Which of the following compounds does not react with $NaHS{{O}_{3}}$?
(A)- HCHO
(B)- ${{C}_{6}}{{H}_{5}}COC{{H}_{3}}$
(C)- $C{{H}_{3}}COC{{H}_{3}}$
(D)- $C{{H}_{3}}CHO$
Answer
615.6k+ views
Hint: The chemical name of $NaHS{{O}_{3}}$ is sodium bisulfite or sodium hydrogen sulphite. It is a white crystalline solid and has rotten egg-like smell. It reacts with compounds having carbonyl groups to form an addition-product.
Complete step by step answer:
Sodium bisulphate (or sodium hydrogen sulphite) reacts with carbonyl compounds mostly aldehydes (RCHO) and some ketones (RCOR) where the hydrocarbon chain, i.e. R-group is a small aliphatic chain like methyl ketone. Aromatic ketones generally do not react with $NaHS{{O}_{3}}$ due to steric effects of large benzene ring,
$NaHS{{O}_{3}}$ addition to aldehydes and ketones is a nucleophilic addition reaction. Nucleophile ($HSO_{3}^{-}$) attacks the electrophilic carbonyl carbon. A tetrahedral alkoxide is formed as an intermediate. Final proton transfer occurs to give bisulfite addition compounds. If a large or bulky R-group is present adjacent to the electrophilic carbonyl carbon then the addition of $HSO_{3}^{-}$ is hindered.
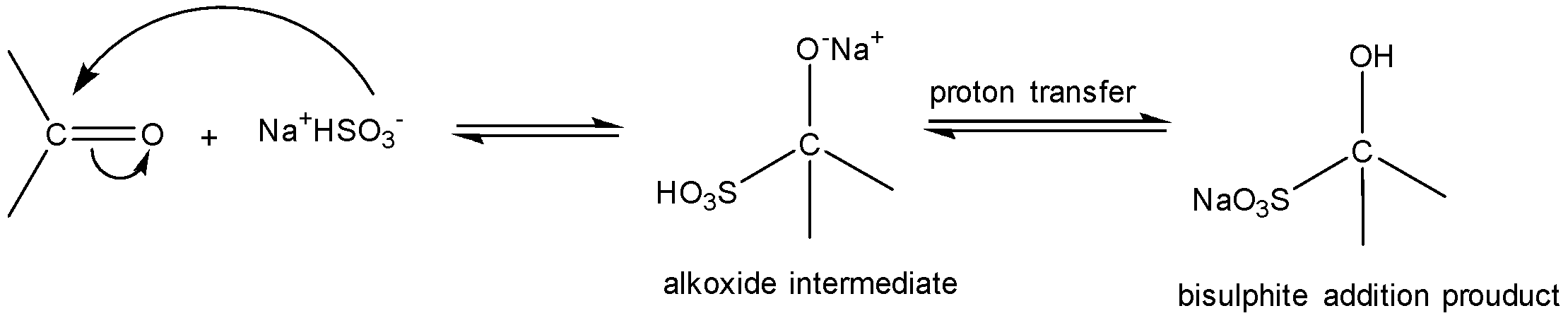
HCHO (formaldehyde), $C{{H}_{3}}CHO$(acetaldehyde) are aldehydes and thus react with $NaHS{{O}_{3}}$ easily to addition products. However, reactivity of ketones is less than that of aldehydes. $C{{H}_{3}}COC{{H}_{3}}$ (acetone) has only methyl substituents and $HSO_{3}^{-}$ addition can occur.
${{C}_{6}}{{H}_{5}}COC{{H}_{3}}$ (acetophenone) has one phenyl group attached and the attack of the nucleophile is sterically favourable. Hence, ${{C}_{6}}{{H}_{5}}COC{{H}_{3}}$ does give addition reaction with $NaHS{{O}_{3}}$.
So, the correct answer is “Option B”.
Additional Information:
The addition bisulphite product formed by nucleophilic addition of $NaHS{{O}_{3}}$ to carbonyl compounds is crystalline and water soluble. It is converted back to the carbonyl compound on treatment with mineral acid or base.
Note: Note that the reaction of a carbonyl compound with $NaHS{{O}_{3}}$ nucleophile attack to carbonyl carbon occurs from sulphur (bisulfite ion) and not oxygen. Sulphur is less electronegative than oxygen and has lesser tendency than oxygen to hold electrons on it. Therefore, sulphur behaves as a better nucleophile than oxygen.
Complete step by step answer:
Sodium bisulphate (or sodium hydrogen sulphite) reacts with carbonyl compounds mostly aldehydes (RCHO) and some ketones (RCOR) where the hydrocarbon chain, i.e. R-group is a small aliphatic chain like methyl ketone. Aromatic ketones generally do not react with $NaHS{{O}_{3}}$ due to steric effects of large benzene ring,
$NaHS{{O}_{3}}$ addition to aldehydes and ketones is a nucleophilic addition reaction. Nucleophile ($HSO_{3}^{-}$) attacks the electrophilic carbonyl carbon. A tetrahedral alkoxide is formed as an intermediate. Final proton transfer occurs to give bisulfite addition compounds. If a large or bulky R-group is present adjacent to the electrophilic carbonyl carbon then the addition of $HSO_{3}^{-}$ is hindered.

HCHO (formaldehyde), $C{{H}_{3}}CHO$(acetaldehyde) are aldehydes and thus react with $NaHS{{O}_{3}}$ easily to addition products. However, reactivity of ketones is less than that of aldehydes. $C{{H}_{3}}COC{{H}_{3}}$ (acetone) has only methyl substituents and $HSO_{3}^{-}$ addition can occur.
${{C}_{6}}{{H}_{5}}COC{{H}_{3}}$ (acetophenone) has one phenyl group attached and the attack of the nucleophile is sterically favourable. Hence, ${{C}_{6}}{{H}_{5}}COC{{H}_{3}}$ does give addition reaction with $NaHS{{O}_{3}}$.
So, the correct answer is “Option B”.
Additional Information:
The addition bisulphite product formed by nucleophilic addition of $NaHS{{O}_{3}}$ to carbonyl compounds is crystalline and water soluble. It is converted back to the carbonyl compound on treatment with mineral acid or base.
Note: Note that the reaction of a carbonyl compound with $NaHS{{O}_{3}}$ nucleophile attack to carbonyl carbon occurs from sulphur (bisulfite ion) and not oxygen. Sulphur is less electronegative than oxygen and has lesser tendency than oxygen to hold electrons on it. Therefore, sulphur behaves as a better nucleophile than oxygen.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




