Which one of the following is a planar molecule
A. \[NH_3\]
B. \[H_3O^{+}\]
C. \[BCl_3\]
D. \[PCl_3\]
Answer
253.8k+ views
Hint: Trigonal planar structure is depicted by a molecule having one central atom bound to the other three atoms resembling the corner of an equilateral triangle. The bond angles are \[120^o\].
Complete step by step solution:Here in this question, we have to find out which of the following molecules has a planar structure.
A. \[NH_3\]
Here N is the central atom. Nitrogen has five electrons in its valence shell.
Three electrons are involved in bond-making and two other electrons pair up.
So, it has three bond pairs and one lone pair.
So, it has a trigonal pyramidal structure.
So, A is incorrect.
B. \[H_3O^{+}\]
Here O is the central atom. It has a positive charge and thus has five electrons in this valence shell.
Three electrons bond with three electrons each from a hydrogen atom. One lone pair is present.
So, there are three bond pairs and one lone pair.
So, it has a trigonal pyramidal structure.
So, B is incorrect.
C. \[BCl_3\]
Here Boron is the central atom.
It includes three electrons in its valence shell.
Three electrons are involved in bond-making.
So, it has three bond pairs.
So, it has a trigonal planar structure.
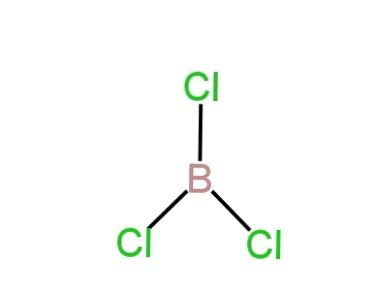
Image: Structure of BCl3
So, C is correct.
D. \[PCl_3\]
Here P is the central atom. It includes five electrons in its valence shell.
Three electrons are involved in bond-making and two other electrons pair up.
So, it has three bond pairs and one lone pair.
So, it has a trigonal pyramidal structure.
So, D is incorrect.
So, option C is correct.
Note: In chemistry, hydronium is the common name for the aqueous cation H3O+.
It is usually considered as the positive ion present when an Arrhenius acid undergoes dissolution in water.
Arrhenius acid molecules in solution lose proton \[H^+\] to the surrounding water molecules forming hydronium ions.
Complete step by step solution:Here in this question, we have to find out which of the following molecules has a planar structure.
A. \[NH_3\]
Here N is the central atom. Nitrogen has five electrons in its valence shell.
Three electrons are involved in bond-making and two other electrons pair up.
So, it has three bond pairs and one lone pair.
So, it has a trigonal pyramidal structure.
So, A is incorrect.
B. \[H_3O^{+}\]
Here O is the central atom. It has a positive charge and thus has five electrons in this valence shell.
Three electrons bond with three electrons each from a hydrogen atom. One lone pair is present.
So, there are three bond pairs and one lone pair.
So, it has a trigonal pyramidal structure.
So, B is incorrect.
C. \[BCl_3\]
Here Boron is the central atom.
It includes three electrons in its valence shell.
Three electrons are involved in bond-making.
So, it has three bond pairs.
So, it has a trigonal planar structure.

Image: Structure of BCl3
So, C is correct.
D. \[PCl_3\]
Here P is the central atom. It includes five electrons in its valence shell.
Three electrons are involved in bond-making and two other electrons pair up.
So, it has three bond pairs and one lone pair.
So, it has a trigonal pyramidal structure.
So, D is incorrect.
So, option C is correct.
Note: In chemistry, hydronium is the common name for the aqueous cation H3O+.
It is usually considered as the positive ion present when an Arrhenius acid undergoes dissolution in water.
Arrhenius acid molecules in solution lose proton \[H^+\] to the surrounding water molecules forming hydronium ions.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Inductive Effect and Its Role in Acidic Strength

Understanding Average and RMS Value in Electrical Circuits

Understanding the Electric Field Due to Infinite Linear Charge and Cylinders

Other Pages
CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reaction - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 8 Organic Chemistry - Some Basic Principles And Techniques - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 5 Thermodynamics - 2025-26

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




