Write the structure of nitrolic acid.
Answer
636.9k+ views
Hint: Nitrolic acid is a weak acid, which is prepared by the action of nitrous acid on any nitrile source.
Complete step by step answer:
Nitrolic acid is a compound with a general formula \[RC(N{{O}_{2}})=NOH\], where R can be any group. It is a weak acid and has a strong conjugate base. It is not a very popular compound and has been formed in laboratories out of curiosity.
Nitrolic acid is prepared by the action of nitrous acid - \[HN{{O}_{2}}\] on primary nitroparaffins - \[RC{{H}_{2}}N{{O}_{2}}\](a nitrile source) and that react with an alkali to give intensely red-colored solutions of their salts. The sample reaction is as follows –
\[RC{{H}_{2}}N{{O}_{2}}+HN{{O}_{2}}\to RC(N{{O}_{2}})=NOH+{{H}_{2}}O\]
This reaction was first demonstrated by Victor Meyer using a compound named - nitroethane. This reaction proceeds via attack on the nitronate anion (nitronate anion is formed as an intermediate in this reaction).
The structure of nitrolic acid is as given below –
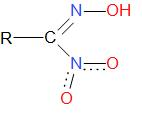
Additional Information:
Nitrous acid - \[HN{{O}_{2}}\] is a monobasic weak acid. Nitrous acid is also used to distinguish primary, secondary and tertiary amines.
Note: Pseudo Nitrile is a class of compounds with the general formula \[RR'C(NO)N{{O}_{2}}\]. It is formed by the action of nitrous acid on a disubstituted nitromethane \[RR'CHN{{O}_{2}}\] . It occurs as a pungent, colorless, solid dimers which when fused or dissolved, it depolymerizes into the monomers of intense and characteristic blue color. The reaction for preparation of Pseudo Nitrile is the same as that of Nitrolic acid –
\[RR'CHN{{O}_{2}}+HN{{O}_{2}}\to RR'C(NO)N{{O}_{2}}=NOH+{{H}_{2}}O\]
Complete step by step answer:
Nitrolic acid is a compound with a general formula \[RC(N{{O}_{2}})=NOH\], where R can be any group. It is a weak acid and has a strong conjugate base. It is not a very popular compound and has been formed in laboratories out of curiosity.
Nitrolic acid is prepared by the action of nitrous acid - \[HN{{O}_{2}}\] on primary nitroparaffins - \[RC{{H}_{2}}N{{O}_{2}}\](a nitrile source) and that react with an alkali to give intensely red-colored solutions of their salts. The sample reaction is as follows –
\[RC{{H}_{2}}N{{O}_{2}}+HN{{O}_{2}}\to RC(N{{O}_{2}})=NOH+{{H}_{2}}O\]
This reaction was first demonstrated by Victor Meyer using a compound named - nitroethane. This reaction proceeds via attack on the nitronate anion (nitronate anion is formed as an intermediate in this reaction).
The structure of nitrolic acid is as given below –

Additional Information:
Nitrous acid - \[HN{{O}_{2}}\] is a monobasic weak acid. Nitrous acid is also used to distinguish primary, secondary and tertiary amines.
Note: Pseudo Nitrile is a class of compounds with the general formula \[RR'C(NO)N{{O}_{2}}\]. It is formed by the action of nitrous acid on a disubstituted nitromethane \[RR'CHN{{O}_{2}}\] . It occurs as a pungent, colorless, solid dimers which when fused or dissolved, it depolymerizes into the monomers of intense and characteristic blue color. The reaction for preparation of Pseudo Nitrile is the same as that of Nitrolic acid –
\[RR'CHN{{O}_{2}}+HN{{O}_{2}}\to RR'C(NO)N{{O}_{2}}=NOH+{{H}_{2}}O\]
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




