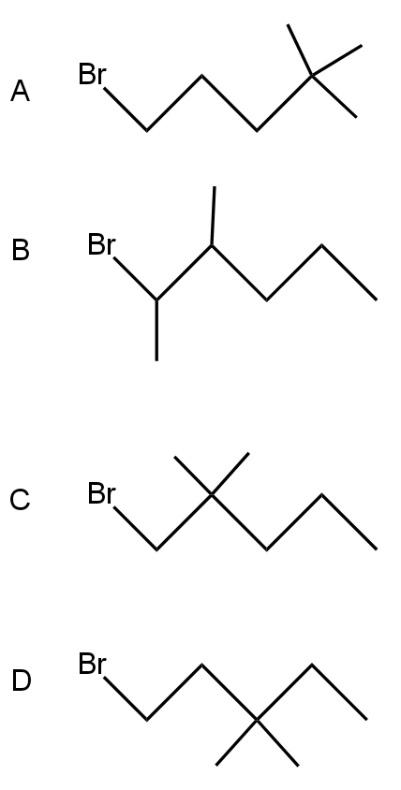
Which compound undergoes nucleophilic substitution with \[NaCN\]at the faster rate?
Answer
258k+ views
Hint: In the given options, bromine is bonded which is a good leaving group. Thus, it leaves compounds easily and generates carbonation. This carbonation will get attacked by nucleophiles. And the rate of this nucleophile attack will be greater at the carbonation which is surrounded with less bulky groups (less steric hindrance).
Complete Step by Step Solution:
Reagent \[NaCN\] is a strong nucleophile. In a nucleophilic substitution reaction, this nucleophile substitutes the nucleophile present in the given compound. Bromine's whose electronegativity is quite high as compared to its substrate (C) is a good leaving group and tends to attain a negative charge after attracting bond electrons towards itself with an inductive effect. This is the reason bromine is a good nucleophile.
As bromine (nucleophile) is a good leaving group thus from all the compounds given in the option, bromine will leave taking bond electron with itself and generating carbonation such as
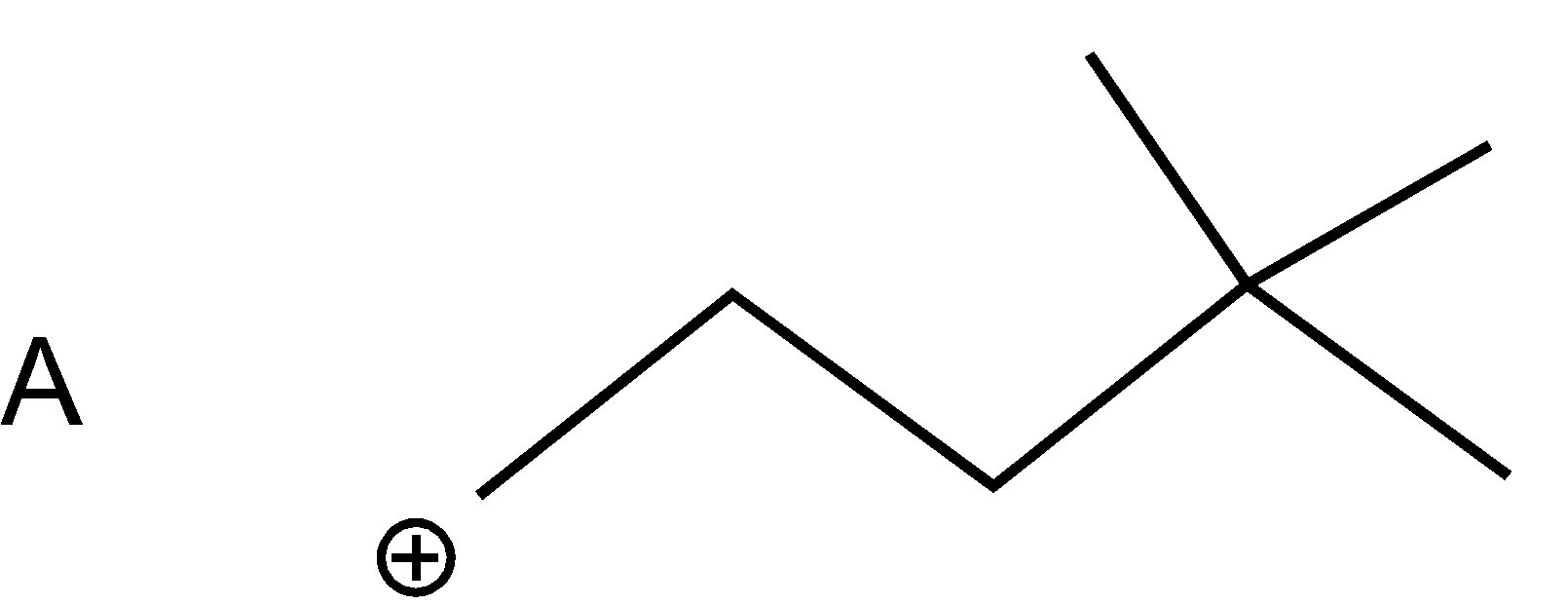
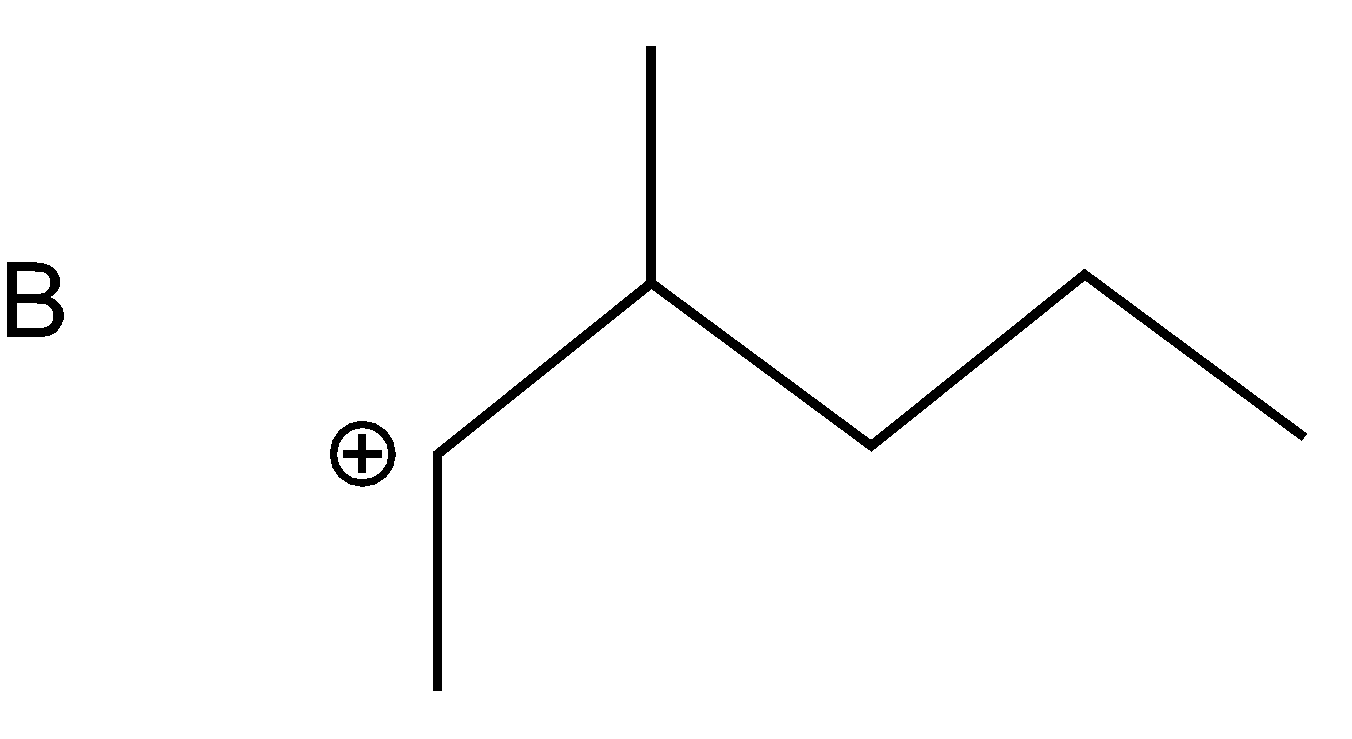
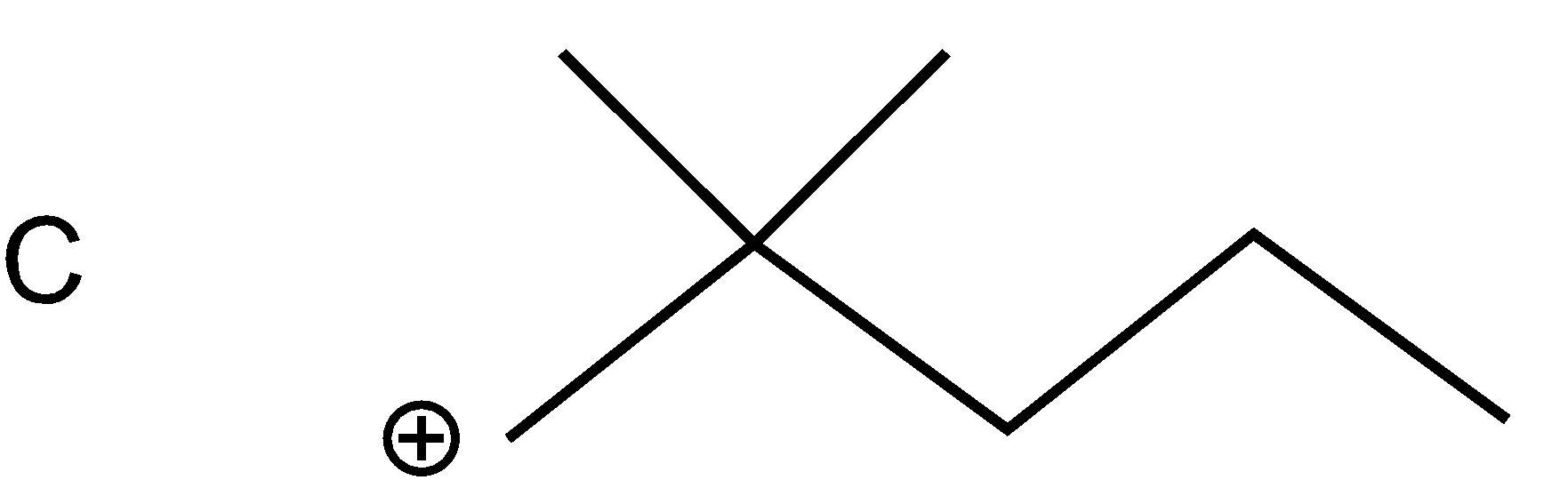
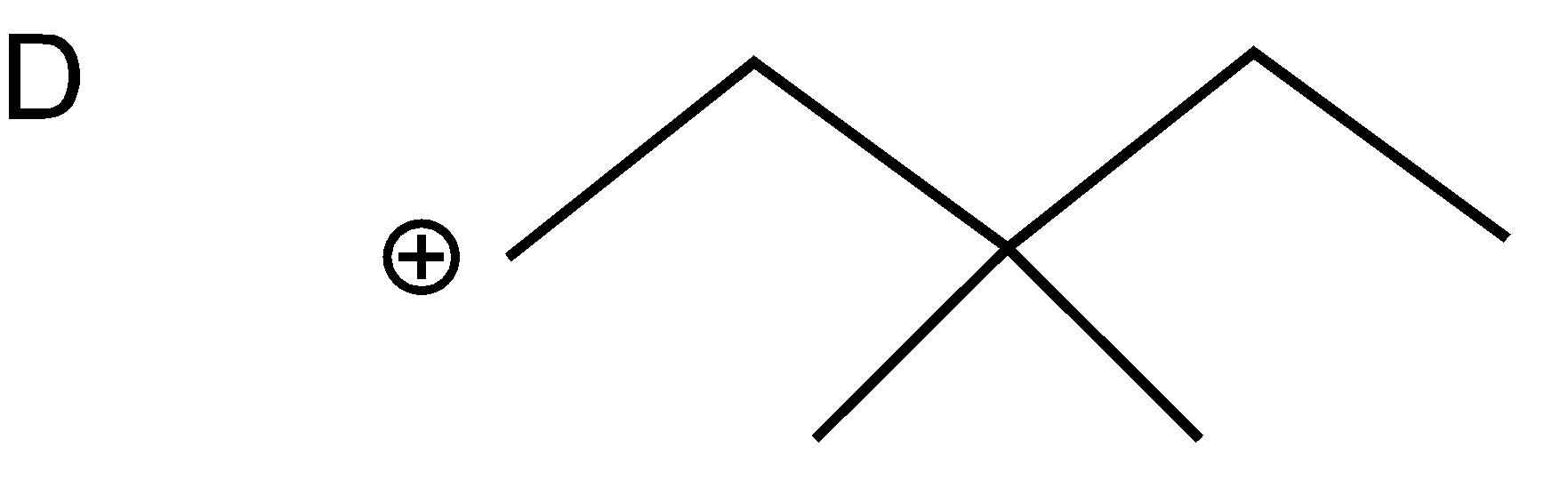
As a nucleophile leaves the compound, carbonation will form at which another nucleophile \[(C{{N}^{-}})\]will attack. Now nucleophiles attack fastly on that cation which is surrounded with a smaller number of groups (at the site of less steric hindrance) and show nucleophilic substitution reaction.
Compound A cation is less hindered thus, nucleophile firstly prefers to attack that compound. Further nucleophile attack on B compound as the bulky groups are far way the action. Compounds D and C are the compounds at which nucleophiles attack with difficulty.
Thus, the correct option is A.
Note: It is important to note that when a compound substrate is bonded with a strong base. Then this base tends to protonate through the hydrogen of carbon adjacent to the substrate and a new one more bond between substrate carbon and carbon to which protons get abstracted (alkane changes to alkene). This is an elimination reaction. If the substrate is bonded to the leaving group (strong nucleophile) thus a nucleophile takes the place of the leaving group and this is a substitution reaction.
Complete Step by Step Solution:
Reagent \[NaCN\] is a strong nucleophile. In a nucleophilic substitution reaction, this nucleophile substitutes the nucleophile present in the given compound. Bromine's whose electronegativity is quite high as compared to its substrate (C) is a good leaving group and tends to attain a negative charge after attracting bond electrons towards itself with an inductive effect. This is the reason bromine is a good nucleophile.
As bromine (nucleophile) is a good leaving group thus from all the compounds given in the option, bromine will leave taking bond electron with itself and generating carbonation such as




As a nucleophile leaves the compound, carbonation will form at which another nucleophile \[(C{{N}^{-}})\]will attack. Now nucleophiles attack fastly on that cation which is surrounded with a smaller number of groups (at the site of less steric hindrance) and show nucleophilic substitution reaction.
Compound A cation is less hindered thus, nucleophile firstly prefers to attack that compound. Further nucleophile attack on B compound as the bulky groups are far way the action. Compounds D and C are the compounds at which nucleophiles attack with difficulty.
Thus, the correct option is A.
Note: It is important to note that when a compound substrate is bonded with a strong base. Then this base tends to protonate through the hydrogen of carbon adjacent to the substrate and a new one more bond between substrate carbon and carbon to which protons get abstracted (alkane changes to alkene). This is an elimination reaction. If the substrate is bonded to the leaving group (strong nucleophile) thus a nucleophile takes the place of the leaving group and this is a substitution reaction.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




