What is meant by the term coordination number in solids? What is the coordination number in a face centered cubic close packing?
Answer
612.9k+ views
Hint:We know that this question is taken from the chapter of solid state. And coordination numbers we can find by counting the number of atoms attached with a single atom.
Complete step by step solution:
> The number of atoms nearest to an atom in the crystalline structure is called the coordination number. Coordination number, also called Ligancy, the number of atoms, ions, or molecules that a central atom or ion holds as its nearest neighbours in a complex or coordination compound or in a crystal.
> The term "closest packed structures" refers to the most tightly packed or space-efficient composition of crystal structures (lattices).
- A unit cell is the smallest representation of an entire crystal. All crystal lattices are built of repeating unit cells.
- The face-centered cubic (fcc) has a coordination number of 12 and contains 4 atoms per unit cell. Let’s take an atom at the corner, this corner atom is attached with 6 other atoms of the corner and 6 atoms of face.
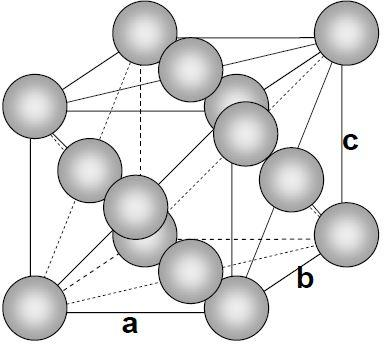
Note: The packing of spheres can describe the solid structures of crystals. In a crystal structure, the centers of atoms, ions, or molecules lie on the lattice points.
Complete step by step solution:
> The number of atoms nearest to an atom in the crystalline structure is called the coordination number. Coordination number, also called Ligancy, the number of atoms, ions, or molecules that a central atom or ion holds as its nearest neighbours in a complex or coordination compound or in a crystal.
> The term "closest packed structures" refers to the most tightly packed or space-efficient composition of crystal structures (lattices).
- A unit cell is the smallest representation of an entire crystal. All crystal lattices are built of repeating unit cells.
- The face-centered cubic (fcc) has a coordination number of 12 and contains 4 atoms per unit cell. Let’s take an atom at the corner, this corner atom is attached with 6 other atoms of the corner and 6 atoms of face.

Note: The packing of spheres can describe the solid structures of crystals. In a crystal structure, the centers of atoms, ions, or molecules lie on the lattice points.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE

No of 5 membered ring in left Caleft EDTA right right2 class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

How many 176Omega resistors in parallel are required class 12 physics CBSE

What is corrosion Explain its advantages and disad class 12 chemistry CBSE




