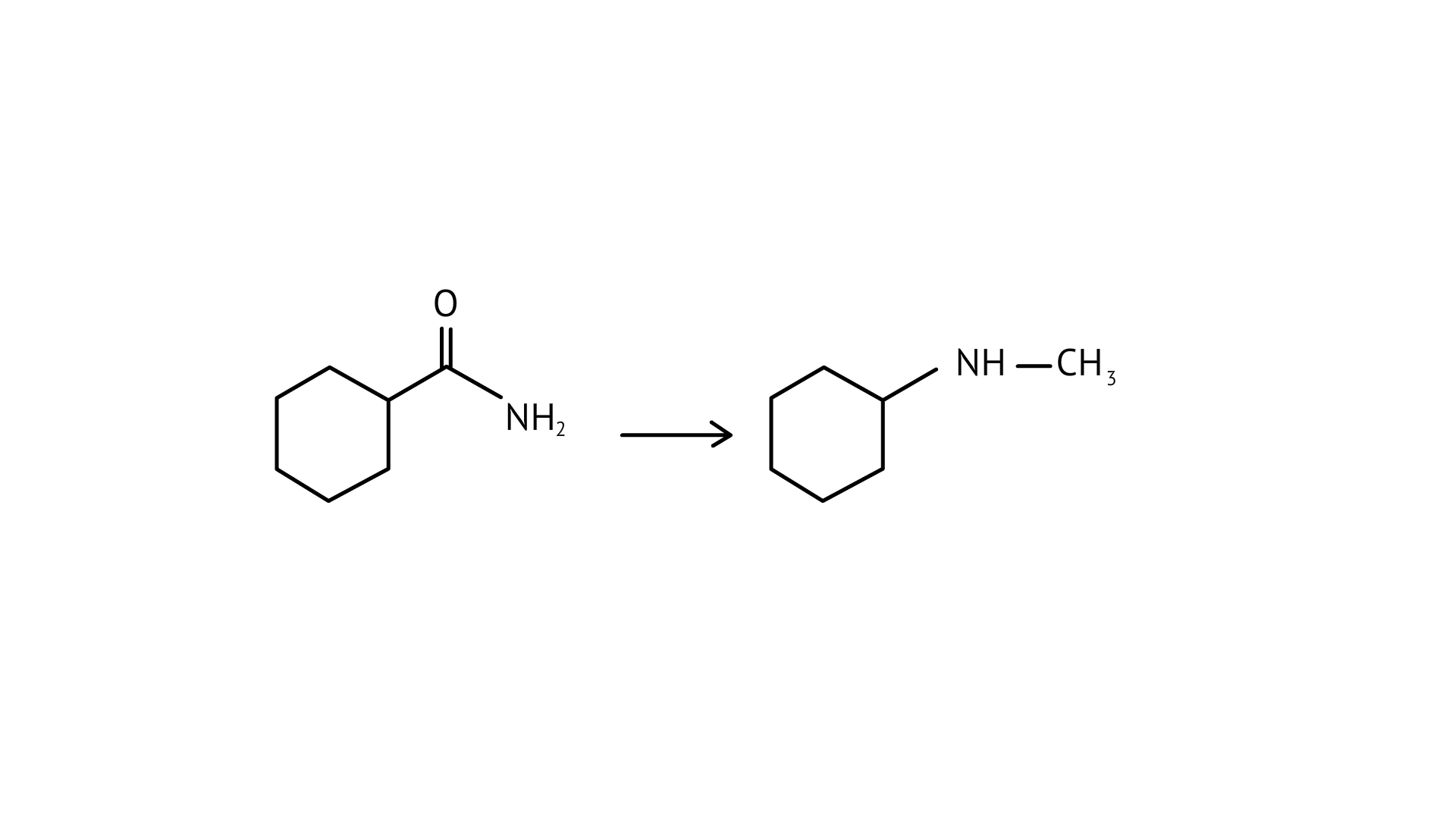
Ans:
58. Identify A and B in the following reaction.
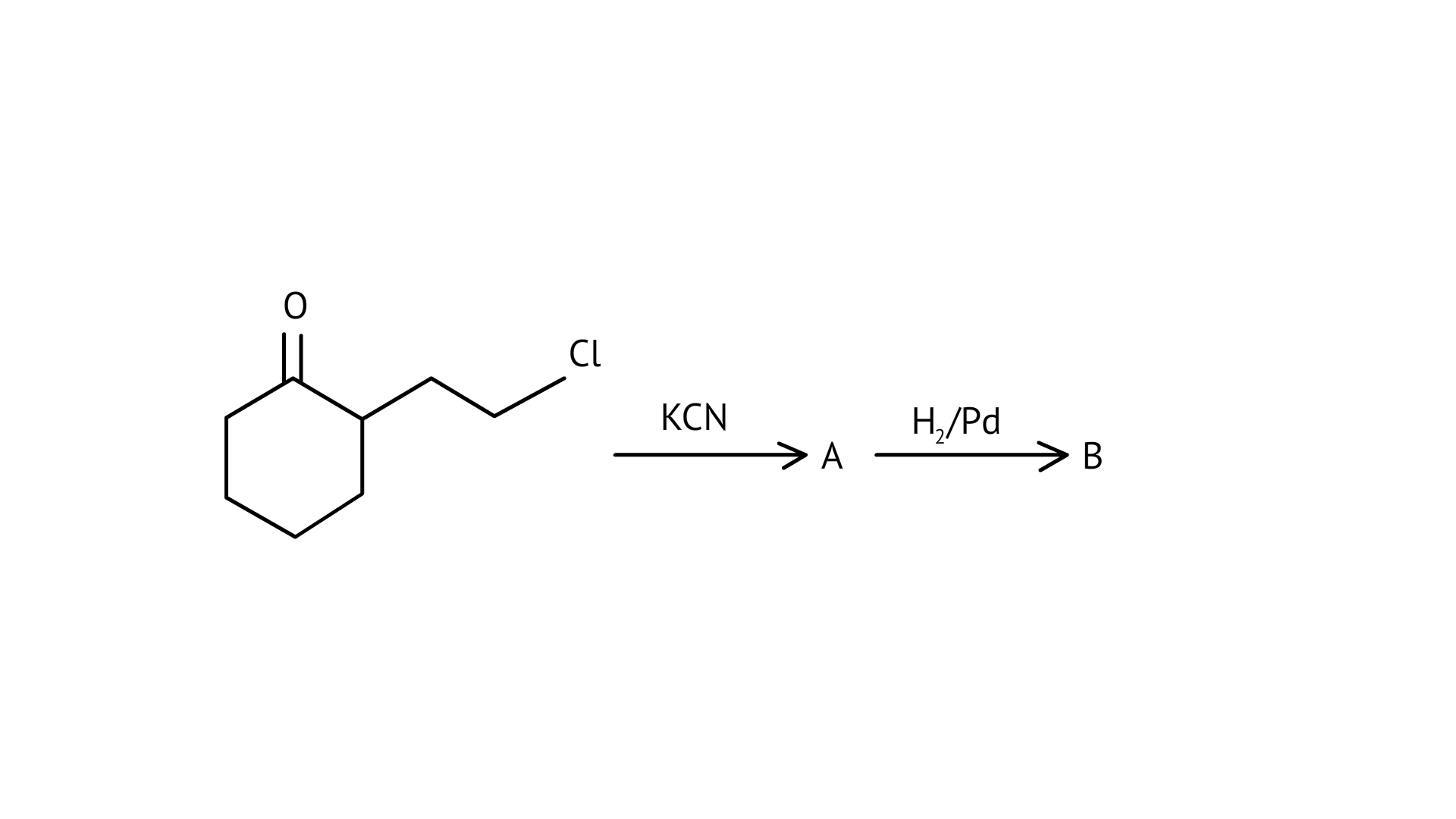
Ans:
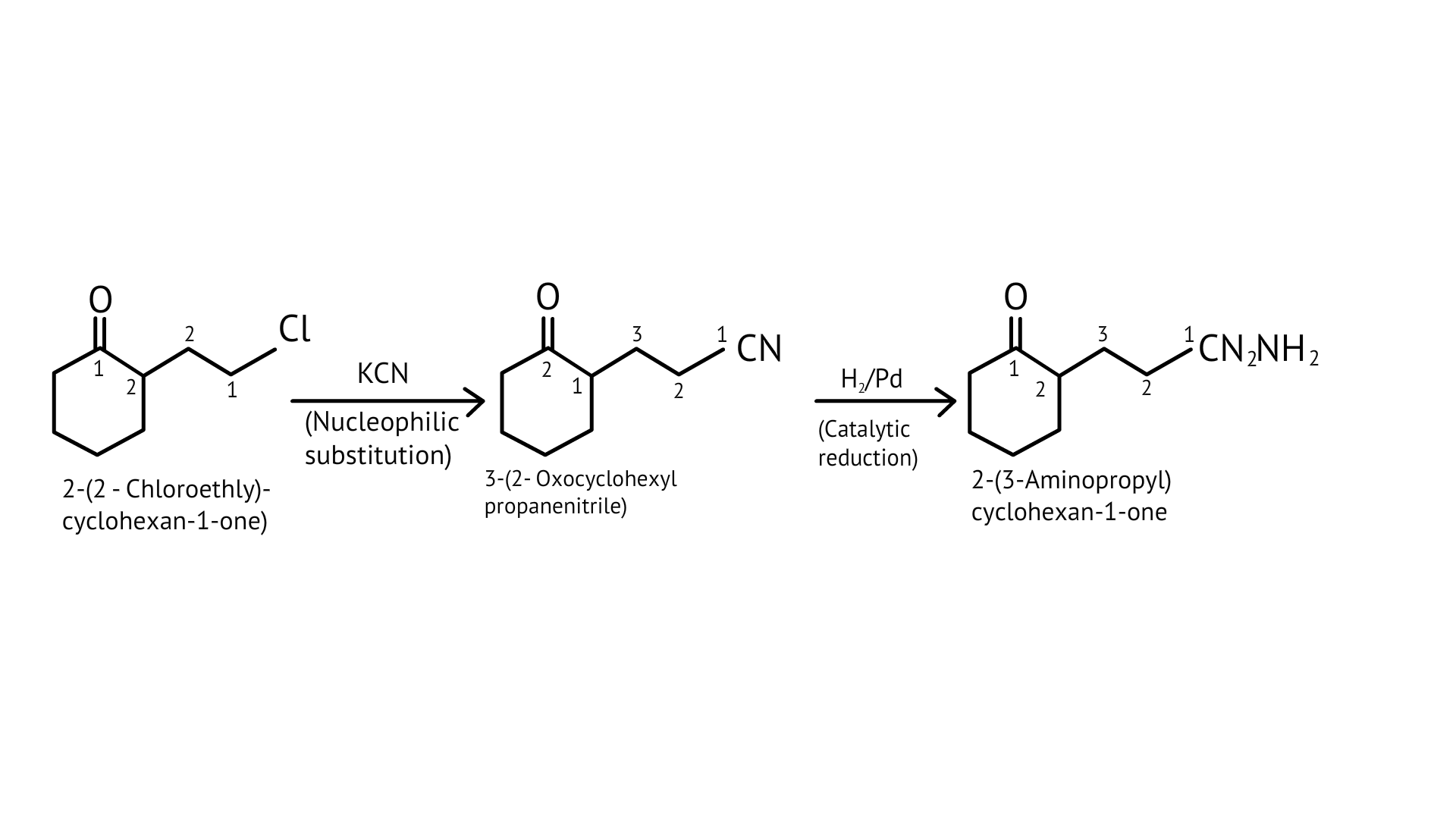
59. How will you carry out the following conversions?
(i) toluene $\rightarrow$ p-toluidine
Ans:
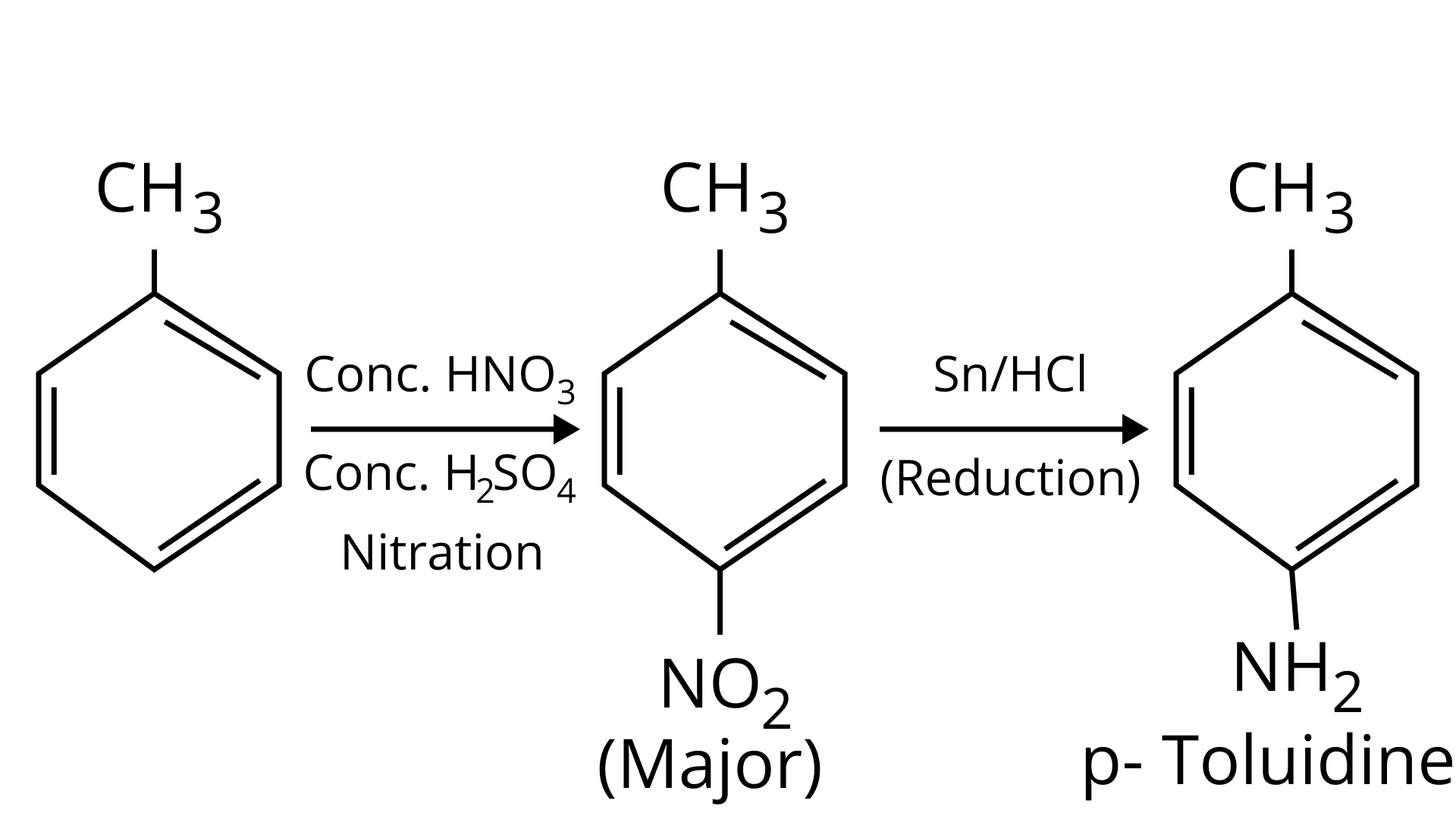
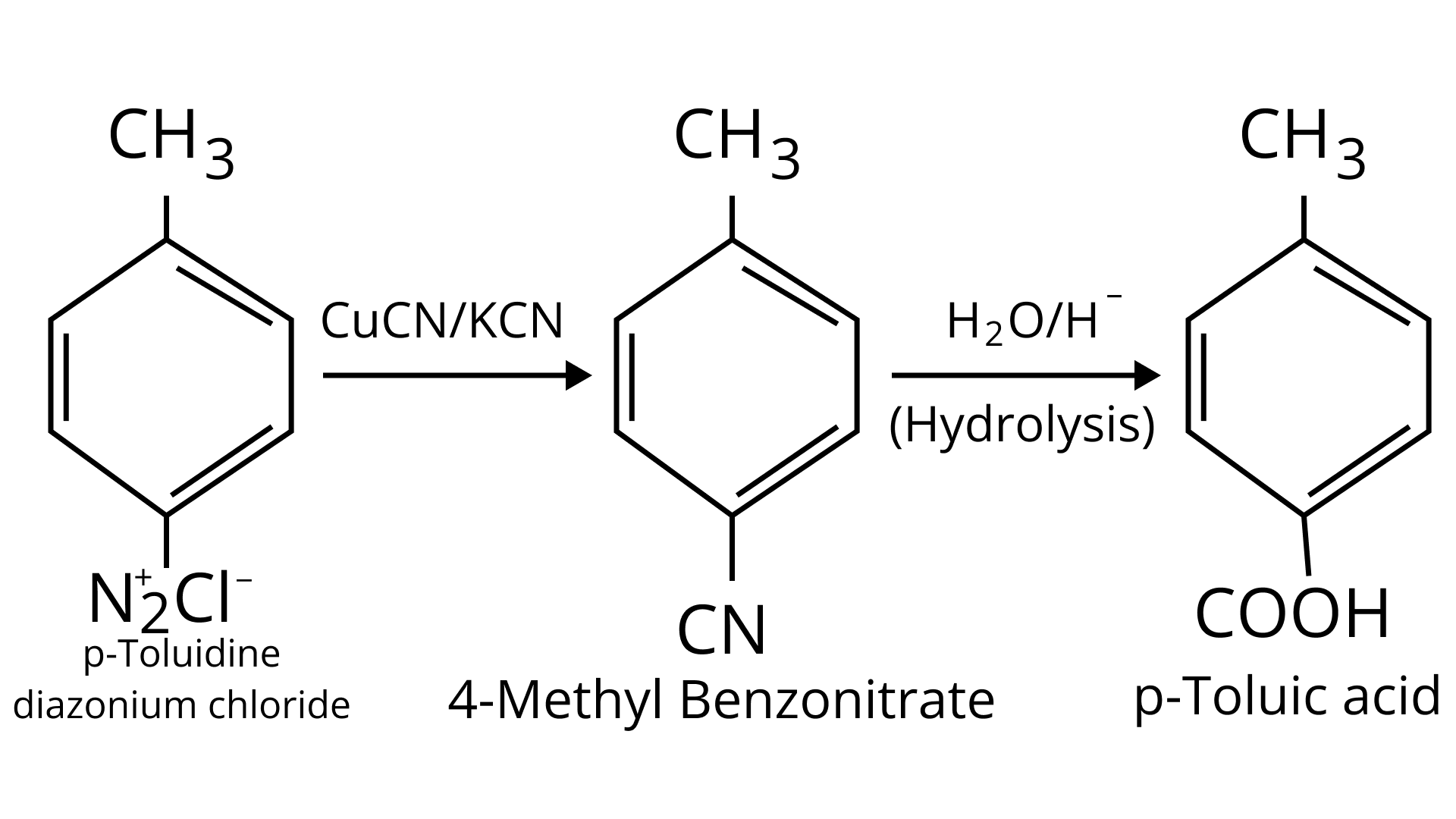
(ii) p-toluidine diazonium chloride $\rightarrow$ p-toluic acid
Ans:
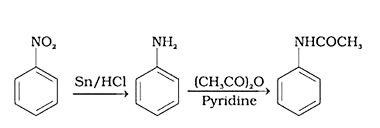
60. Write following conversions:
(i) nitrobenzene $\rightarrow$ acetanilide
Ans:
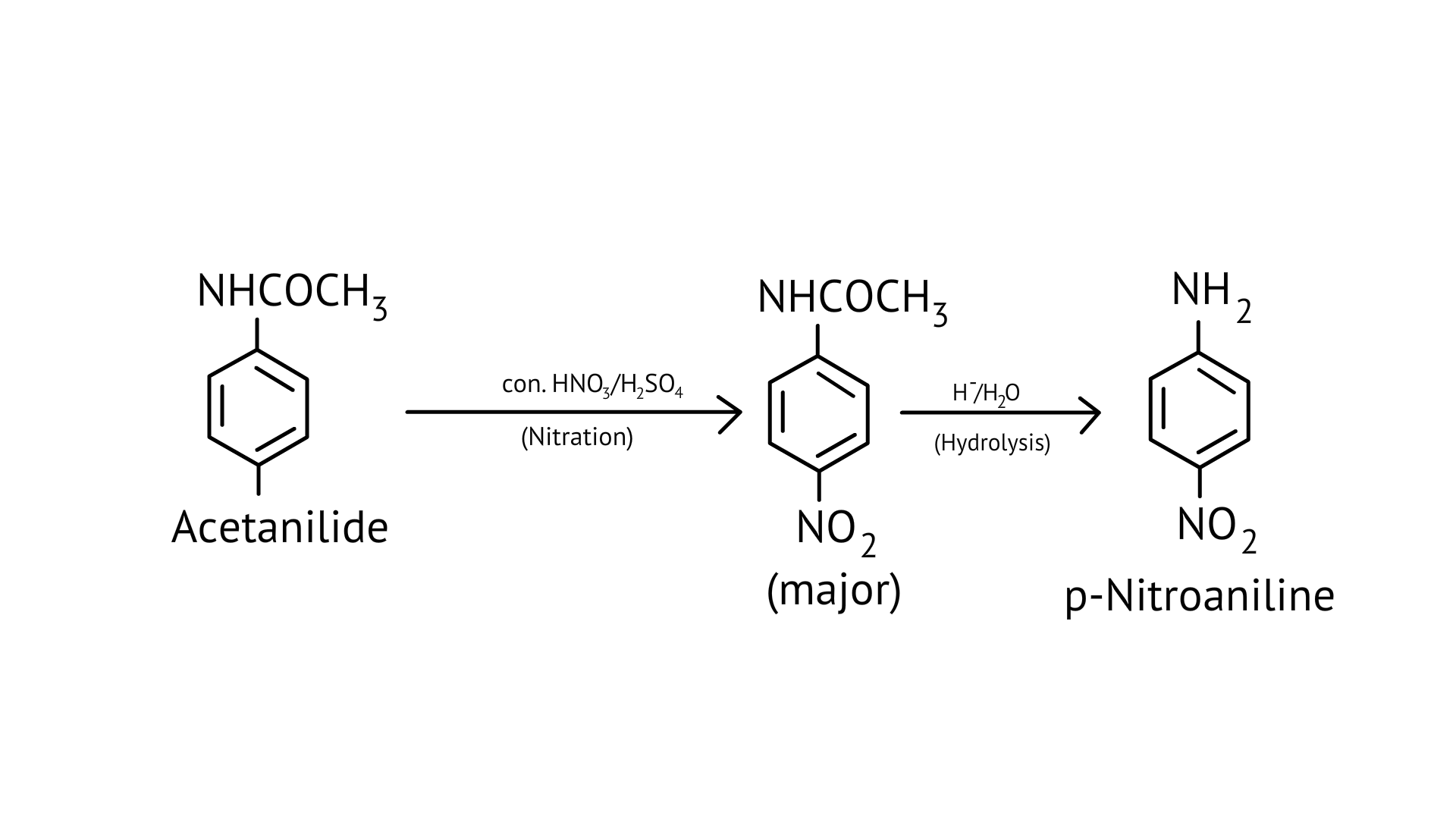
(ii) acetanilide $\rightarrow$ p-nitroaniline
Ans:
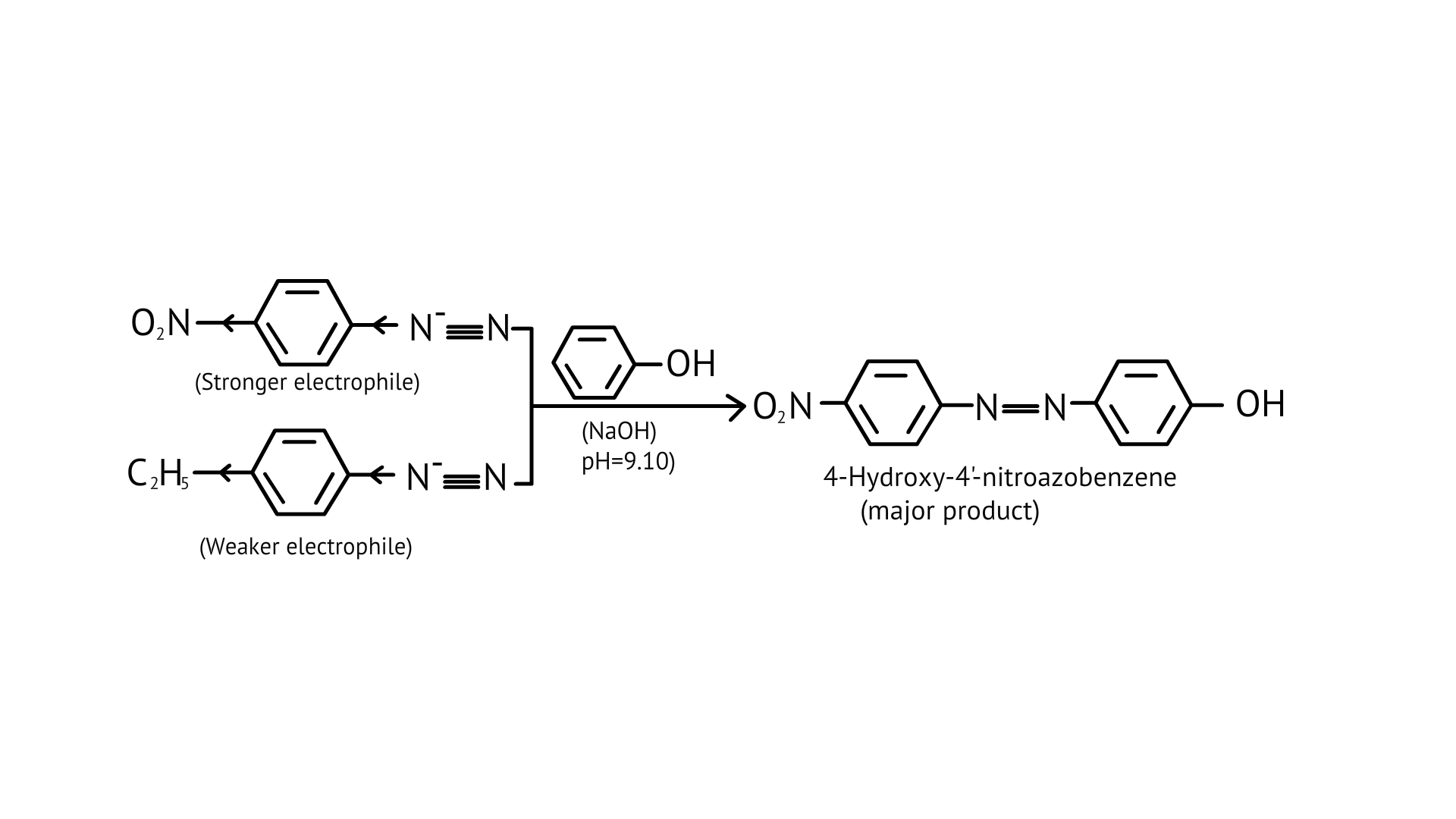
61. A solution contains 1 g mol each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this 1 g mol of alkaline solution of phenol is added. Predict the major product. Explain your answer.
Ans:
This is an example of an electrophilic aromatic substitution reaction. phenol generates phenoxide ion in alkaline medium, which is more electron-rich than phenol and thus more reactive to electrophilic attack. In this reaction, the electrophile is an aryldiazonium cation.
The faster the reaction, the stronger the electrophile.
The cation p-nitrophenyldiazonium is a stronger electrophile than the cation p-toluene diazonium.
As a result, it preferentially couples with phenol.
62. How will you bring out the following conversion?
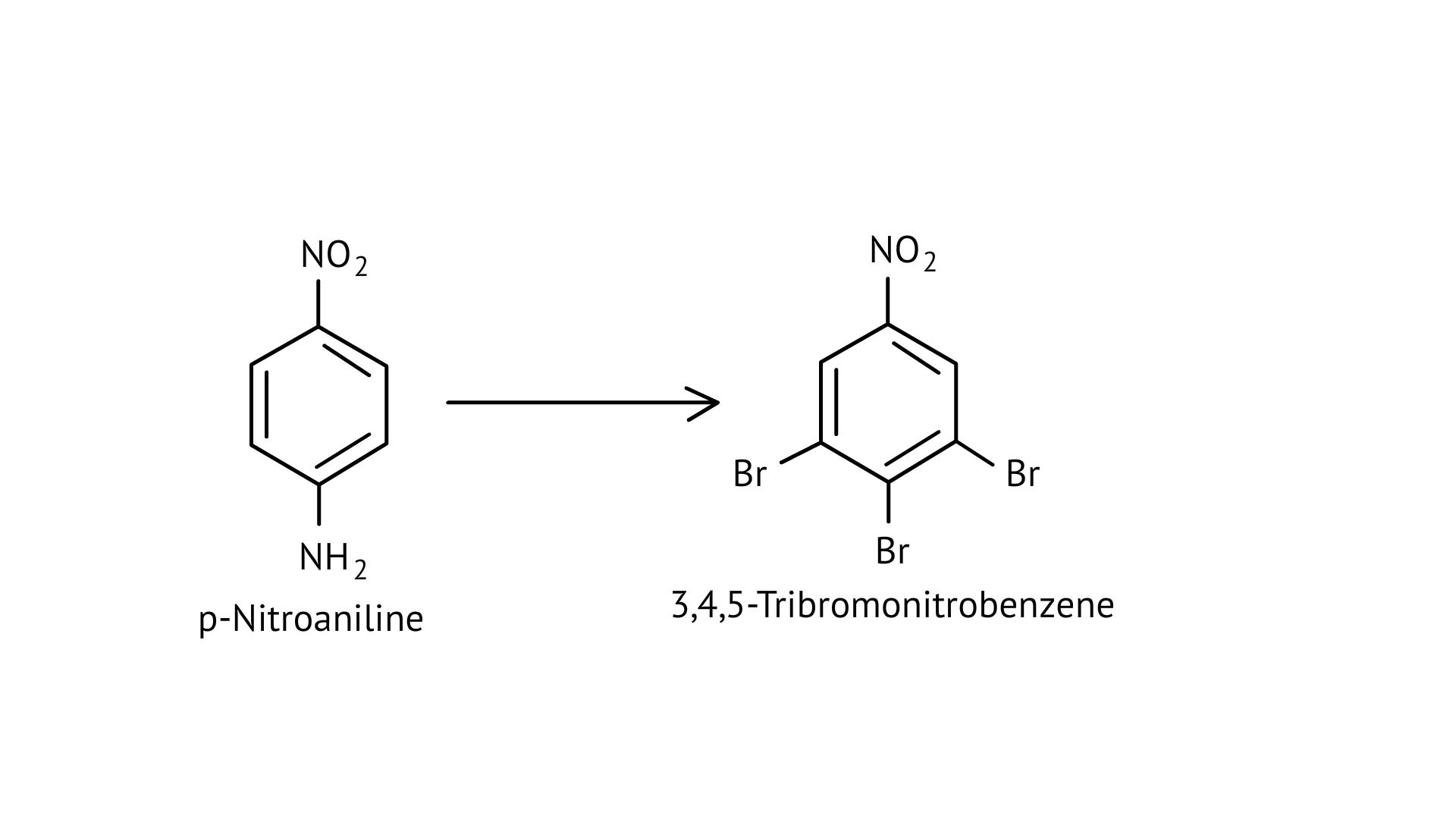
Ans:
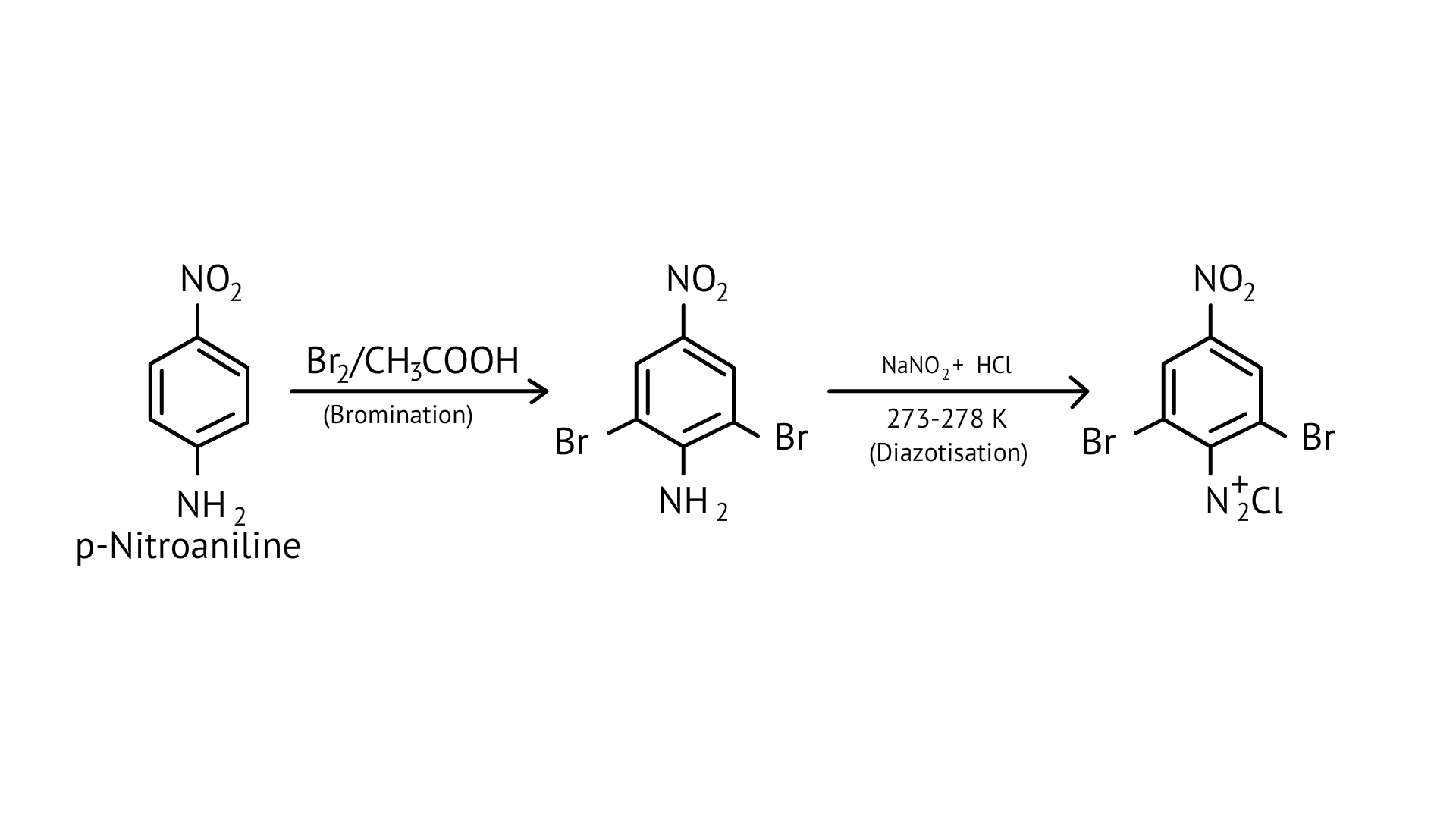
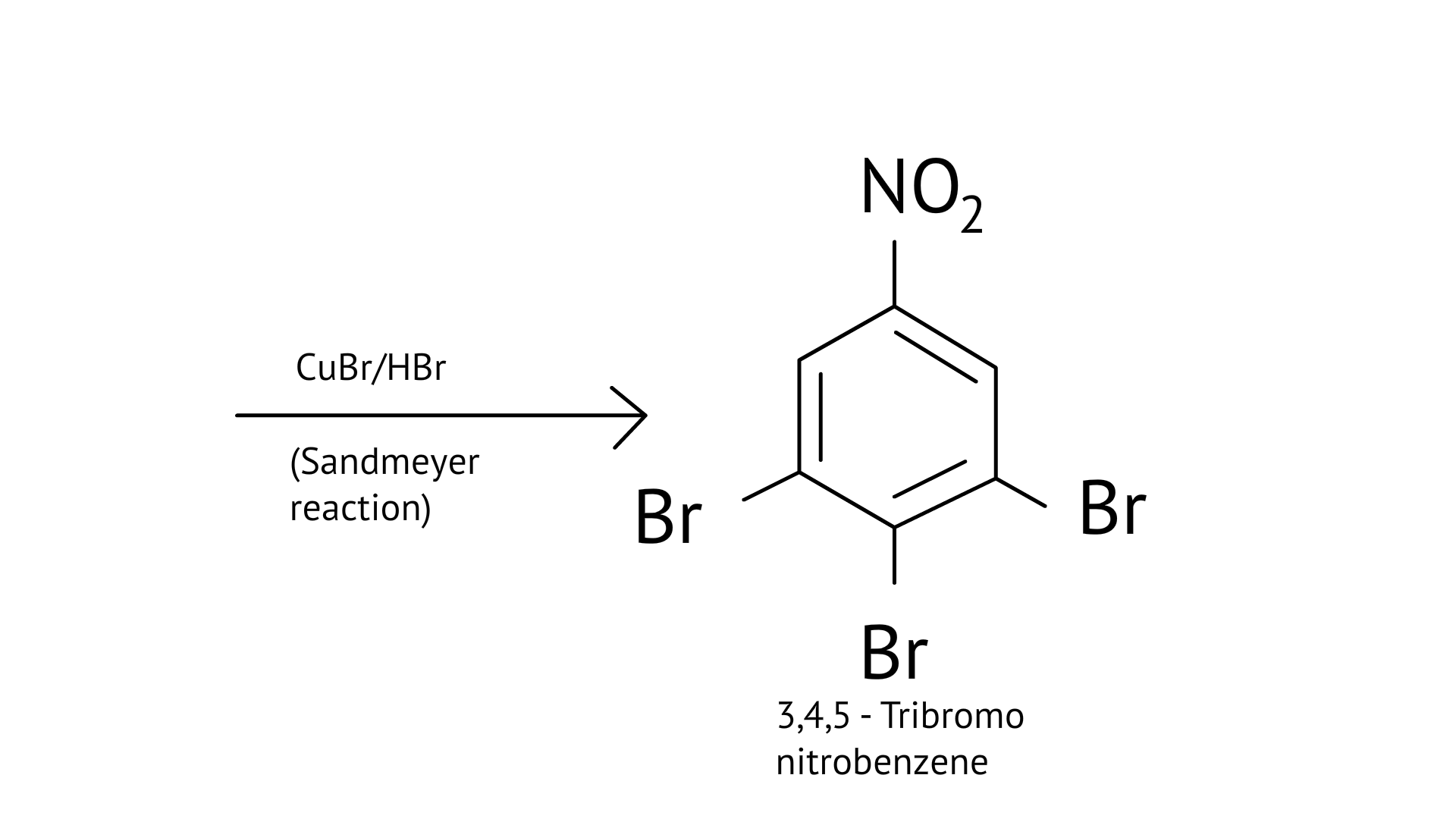
63. How will you carry out the following conversion?
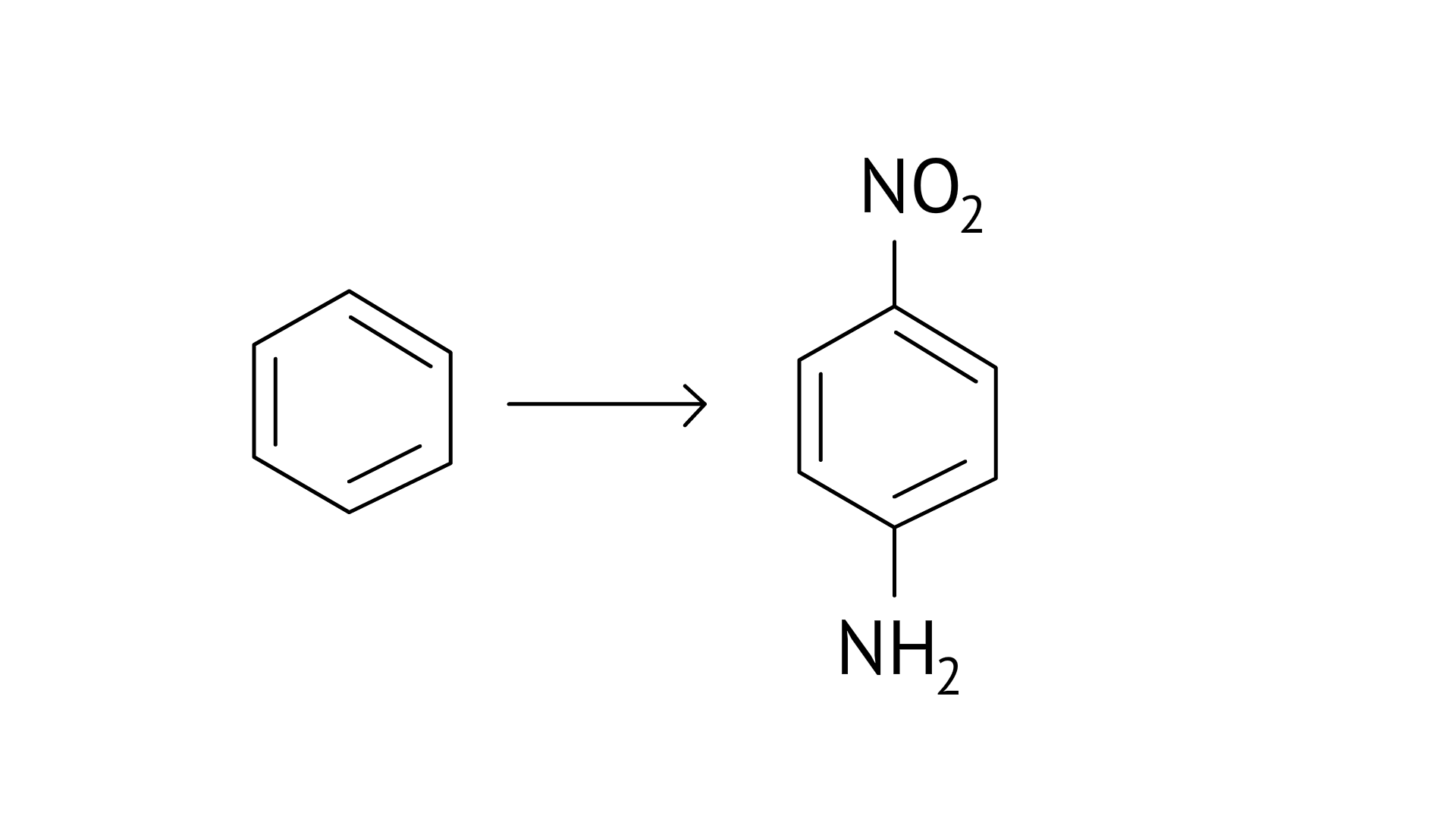
Ans:
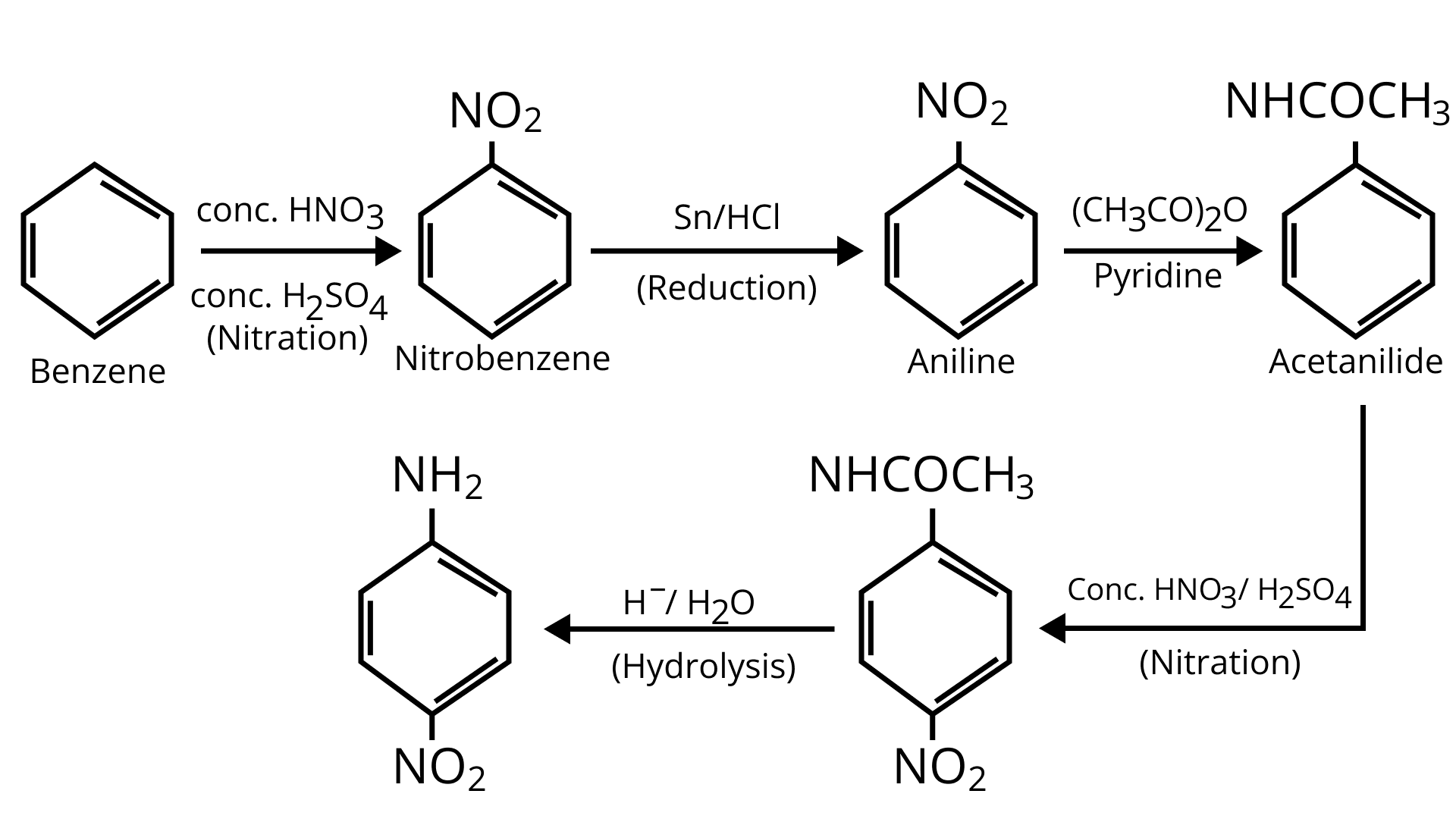
64. How will you carry out the following conversion?
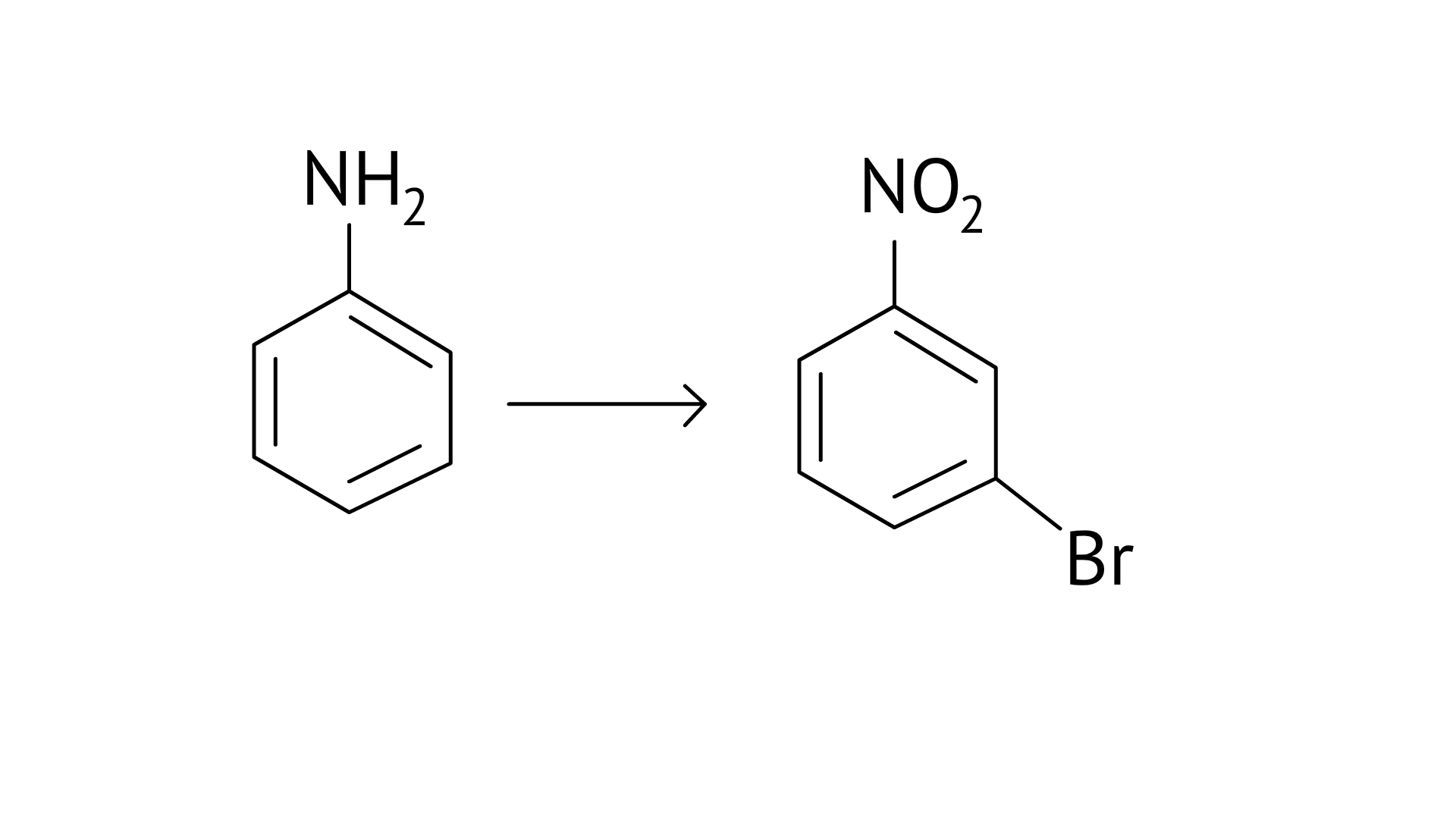
Ans:
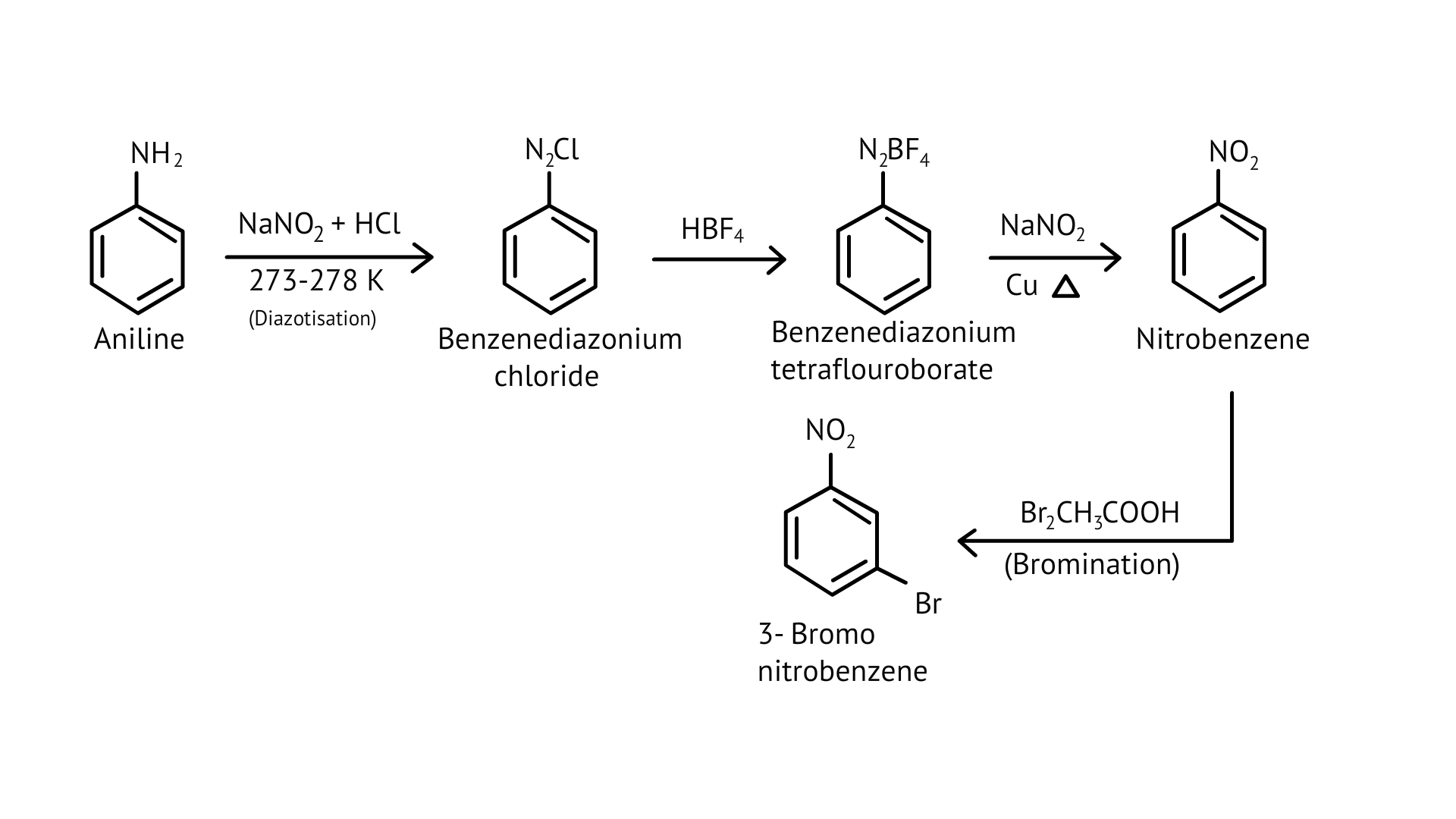
65. How will you carry out the following conversion?
(i)
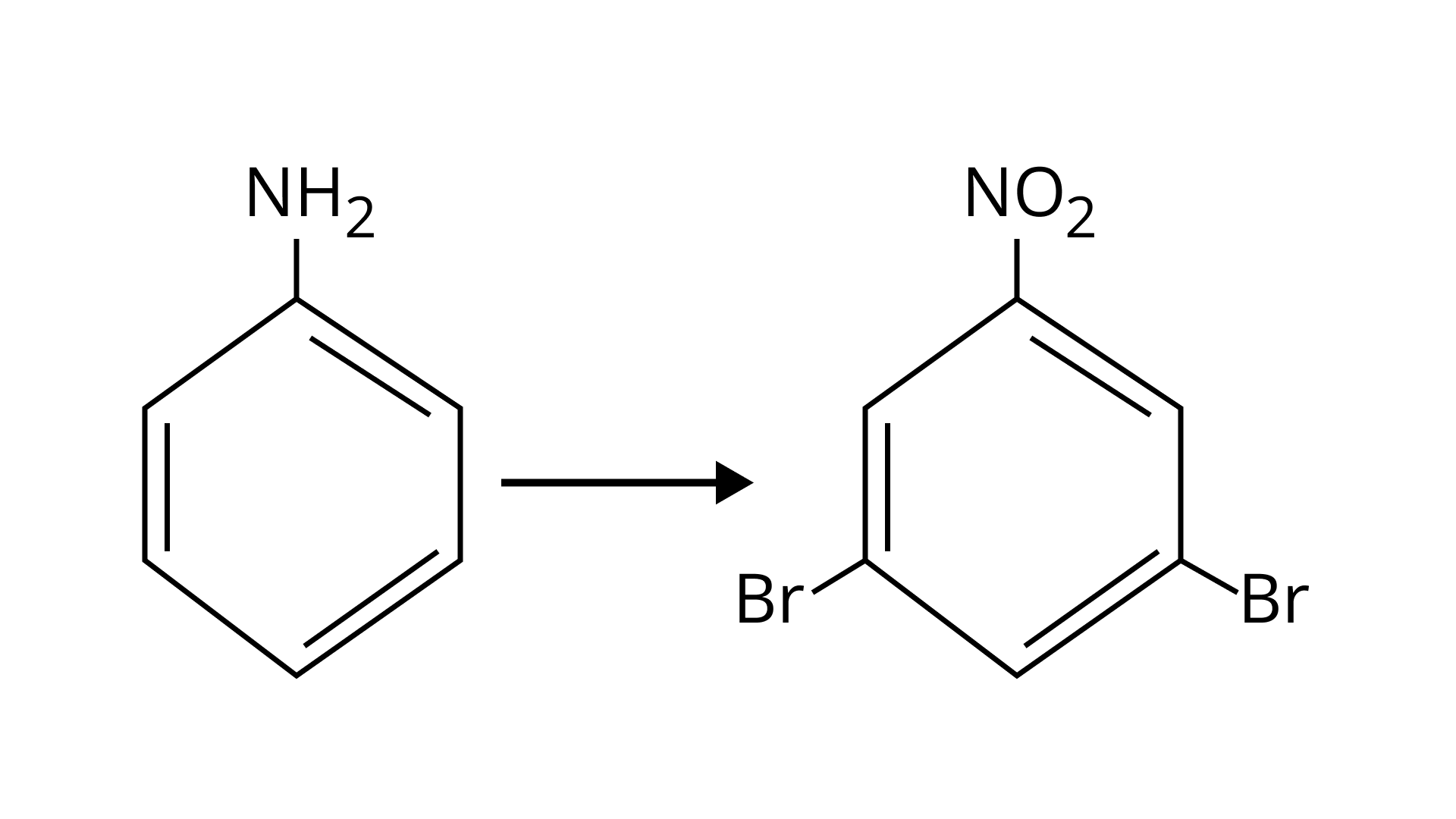
Ans:
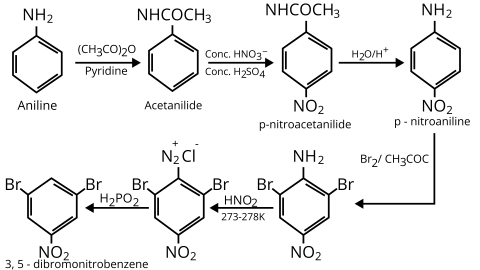
(ii)
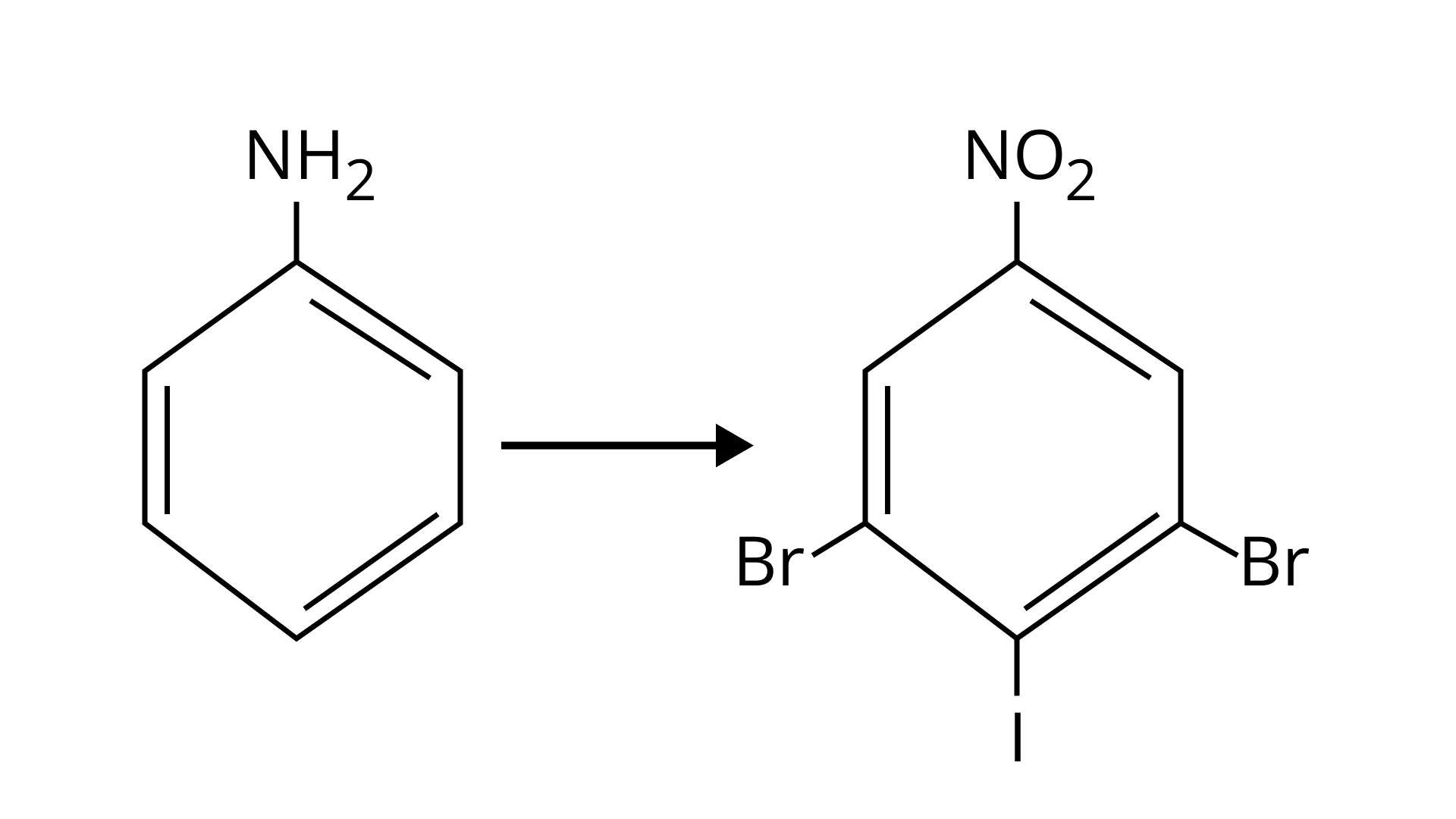
Ans:
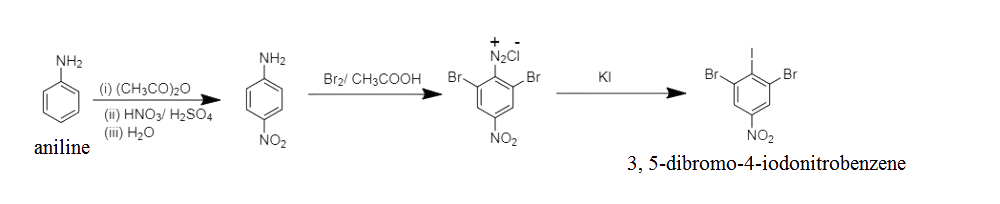
MATCH TYPE:
66. Match the reactions given in Column I with the statements given in Column II.
Column I | Column II |
(i) Ammonolysis | (a) Amine wth lesser number of carbon atoms |
(ii)Gabriel phthalimide synthesis | (B) Detection test for primary amines. |
(iii) Hoffmann Bromide reaction | (c) Reaction of phthalimide with KOH and R-X |
(iv) Carbylamine reaction | (d) Reaction of aljylhalides with $\mathrm{NH}_{3}$ |
Ans: (i)-(d), (ii)-(c), (iii)-(a), (iv)-(B)
Ammonolysis: $\mathrm{R}-\mathrm{X}+\mathrm{NH}_{3} \rightarrow \mathrm{RNH}_{2}+\mathrm{HCl}$
Carbylamine reaction:
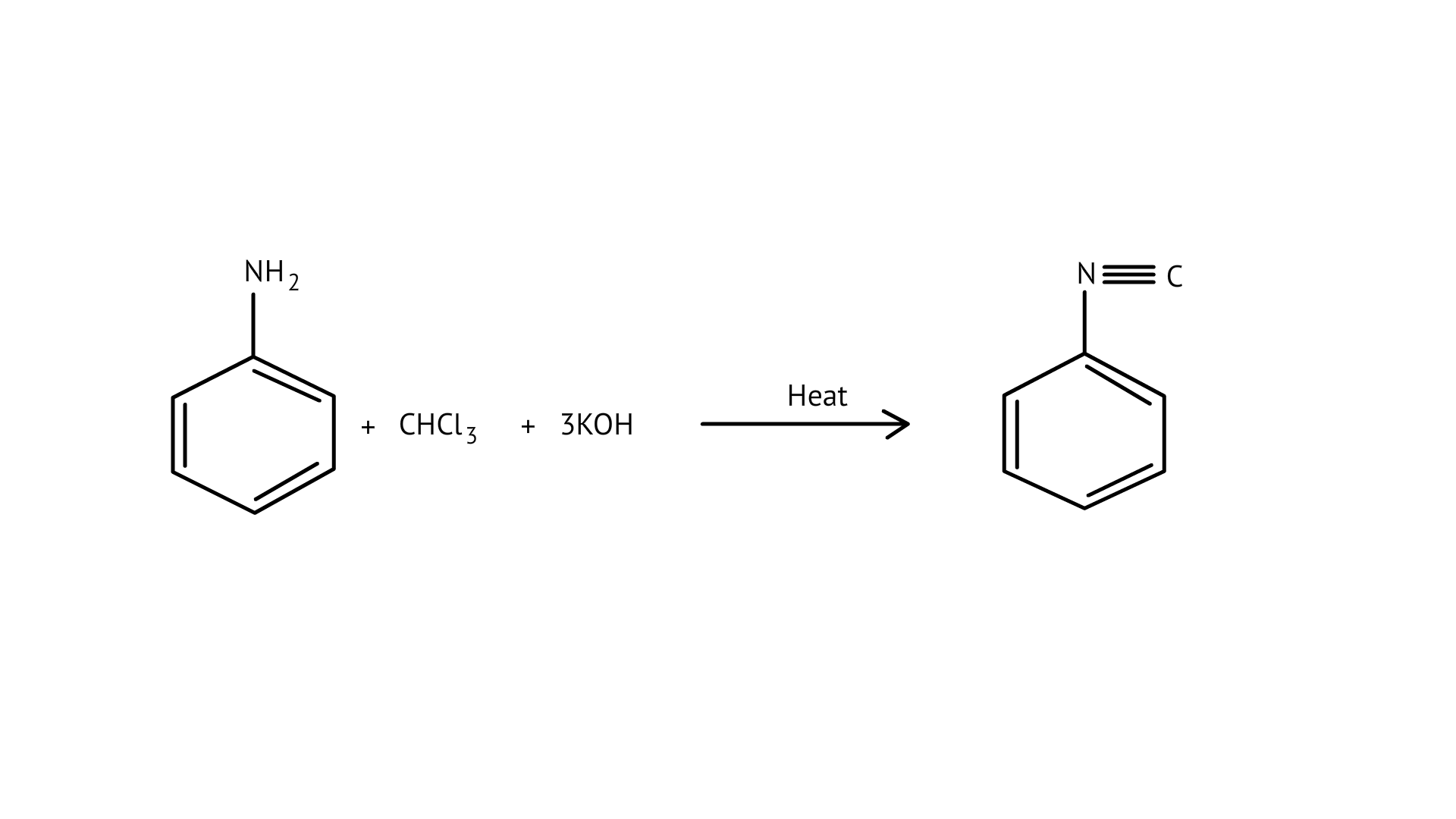
Hoffmann bromamide reaction:
$\left(\mathrm{C}_{6} \mathrm{H}_{5}\right)\left(\mathrm{CH}_{3}\right) \mathrm{CH}-\mathrm{CONH}_{2} \frac{\mathrm{Br} 2 / \mathrm{NaOH}}{\underset{\text { reaction }}{\text { Hoffmannmbromamide }}}\left(\mathrm{C}_{6} \mathrm{H}_{5}\right)\left(\mathrm{CH}_{3}\right) \mathrm{CH}-\mathrm{NH}_{2}$
67. Match the compounds given in Column I with the items given in Column II.
Column I | Column II |
(i) Benzene sulphonyl chloride | (a) Zwitter ion |
(ii) Sulphanilic acid | (B) Hinsberg reagent |
(iii) Alkyl diazonium salts | (c) Dyes |
(iv) Aryl diazonium salts | (d) Conversion to alcohols |
Ans:
Column I | Column II |
(i) Benzene sulphonyl chloride | (B) Hinsberg reagent |
(ii) Sulphanilic acid | (a) Zwitter ion |
(iii) Alkyl diazonium salts | (d) Conversion to alcohols |
(iv) Aryl diazonium salts | (c) Dyes |
ASSERTION REASON TYPE:
In the following questions, a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct answer out of the following choices:
(a) Both Assertion and Reason are wrong.
(B) Both Assertion and Reason are correct but Reason is not the correct explanation of Assertion.
(c) Assertion is correct but Reason is wrong.
(d) Both Assertion and Reason are correct and Reason is the correct explanation of Assertion.
(e) Assertion is wrong but Reason is correct.
68. Assertion (A): Acylation of amines gives a monosubstituted product, whereas alkylation of amines gives polysubstituted product.
Reason (R): Acyl group sterically hinders the approach of further acyl groups.
Ans: (c)
In the acryl derivative, the delocalisation of electrons of the nitrogen atom occur over the carbonyl group, this decreases the electron density on the the nitrogen atom that it no more perform as nucleophile and don’t react with next acylating alkaline molecule.
Therefore, Assertion statement is correct but reason is incorrect.
69. Assertion (A): Hofmann’s bromamide reaction is given by primary amines. Reason (R): Primary amines are more basic than secondary amines.
Ans: (a)
In Hoffmann bromamide degradation reaction as in this reaction primary amide group is treated with halogen first bromine then the halogen substituted amide product is coverts to primary amine with the release of carbon dioxide gas. Both the statements assertion and reason are incorrect therefore the correct option is a.
70. Assertion (A): N-Ethylbenzene sulphonamide is soluble in alkali.
Reason (R): Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Ans: (d)
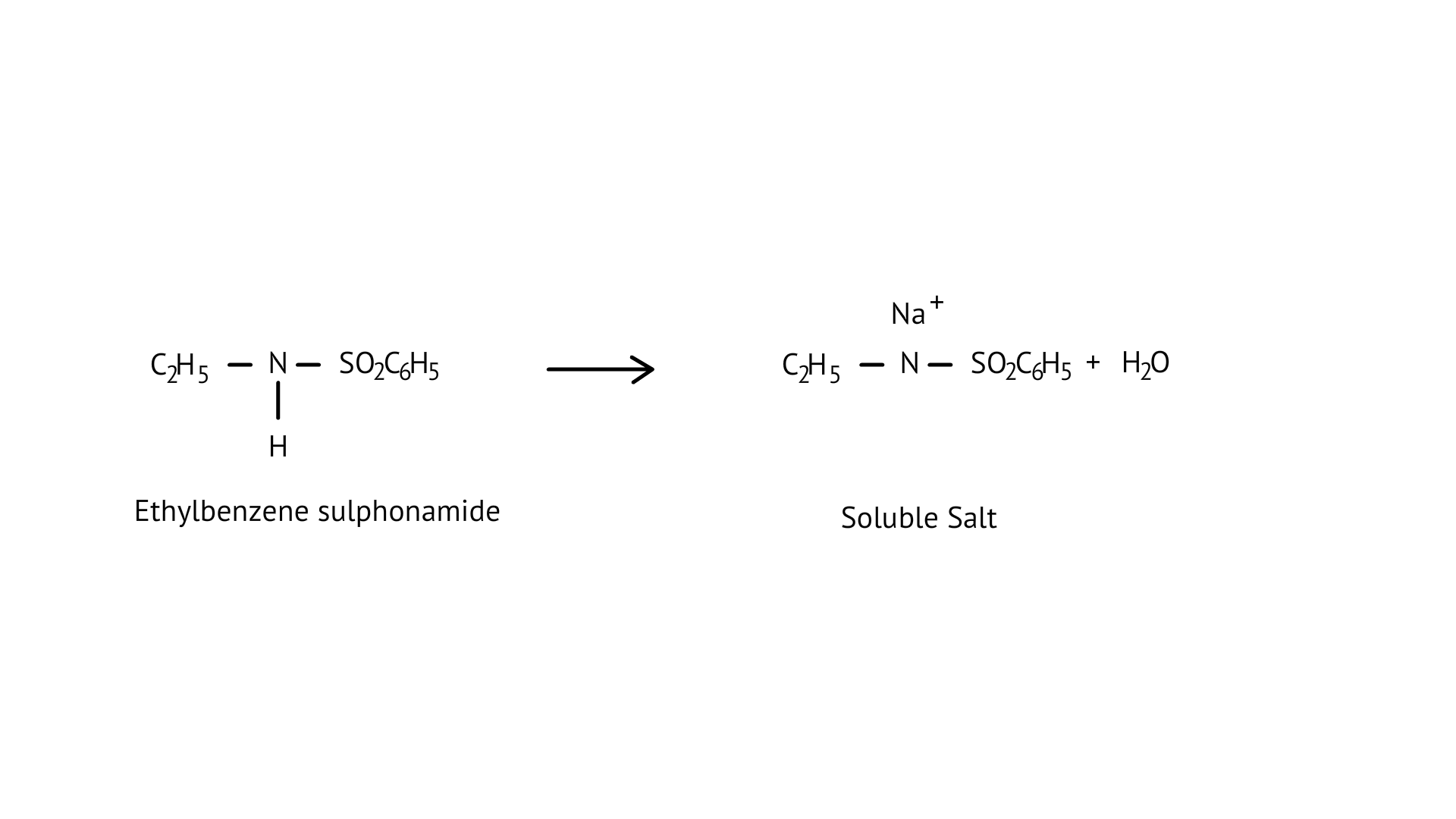
Ethylbenzene sulphonamide is soluble in alkali because it has acidic hydrogen.
71. Assertion (A): N, N-diethylbenzene sulphonamide is insoluble in alkali.
Reason (R): Sulphonyl group attached to nitrogen atom is strong electron withdrawing group.
Ans: (b)
N, N-die ethyl benzene sulphonamide, there is no acidic hydrogen present on the N-atom which can make it soluble. Therefore, it is insoluble in alkali.
72. Assertion (A): Only a small amount of HCl is required in the reduction of nitro compounds with iron scrap and HCl in the presence of steam.
Reason (R): $\mathrm{FeCl}_{2}$ formed gets hydrolysed to release $\mathrm{HCl}$ during the reaction.
Ans: (d)
$\mathrm{Fe}+2 \mathrm{HCl} \rightarrow \mathrm{FeCl}_{2}+2[\mathrm{H}]$ $\mathrm{FeCl}_{2}+\mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \rightarrow \mathrm{FeO}+2 \mathrm{HCl}$
The nascent hydrogen formed act as reducing agent for the reduction of nitro compounds.
73. Assertion (A): Aromatic 1° amines can be prepared by Gabriel phthalimide synthesis.
Reason (R): Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Ans: (a)
Aromatic primary amines cannot be prepared using Gabriel phthalimide synthesis as aryl halides do not undergo nucleophilic substitution with anion formed by phthalimide. Both the statement's assertion and reason are incorrect, hence the correct answer is a.
74. Assertion (A): Acetanilide is less basic than aniline.
Reason (R): Acetylation of aniline results in decrease of electron density on nitrogen.
Ans: (d)
Acetanilide is less basic than aniline because electron density of nitrogen is lowered by acetyl group.
LONG ANSWER TYPE:
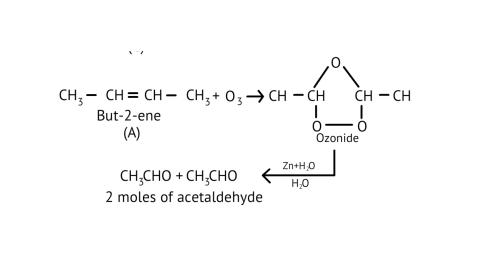
75. A hydrocarbon ' $\mathrm{A}$ ', $\left(\mathrm{C}_{4} \mathrm{H}_{8}\right)$ on reaction with $\mathrm{HCl}$ gives a compound ' $\mathrm{B}$ ' $\left(\mathrm{C}_{4} \mathrm{H}_{9} \mathrm{Cl}\right)$, which on reaction with $1 \mathrm{~mol}$ of $\mathrm{NH}_{3}$ gives compound ' $\mathrm{C}$ ', $\left(\mathrm{C}_{4} \mathrm{H}_{11} \mathrm{~N}\right) .$ On reacting with $\mathrm{NaNO}_{2}$ and $\mathrm{HCl}$ followed by treatment with water, compound ' $\mathrm{C}$ ' yields an optically active alcohol, ' $D$ '. Ozonolysis of ' $A$ ' gives 2 moles of acetaldehyde. Identify compounds 'A' to 'D'. Explain the reactions involved.
Ans:
$\underset{\text { (A) }}{\mathrm{C}_{4} \mathrm{H}_{8}} \stackrel{\mathrm{HCl}}{\longrightarrow} \underset{\text { (B) }}{\mathrm{C}_{4} \mathrm{H}_{9} \mathrm{Cl}} \stackrel{\mathrm{NH}_{3}}{\longrightarrow} \underset{\text { (C) }}{\mathrm{C}_{4} \mathrm{H}_{1,} \mathrm{~N} \text { or } \mathrm{C}_{4} \mathrm{H}_{9} \mathrm{NH}_{2}}$(ii)
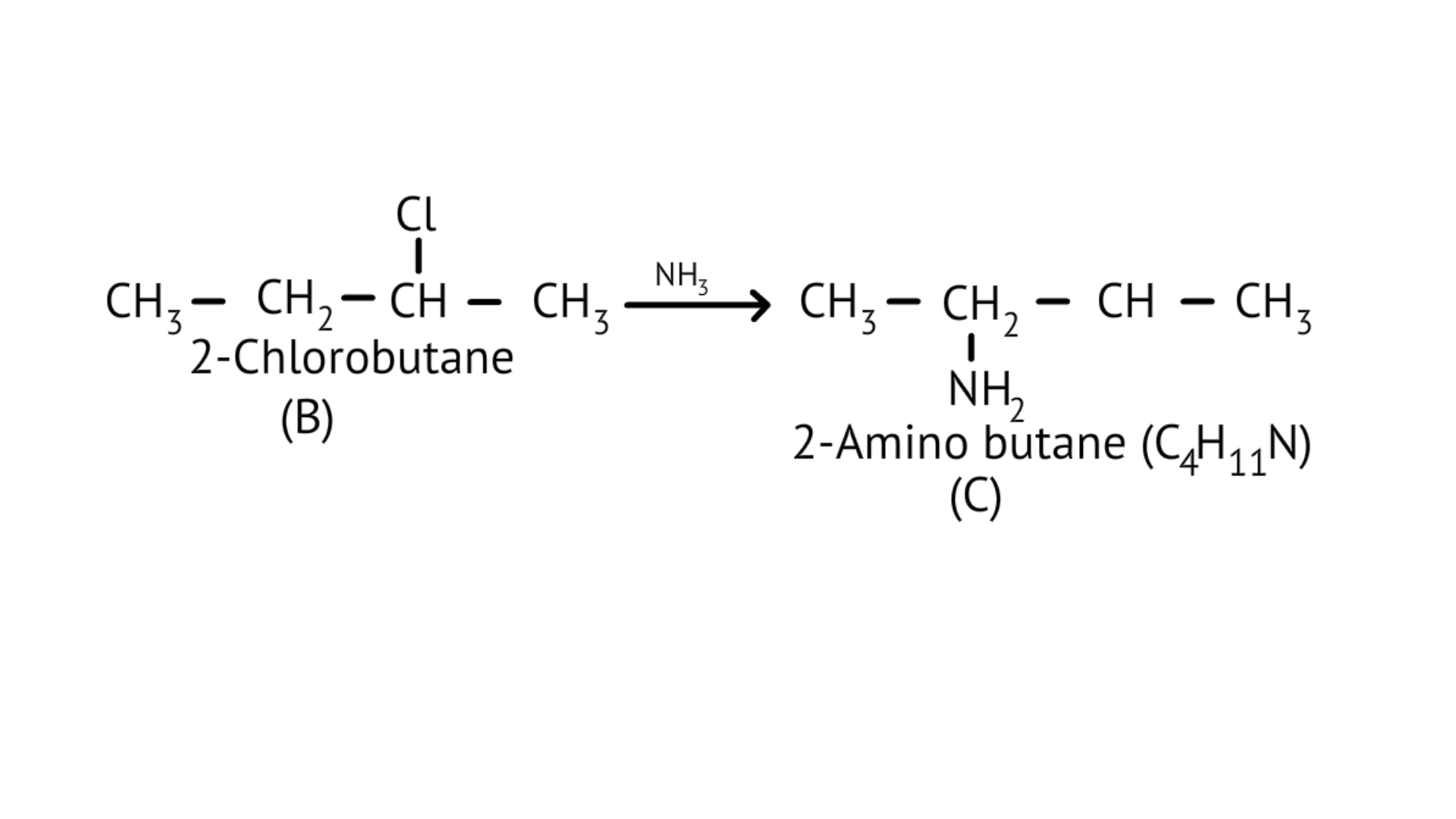
(iii)
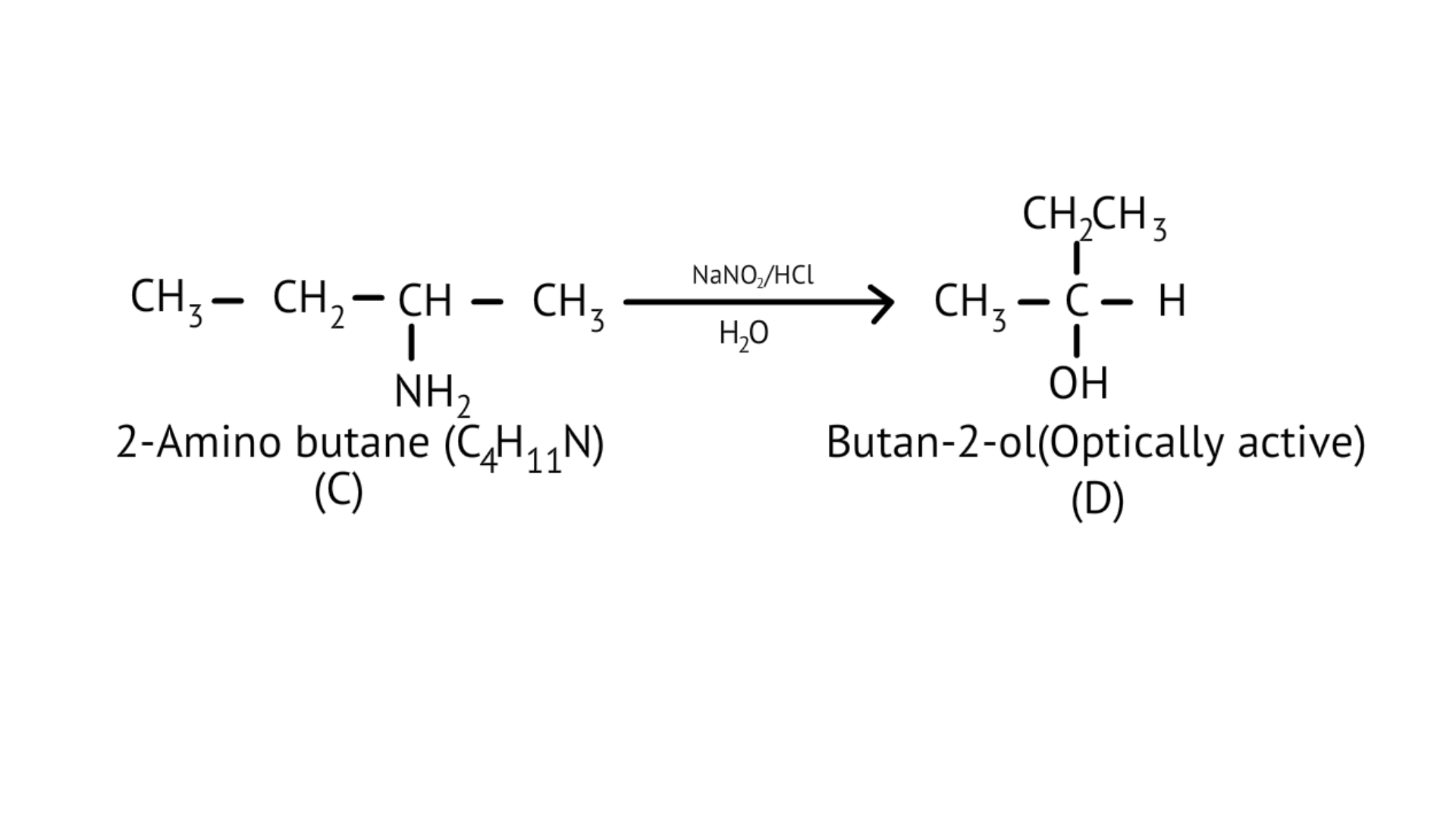
(iii)
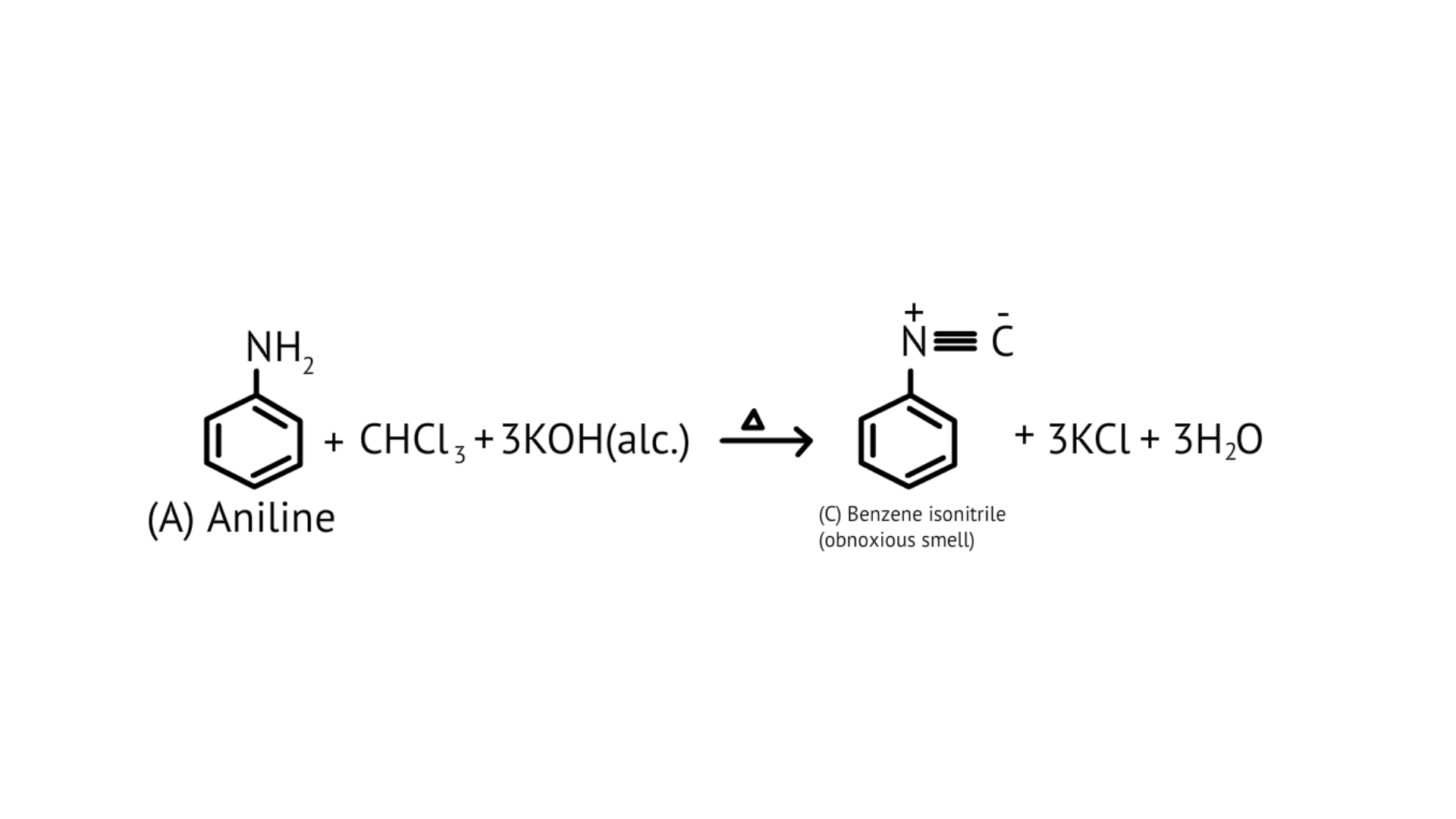
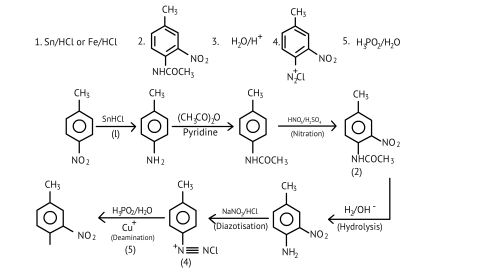
76. A colourless substance ' $\mathrm{A}^{\prime}\left(\mathrm{C}_{6} \mathrm{H}_{7} \mathrm{~N}\right)$ is sparingly soluble in water and gives a water soluble compound ' $\mathrm{B}$ ' on treatment with mineral acid. On reacting with $\mathrm{CHCl}_{3}$ and alcoholic potash 'A' produces an obnoxious smell due to the formation of compound ' $C$ '. Reaction of 'A' with benzenesulphonyl chloride gives compound ' $D$ ' which is soluble in alkali. With $\mathrm{NaNO}_{2}$ and, ' $\mathrm{A}$ ' forms compound ' $\mathrm{E}$ ' which reacts with phenol in alkaline medium to give an orange dye ' $F$ ' Identify compounds ' $A$ ' to ' $F$ '.
Ans:
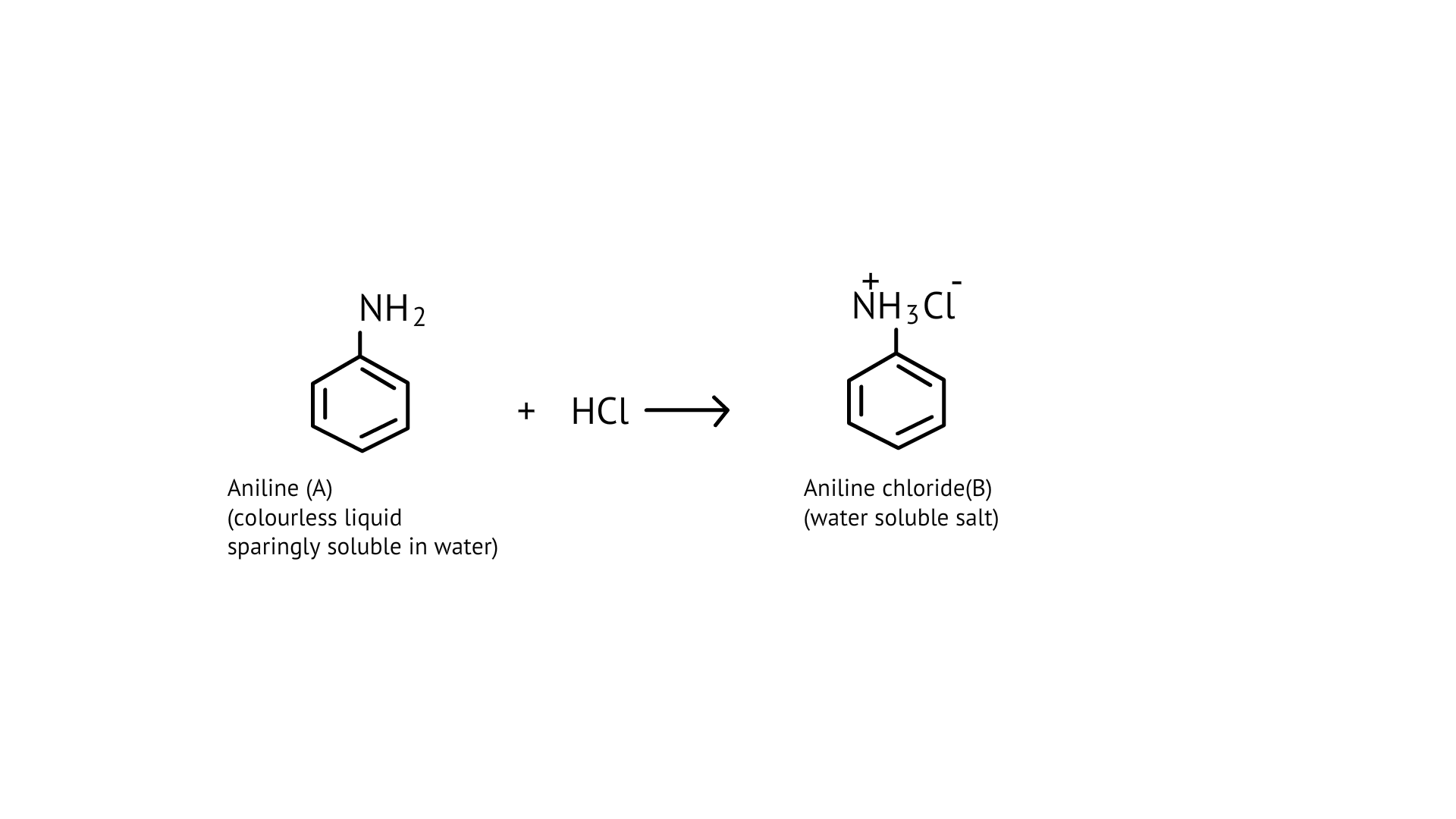
(i)
(ii)
(iii)
77. Predict the reagent or the product in the following reaction sequence.
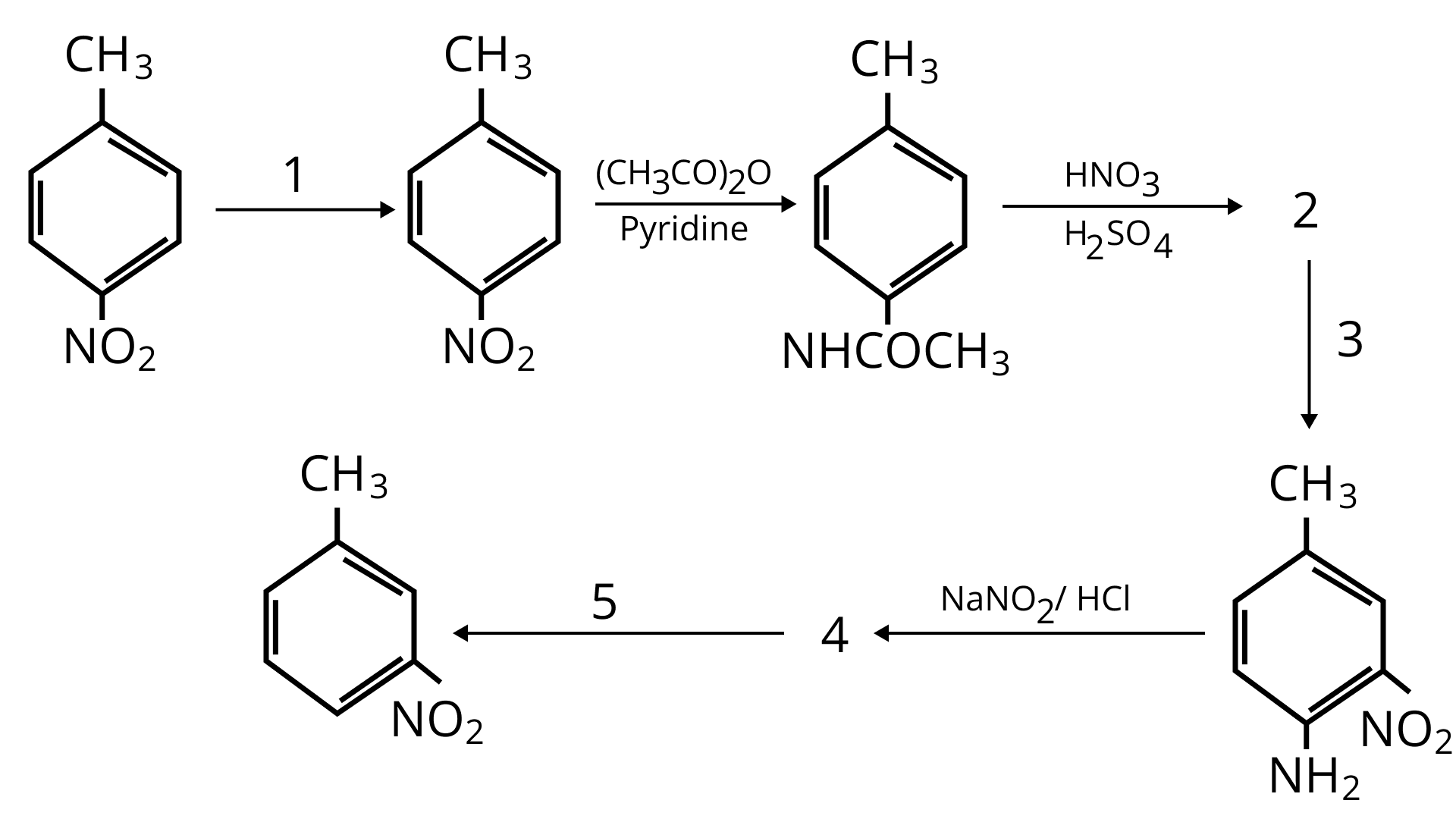
Ans:
Summary of the Chapter
Chapter 13 on Animes begins with its definition and goes on to explain its classification and structure. Physical properties of Amines come next along with its preparation. Multiple examples have been used throughout the book for the purpose of coherence. Students then learn about the reaction of Amines. The procedure of preparing aromatic amines and their subsequent reactions is next in line. The particular chapter ends with the analysis of Amines.
FAQs on NCERT Exemplar for Class 12 Chemistry - Amines - Free PDF Download
1. Where will I find details on the reactions of Amines?
Most of the information on Amines will be found on NCERT Exemplar for Class 12 Chemistry Chapter 13. The book has all the important matters that are needed for students to prepare for their assessments as well as for the sake of practice. Reactions are a complex concept to grasp and not understanding the basics could impede learning about them in-depth in future. The solved book of questions has challenging exercises for the sake of practice and will instill good understanding in the students who refer to it.
2. Where can I find chapter 13 of class 12 Chemistry online?
You will find this particular chapter on Vedantu if you go to NCERT Exemplar for Class 12 Chemistry Chapter 13 - Amines. The book has been created in tandem with Chemistry teachers who know the topics inside out and are also familiar with the NCERT guidelines in the proper sense. Solving this book will help boost confidence and also propel you to secure higher marks in all your examinations. Do not skip any of the questions that are present in the book to be on the safe side.
3. How can I make comparative notes for Class 12 Chemistry’s Chapter 13?
You can refer to NCERT Exemplar for Class 12 Chemistry Chapter 13 - Amines present on Vedantu.com. Vedantu is India’s top-most education portal for students seeking online tuitions and notes. It is trustworthy and has study material that won’t cost you even a penny. You can scan all notes and solved papers online or you can download the PDFs available so that they are always at your disposal. Making notes by referring to the solved book of exercises will equip you properly before all the possible examinations.
4. Where can I find notes on Simple Amines?
Simple amines are named as per the number of carbons in the longest continuous chain of carbon atoms that are present in any of the R groups that are attached to Nitrogen. More such information on Amines will be found in NCERT Exemplar for Class 12 Chemistry Chapter 13 - Amines. The solved book of exercises comes as a huge relief for those who are in desperate need of papers to solve from. All the explanations are lucid and in tandem with the NCERT syllabus. Free resources available for download on Vedantu. These include syllabus, notes and even chapter wise solutions for subjects.
5. Is it important to go through all the questions in NCERT Exemplar for Class 12 Chemistry Chapter 13 - Amines?
Absolutely. You must not skip any of the solved exercise questions as that would be important to selective studying. It is vital to go through all the questions in the NCERT Exemplar for Class 12 Chemistry 13- Amines to get an overall idea of what’s important and what’s not quite important. The questions are a summarized version of what’s in the main textbook and so, solving them will in turn ensure that you have properly revised the entire chapter.

























