Which of the following statements regarding an anion is not true?
(A) The gain of an electron leads to the formation of an anion.
(B) The radius of an anion is larger than the atomic radius of its parent atom.
(C) The effective nuclear charge increases when an anion is formed.
(D) Electron clouds expand due to increased repulsion among the electrons.
Answer
267.9k+ views
Hint: (1) An anion is attracted towards the anode in electrolysis and hence the name anion.
(2) Increase in the number of electrons leads to increase in the atomic size.
(3) Increase in the number of electrons in an atom leads to decrease in the effective nuclear charge experienced by the outermost electron.
(4) An electron cloud defines the region which has a high probability of containing electrons.
Complete step by step answer: An ion which results from an atom or molecule when it gains one or more valence electrons thereby giving a negative charge is called an anion. Thus, anions have more electrons than protons. Therefore, the statement A is true.
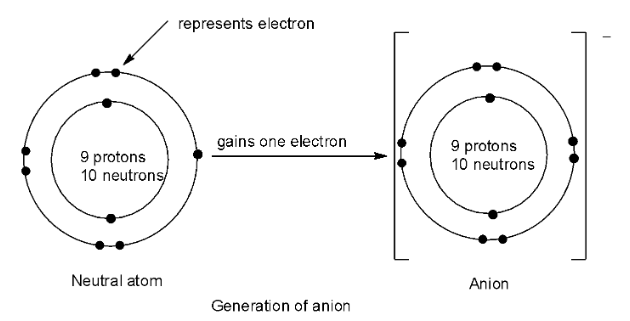
The ionic radius of an ion is equal to the distance between the centre of the nucleus of the ion and its outermost shell containing electrons. When extra electrons are added, this distance will increase and hence the ionic radius of an anion will increase and will be larger than the parent atom. So, statement B is true.
Effective nuclear charge is the attractive force faced by the electron when it is shielded by the core electrons. When the number of electrons increases, the shielding of the outermost electron by the core electrons will increase and hence the nuclear charge experienced by it will be reduced. This happens in the case of an anion. So, statement C is not true.
Due to increase in electrons in an anion, the repulsion among the electrons increases leading to the increase in the size of the electron cloud and thus the electron cloud expands. So, the statement D is true.
So, thus option C is not true.
Note: The ion which is formed by the loss of electrons is known as cation. It has more protons and electrons and hence, it has positive charge. The ionic radius of the cation is smaller than the radius of the parent atom which also means that the radius of the cation is smaller than the radius of the corresponding anion. Moreover, the effective nuclear charge will increase when a cation is formed.
(2) Increase in the number of electrons leads to increase in the atomic size.
(3) Increase in the number of electrons in an atom leads to decrease in the effective nuclear charge experienced by the outermost electron.
(4) An electron cloud defines the region which has a high probability of containing electrons.
Complete step by step answer: An ion which results from an atom or molecule when it gains one or more valence electrons thereby giving a negative charge is called an anion. Thus, anions have more electrons than protons. Therefore, the statement A is true.

The ionic radius of an ion is equal to the distance between the centre of the nucleus of the ion and its outermost shell containing electrons. When extra electrons are added, this distance will increase and hence the ionic radius of an anion will increase and will be larger than the parent atom. So, statement B is true.
Effective nuclear charge is the attractive force faced by the electron when it is shielded by the core electrons. When the number of electrons increases, the shielding of the outermost electron by the core electrons will increase and hence the nuclear charge experienced by it will be reduced. This happens in the case of an anion. So, statement C is not true.
Due to increase in electrons in an anion, the repulsion among the electrons increases leading to the increase in the size of the electron cloud and thus the electron cloud expands. So, the statement D is true.
So, thus option C is not true.
Note: The ion which is formed by the loss of electrons is known as cation. It has more protons and electrons and hence, it has positive charge. The ionic radius of the cation is smaller than the radius of the parent atom which also means that the radius of the cation is smaller than the radius of the corresponding anion. Moreover, the effective nuclear charge will increase when a cation is formed.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




