Assertion
Borazole is more reactive than benzene.
Reason
Electrophiles attack at nitrogen and not at boron atoms in inorganic benzene.
(A) The Assertion is true, Reason is true and Reason is the correct explanation for Assertion.
(B) The Assertion is true, Reason is true and Reason is NOT the correct explanation for Assertion.
(C) The Assertion is true, Reason is false.
(D) The Assertion is false, Reason is true.
Answer
622.5k+ views
Hint: Polarity induces reactivity in any compound. Electrophiles are species which are electron-deficient.
Complete answer:
-Borazole is a six-membered heterocyclic compound having alternate boron and nitrogen atoms and alternate double bonds. It is also known as Borazine.
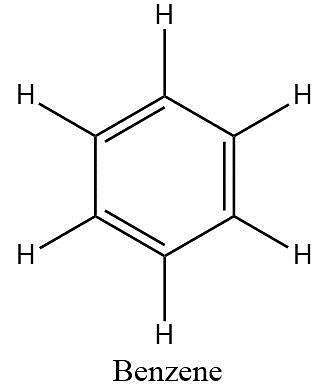
-Borazine is also known as inorganic benzene. It is isostructural to benzene. It contains three double bonds.
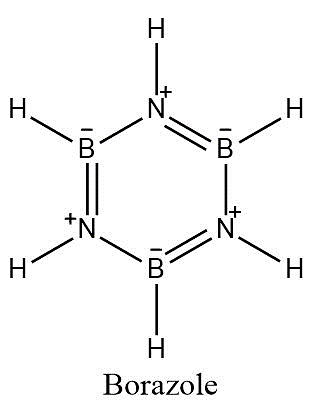
-We are aware that nitrogen is more electron-rich than boron. Nitrogen atoms have one lone pair. Nitrogen donates its lone pair to boron which is electron-deficient which gives rise to double bond between them.
-Now, nitrogen develops a positive charge and boron has a negative charge. So, there is polarity induced in the molecule. Benzene is a non-polar solvent.
-As the polarity increases, reactivity increases and thus, borazole is more polar than benzene. So, the assertion is true.
-When electrophiles attack on borazole, nitrogen atoms donate electrons to the electrophile. Boron being itself electrophilic doesn’t react with electrophiles. So, the reason is also true. But it fails to explain higher stability of benzene in contrast to borazole.
-Therefore, the correct option is option(B) The Assertion is true, Reason is true and Reason is NOT the correct explanation for Assertion.
So, the correct answer is “Option B”.
Note: Benzene and borazine are both resonating structures but due to dissimilar atoms present in borazine, it has less stability. Also, each $N=B$ bond present in a borazine ring is a polar bond which further reduces stability of the entire structure and makes it more reactive.
Complete answer:
-Borazole is a six-membered heterocyclic compound having alternate boron and nitrogen atoms and alternate double bonds. It is also known as Borazine.
-Borazine is also known as inorganic benzene. It is isostructural to benzene. It contains three double bonds.


-We are aware that nitrogen is more electron-rich than boron. Nitrogen atoms have one lone pair. Nitrogen donates its lone pair to boron which is electron-deficient which gives rise to double bond between them.
-Now, nitrogen develops a positive charge and boron has a negative charge. So, there is polarity induced in the molecule. Benzene is a non-polar solvent.
-As the polarity increases, reactivity increases and thus, borazole is more polar than benzene. So, the assertion is true.
-When electrophiles attack on borazole, nitrogen atoms donate electrons to the electrophile. Boron being itself electrophilic doesn’t react with electrophiles. So, the reason is also true. But it fails to explain higher stability of benzene in contrast to borazole.
-Therefore, the correct option is option(B) The Assertion is true, Reason is true and Reason is NOT the correct explanation for Assertion.
So, the correct answer is “Option B”.
Note: Benzene and borazine are both resonating structures but due to dissimilar atoms present in borazine, it has less stability. Also, each $N=B$ bond present in a borazine ring is a polar bond which further reduces stability of the entire structure and makes it more reactive.
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




