Vinyl chloride reacts with HCl to form major product of
(A) 1,3-dichloroethane
(B) 1,2-dichloroethane
(C) Tetrachloroethylene
(D) 1,1-dichloroethane
Answer
269.7k+ views
Hint: Vinyl chloride is a gas which has no colour. It is toxic and flammable in nature. It is also termed chloroethylene. Globally, it is one of the twenty most used petrochemicals.
Complete Step by Step Solution:
Let's first understand the structure of Vinyl chloride. It is an alkene having one halogen group. The chemical structure of the compound vinyl chloride is,
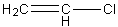
Image: Vinyl Chloride
Let's understand Markovnikov's addition reaction in detail. In Markovnikov's addition, when an alkene or alkyne undergoes a reaction with Hydrogen chloride, a proton gets attached to that C atom of double bond which has the largest count of Hydrogen atoms. And the halogen atom gets added to the other carbon atom present in the double bond.
Here, the reaction of Vinyl chloride with HCl follows Markovnikov's addition. So, the H atom of the HCl gets attached to that carbon atom of the vinyl chloride which has a larger number of H atoms. So, the reaction is,
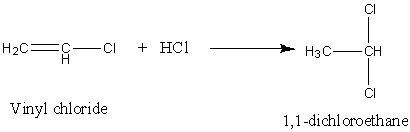
Image: Vinyl chloride undergoes reaction with HCl.
Therefore, the reaction of Vinyl Chloride and HCl gives 1,1-dichloroethane. Hence, option (D) is right.
Additional Information:
In anti-Markovnikov's addition, the addition of the proton to that C atom of the double bond occurs, where there are fewer Hydrogen atoms present. So, it is the opposite of Markovnikov's addition.
Note: Nowadays, exposure to vinyl chloride causes many health impacts. Normally, the exposure occurs because of polluted air and polluted water. Also, workers in the manufacturing industries of vinyl chloride are exposed to vinyl chloride due to inhalation.
Complete Step by Step Solution:
Let's first understand the structure of Vinyl chloride. It is an alkene having one halogen group. The chemical structure of the compound vinyl chloride is,
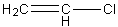
Image: Vinyl Chloride
Let's understand Markovnikov's addition reaction in detail. In Markovnikov's addition, when an alkene or alkyne undergoes a reaction with Hydrogen chloride, a proton gets attached to that C atom of double bond which has the largest count of Hydrogen atoms. And the halogen atom gets added to the other carbon atom present in the double bond.
Here, the reaction of Vinyl chloride with HCl follows Markovnikov's addition. So, the H atom of the HCl gets attached to that carbon atom of the vinyl chloride which has a larger number of H atoms. So, the reaction is,
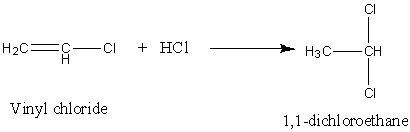
Image: Vinyl chloride undergoes reaction with HCl.
Therefore, the reaction of Vinyl Chloride and HCl gives 1,1-dichloroethane. Hence, option (D) is right.
Additional Information:
In anti-Markovnikov's addition, the addition of the proton to that C atom of the double bond occurs, where there are fewer Hydrogen atoms present. So, it is the opposite of Markovnikov's addition.
Note: Nowadays, exposure to vinyl chloride causes many health impacts. Normally, the exposure occurs because of polluted air and polluted water. Also, workers in the manufacturing industries of vinyl chloride are exposed to vinyl chloride due to inhalation.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




