How many sigma and pi bonds are present in buta $ - 1,3 - $ diene?
Answer
591k+ views
Hint: To determine the sigma and pi bond present in buta $ - 1,3 - $ diene we should know the IUPAC rules for naming so, we can draw the structure of the given compound. After drawing the structure we will count one single bond as one sigma, one double bond as one sigma and one pi, triple bond as one sigma and two pi bonds.
Complete step by step answer:
The name buta $ - 1,3 - $ diene is an IUPAC name of an organic compound. From the IUPAC rules, we know that ‘buta’ tells that there are four carbon atoms in the parent chain. ‘ene’ suffix is used for the alkene. So, ‘diene’ means there are two double bonds present in the four-carbon atom parent chain. The $ - 1,3 - $ shows the position of the double bond. The rest of the valences of carbon atoms are satisfied by hydrogen atoms.
So, we can write the structure of the buta $ - 1,3 - $ diene as follows:
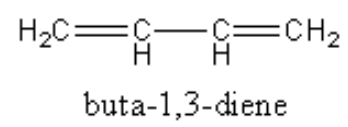
We have to open the structure to count the all sigma and pi bonds.
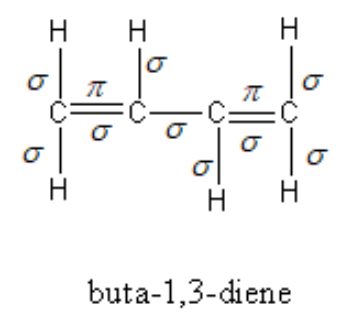
Each C-H bond is a sigma bond and C-C bond is also a sigma bond so, total of seven sigma bonds are present in the form of C-H and C-C bonds. Each C=C contains one sigma and one pi bond, so two pi bonds and two sigma bonds are present in the form of C=C.
Therefore, a total of nine sigma bonds and two pi bonds are present in buta $ - 1,3 - $ diene.
Note: The longest hydrocarbon skeleton is known as the parent chain. Sigma bond is formed by axial overlapping. s-orbitals always form sigma bonds. Hydrogen contains only one s-orbital and carbon has p-orbitals, so the C-H sigma bond forms by axial overlapping of s-p orbitals. The C-C sigma bond is formed by axial overlapping of the p-p orbital. The pi bond is formed by collateral overlapping of orbitals. The C=C forms by collateral overlapping of two p-p orbitals.
Complete step by step answer:
The name buta $ - 1,3 - $ diene is an IUPAC name of an organic compound. From the IUPAC rules, we know that ‘buta’ tells that there are four carbon atoms in the parent chain. ‘ene’ suffix is used for the alkene. So, ‘diene’ means there are two double bonds present in the four-carbon atom parent chain. The $ - 1,3 - $ shows the position of the double bond. The rest of the valences of carbon atoms are satisfied by hydrogen atoms.
So, we can write the structure of the buta $ - 1,3 - $ diene as follows:
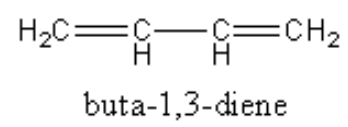
We have to open the structure to count the all sigma and pi bonds.

Each C-H bond is a sigma bond and C-C bond is also a sigma bond so, total of seven sigma bonds are present in the form of C-H and C-C bonds. Each C=C contains one sigma and one pi bond, so two pi bonds and two sigma bonds are present in the form of C=C.
Therefore, a total of nine sigma bonds and two pi bonds are present in buta $ - 1,3 - $ diene.
Note: The longest hydrocarbon skeleton is known as the parent chain. Sigma bond is formed by axial overlapping. s-orbitals always form sigma bonds. Hydrogen contains only one s-orbital and carbon has p-orbitals, so the C-H sigma bond forms by axial overlapping of s-p orbitals. The C-C sigma bond is formed by axial overlapping of the p-p orbital. The pi bond is formed by collateral overlapping of orbitals. The C=C forms by collateral overlapping of two p-p orbitals.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




