Calcium butanoate on heating followed by 1,2-ethanediol in the presence of catalytic amount of acid, produces a major product which is:
A.
B.
C.
D.




Answer
612.9k+ views
Hint: When ever organic reaction takes place, two types of product can be formed. One is major and the other is minor. Major product is that product which is more likely to be formed in a reaction. Minor products are those which are less likely to be formed in a chemical reaction.
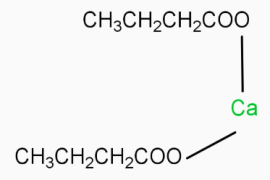 is the chemical formula for calcium butanoate.
is the chemical formula for calcium butanoate.
Complete step by step solution:
-
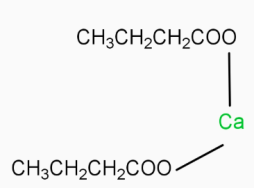 ( Calcium butanoate ) \[\underrightarrow{\Delta }\]
( Calcium butanoate ) \[\underrightarrow{\Delta }\]
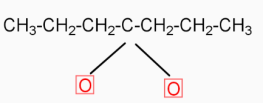
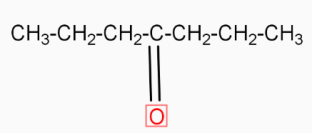 ( 4-heptanone ) Calcium butanoate when heated eliminates $\text{CaC}{{\text{O}}_{\text{3}}}$( calcium carbonate ). The product formed in this reaction is 4-heptanone
( 4-heptanone ) Calcium butanoate when heated eliminates $\text{CaC}{{\text{O}}_{\text{3}}}$( calcium carbonate ). The product formed in this reaction is 4-heptanone
- After heating it is given that it is reacted with 1,2-ethanediol in the presence of catalytic amount of acid. Acid means in the presence of ${{\text{H}}^{\text{+}}}$.
-
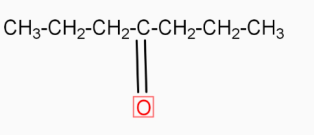 ( 4-heptanone ) in the presence of
( 4-heptanone ) in the presence of
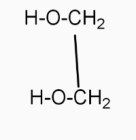 (1,2-ethanediol ) and $\underset{{}}{\overset{{{\text{H}}^{\text{+}}}}{\mathop{\to }}}\,$
(1,2-ethanediol ) and $\underset{{}}{\overset{{{\text{H}}^{\text{+}}}}{\mathop{\to }}}\,$
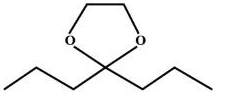
 (2,2-dipropyl-1,3-dioxolane ).
(2,2-dipropyl-1,3-dioxolane ).
So, we can say that option A is correct.
Additional Information:
- Butyrate or butanoate the name is used for the conjugate base of butyric acid.
- Butyrate is used as food for cells lining the mammalian colon.
- In the human body it is produced by our gut bacteria and supports digestive health and disease prevention.
Note: In the above reaction we use acid as a catalyst, the use of acid catalyst for organic chemical reaction is they serve as a source of protons. In the above reaction ${{\text{H}}^{\text{+}}}$ combines with the OH group to form ${{\text{H}}_{\text{2}}}\text{O}$ which is eliminate to form the final product.

Complete step by step solution:
-


- After heating it is given that it is reacted with 1,2-ethanediol in the presence of catalytic amount of acid. Acid means in the presence of ${{\text{H}}^{\text{+}}}$.
-



So, we can say that option A is correct.
Additional Information:
- Butyrate or butanoate the name is used for the conjugate base of butyric acid.
- Butyrate is used as food for cells lining the mammalian colon.
- In the human body it is produced by our gut bacteria and supports digestive health and disease prevention.
Note: In the above reaction we use acid as a catalyst, the use of acid catalyst for organic chemical reaction is they serve as a source of protons. In the above reaction ${{\text{H}}^{\text{+}}}$ combines with the OH group to form ${{\text{H}}_{\text{2}}}\text{O}$ which is eliminate to form the final product.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Which state in the country is at the forefront in controlling class 12 social science CBSE

Mention the role of cyanobacteria as a biofertiliz class 12 biology ICSE

Where is the largest hydroelectric power station located class 12 biology CBSE

Which country did Danny Casey play for class 12 english CBSE




