Thiol group is present in:
A. cystine
B. cysteine
C. methionine
D. cytosine
Answer
267.6k+ views
Hint:
Thiol group is –SH. The amino acid containing thiol group is a triprotic acid with three ionizable functional groups including a carboxylic acid, an amino, and a sulfhydryl group and is polar and non – charged.
Complete step by step answer:
Thiol is any organosulfur compound of the form R−SH, where R can be an alkyl or aryl group. Structure :
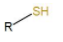
The –SH functional group is referred to as either a thiol group or a sulfanyl group.
In Thiols sulphur takes the place of oxygen in the hydroxyl group (-OH) of an alcohol.
In this question we are required to identify the compound containing thiol , among the options we can see that only cysteine contains the thiol (-SH) group.
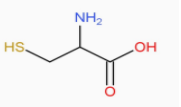
Cysteine symbol - Cys it is a polar , non- charged semi - essential proteinogenic amino acid with the formula $H{{O}_{2}}CCH(N{{H}_{2}})C{{H}_{2}}SH$.
As we can see in the diagram the thiol side chain in cysteine often participates in enzymatic reactions, as a nucleophile.
It is observed that the thiol on oxidation gives disulfide derivative cystine, which serves an important structural role in many proteins.
According to the new R/S system , based on the atomic numbers of atoms near the asymmetric carbon, we can say that cysteine shows R-chirality because sulphur is the second neighbour to the asymmetric carbon.
Cysteine shows amphoteric character in its monomeric form; it means that it can react both as an acid as well as a base.
As previously stated Cysteine is a semi – essential amino acid but for individuals with certain metabolic diseases or who suffer from malabsorption syndromes it is important.
So, the correct option is (B).
Note:
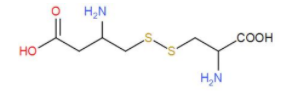
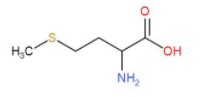
Methionine and cystine are both sulphur containing amino acids but (–SH) thiol group is not present and cystine is formed by joining two cysteine molecules together while cytosine does not have any sulphur. For better understanding study the diagrams:
Cystine -
Methionine –
Cytosine –
Thiol group is –SH. The amino acid containing thiol group is a triprotic acid with three ionizable functional groups including a carboxylic acid, an amino, and a sulfhydryl group and is polar and non – charged.
Complete step by step answer:
Thiol is any organosulfur compound of the form R−SH, where R can be an alkyl or aryl group. Structure :

The –SH functional group is referred to as either a thiol group or a sulfanyl group.
In Thiols sulphur takes the place of oxygen in the hydroxyl group (-OH) of an alcohol.
In this question we are required to identify the compound containing thiol , among the options we can see that only cysteine contains the thiol (-SH) group.

Cysteine symbol - Cys it is a polar , non- charged semi - essential proteinogenic amino acid with the formula $H{{O}_{2}}CCH(N{{H}_{2}})C{{H}_{2}}SH$.
As we can see in the diagram the thiol side chain in cysteine often participates in enzymatic reactions, as a nucleophile.
It is observed that the thiol on oxidation gives disulfide derivative cystine, which serves an important structural role in many proteins.
According to the new R/S system , based on the atomic numbers of atoms near the asymmetric carbon, we can say that cysteine shows R-chirality because sulphur is the second neighbour to the asymmetric carbon.
Cysteine shows amphoteric character in its monomeric form; it means that it can react both as an acid as well as a base.
As previously stated Cysteine is a semi – essential amino acid but for individuals with certain metabolic diseases or who suffer from malabsorption syndromes it is important.
So, the correct option is (B).
Note:
Methionine and cystine are both sulphur containing amino acids but (–SH) thiol group is not present and cystine is formed by joining two cysteine molecules together while cytosine does not have any sulphur. For better understanding study the diagrams:
Cystine -

Methionine –

Cytosine –

Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




