Which of the following complexes is an outer orbital complex?
(A) ${{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}$
(B) ${{[Mn{{(CN)}_{6}}]}^{4-}}$
(C) ${{[Fe{{(CN)}_{6}}]}^{4-}}$
(D) ${{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}$
Answer
266.1k+ views
Hint: The octahedral complexes are formed when there are six ligands attached to the central metal atom in a coordination complex. The hybridization of octahedral complexes is $s{{p}^{3}}{{d}^{2}}$or ${{d}^{2}}s{{p}^{3}}$ .
Complete step by step solution:
-Coordination complexes are molecular structures that have a central metal atom surrounded by different atoms or groups of atoms known as the ligand. The central metal atom is a transition metal atom.
-Ligands are bonded to central metal atoms through coordinate covalent bonds which are formed between ‘s’ and ‘p’ molecular orbital of ligands and the ‘d’ atomic orbitals of the central metal atoms.
-When hybridization occurs between atomic orbitals of the ligands and that of the metal atom, it can form two types of complexes- inner orbital complexes and outer orbital complexes.
-When the hybridization of the atomic orbitals of the central metal atom utilizes inner d-orbitals, then the complexes are said to be inner orbital complexes. The hybridization in such complexes is ${{d}^{2}}s{{p}^{3}}$.
-When the hybridization of the atomic orbitals of the central metal atom utilizes outer d-orbitals, then the complexes are said to be outer orbital complexes. The hybridization in such complexes is $s{{p}^{3}}{{d}^{2}}$.
-The inner orbital complexes are called low spin complexes, they are diamagnetic or less paramagnetic.
-The outer orbital complexes are called high spin complexes, as they have a large number of unpaired complexes.
- In option A, that is ${{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}$
The electronic configuration of Copper in the ground state (Co) $=[Ar]3{{d}^{7}}4{{s}^{2}}$
The electronic configuration of Copper in an excited state $(C{{o}^{3+}})$ \[=[Ar]3{{d}^{6}}4{{s}^{0}}\]
Since$N{{H}_{3}}$is a strong field ligand, so it will cause the pairing of the electron,
Now, the electronic configuration of Copper when ligand $N{{H}_{3}}$approaches it =
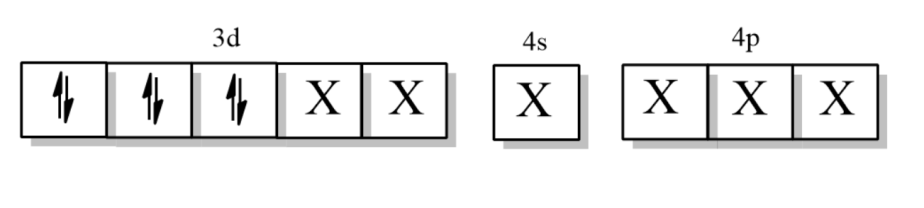
Where X is the orbitals occupied by $N{{H}_{3}}$ligand.
So, the hybridization is ${{d}^{2}}s{{p}^{3}}$, which means it is an inner orbital complex.
-In option B, that is ${{[Mn{{(CN)}_{6}}]}^{4-}}$
The electronic configuration of Manganese in the ground state (Mn)$=[Ar]3{{d}^{5}}4{{s}^{2}}$
The electronic configuration of Manganese in the excited state $(M{{n}^{2+}})=[Ar]3{{d}^{5}}4{{s}^{0}}$
Since CN is a strong field ligand, it will cause the pairing of electrons.
Now, the electronic configuration of Manganese when ligand CN approaches it =
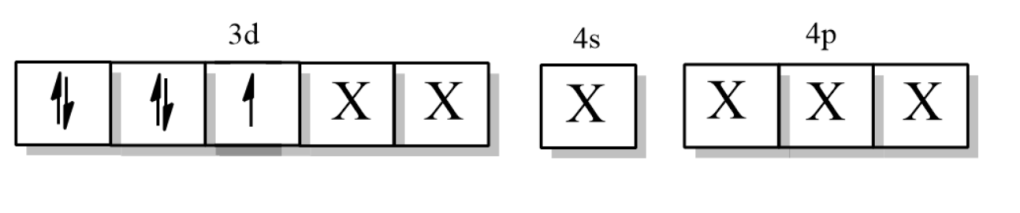
Where X is the orbitals by CN ligand.
So, the hybridization is ${{d}^{2}}s{{p}^{3}}$, which means it is an inner orbital complex.
-In option C, that is ${{[Fe{{(CN)}_{6}}]}^{4-}}$
The electronic configuration of Iron in the ground state (Fe) $=[Ar]3{{d}^{6}}4{{s}^{2}}$
The electronic configuration of Iron in an excited state $(F{{e}^{2+}})=[Ar]3{{d}^{6}}4{{s}^{0}}$
Since CN is a strong field ligand, it will cause the pairing of electrons.
Now, the electronic configuration of Iron when ligand CN approaches it =
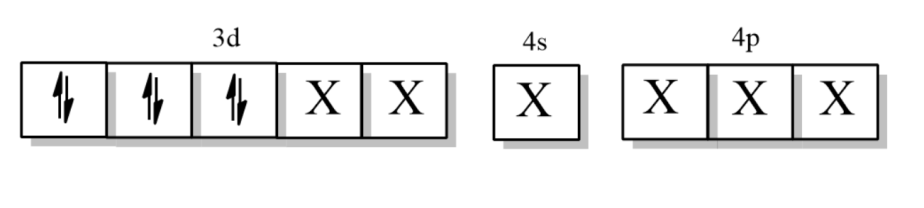
Where X is the orbitals occupied by $N{{H}_{3}}$ligand.
So, the hybridization is ${{d}^{2}}s{{p}^{3}}$, which means it is an inner orbital complex.
-In option D, that is ${{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}$
The electronic configuration of Nickel in the ground state (Ni)$=[Ar]3{{d}^{8}}4{{s}^{2}}$
The electronic configuration of Nickel in an excited state $(N{{i}^{2+}})=[Ar]3{{d}^{8}}4{{s}^{0}}$
Here, $N{{H}_{3}}$acts as a weak field ligand, so it will not cause the pairing of electrons.
Now, the electronic configuration of Nickel when CN ligand approaches it =
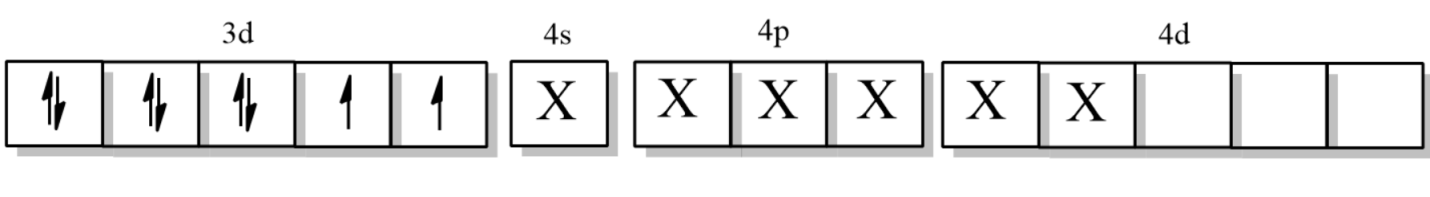
So, the hybridization is $s{{p}^{3}}{{d}^{2}}$, which means it is an outer orbital complex.
So, the correct answer is option D.
Note: According to valence bond theory, the octahedral complexes of $C{{o}^{+2}}, N{{i}^{2+}}, C{{u}^{+2}}$and $Z{{n}^{2+}}$are always outer orbital, hence the complexes formed with these ligands are always outer orbital complexes. These ions always form outer orbital complexes because they contain seven or more electrons in their inner 3d-orbital, hence in the formation of octahedral complexes they can’t attain ${{d}^{2}}s{{p}^{3}}$hybridization.
Complete step by step solution:
-Coordination complexes are molecular structures that have a central metal atom surrounded by different atoms or groups of atoms known as the ligand. The central metal atom is a transition metal atom.
-Ligands are bonded to central metal atoms through coordinate covalent bonds which are formed between ‘s’ and ‘p’ molecular orbital of ligands and the ‘d’ atomic orbitals of the central metal atoms.
-When hybridization occurs between atomic orbitals of the ligands and that of the metal atom, it can form two types of complexes- inner orbital complexes and outer orbital complexes.
-When the hybridization of the atomic orbitals of the central metal atom utilizes inner d-orbitals, then the complexes are said to be inner orbital complexes. The hybridization in such complexes is ${{d}^{2}}s{{p}^{3}}$.
-When the hybridization of the atomic orbitals of the central metal atom utilizes outer d-orbitals, then the complexes are said to be outer orbital complexes. The hybridization in such complexes is $s{{p}^{3}}{{d}^{2}}$.
-The inner orbital complexes are called low spin complexes, they are diamagnetic or less paramagnetic.
-The outer orbital complexes are called high spin complexes, as they have a large number of unpaired complexes.
- In option A, that is ${{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}$
The electronic configuration of Copper in the ground state (Co) $=[Ar]3{{d}^{7}}4{{s}^{2}}$
The electronic configuration of Copper in an excited state $(C{{o}^{3+}})$ \[=[Ar]3{{d}^{6}}4{{s}^{0}}\]
Since$N{{H}_{3}}$is a strong field ligand, so it will cause the pairing of the electron,
Now, the electronic configuration of Copper when ligand $N{{H}_{3}}$approaches it =

Where X is the orbitals occupied by $N{{H}_{3}}$ligand.
So, the hybridization is ${{d}^{2}}s{{p}^{3}}$, which means it is an inner orbital complex.
-In option B, that is ${{[Mn{{(CN)}_{6}}]}^{4-}}$
The electronic configuration of Manganese in the ground state (Mn)$=[Ar]3{{d}^{5}}4{{s}^{2}}$
The electronic configuration of Manganese in the excited state $(M{{n}^{2+}})=[Ar]3{{d}^{5}}4{{s}^{0}}$
Since CN is a strong field ligand, it will cause the pairing of electrons.
Now, the electronic configuration of Manganese when ligand CN approaches it =

Where X is the orbitals by CN ligand.
So, the hybridization is ${{d}^{2}}s{{p}^{3}}$, which means it is an inner orbital complex.
-In option C, that is ${{[Fe{{(CN)}_{6}}]}^{4-}}$
The electronic configuration of Iron in the ground state (Fe) $=[Ar]3{{d}^{6}}4{{s}^{2}}$
The electronic configuration of Iron in an excited state $(F{{e}^{2+}})=[Ar]3{{d}^{6}}4{{s}^{0}}$
Since CN is a strong field ligand, it will cause the pairing of electrons.
Now, the electronic configuration of Iron when ligand CN approaches it =

Where X is the orbitals occupied by $N{{H}_{3}}$ligand.
So, the hybridization is ${{d}^{2}}s{{p}^{3}}$, which means it is an inner orbital complex.
-In option D, that is ${{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}$
The electronic configuration of Nickel in the ground state (Ni)$=[Ar]3{{d}^{8}}4{{s}^{2}}$
The electronic configuration of Nickel in an excited state $(N{{i}^{2+}})=[Ar]3{{d}^{8}}4{{s}^{0}}$
Here, $N{{H}_{3}}$acts as a weak field ligand, so it will not cause the pairing of electrons.
Now, the electronic configuration of Nickel when CN ligand approaches it =

So, the hybridization is $s{{p}^{3}}{{d}^{2}}$, which means it is an outer orbital complex.
So, the correct answer is option D.
Note: According to valence bond theory, the octahedral complexes of $C{{o}^{+2}}, N{{i}^{2+}}, C{{u}^{+2}}$and $Z{{n}^{2+}}$are always outer orbital, hence the complexes formed with these ligands are always outer orbital complexes. These ions always form outer orbital complexes because they contain seven or more electrons in their inner 3d-orbital, hence in the formation of octahedral complexes they can’t attain ${{d}^{2}}s{{p}^{3}}$hybridization.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




