The shape of ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$is similar to:
(A) ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$
(B) ${{\text{O}}_{\text{2}}}{{\text{H}}_{\text{2}}}$
(C) ${{\text{H}}_{\text{2}}}{{\text{F}}_{\text{2}}}$
(D) ${{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}$
Answer
261k+ views
Hint: The bonding in the oxygen in dioxygen difluoride is different than the usual. This is particularly because of the very short distance between $\text{O-O}$ the bond and a long-distance along with the $\text{O-F}$bond. Thus the two $\text{O-F}$ bonds are arranged in the two planes perpendicular to each other. Such that the ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ exhibit open book structure.
Complete step by step solution:
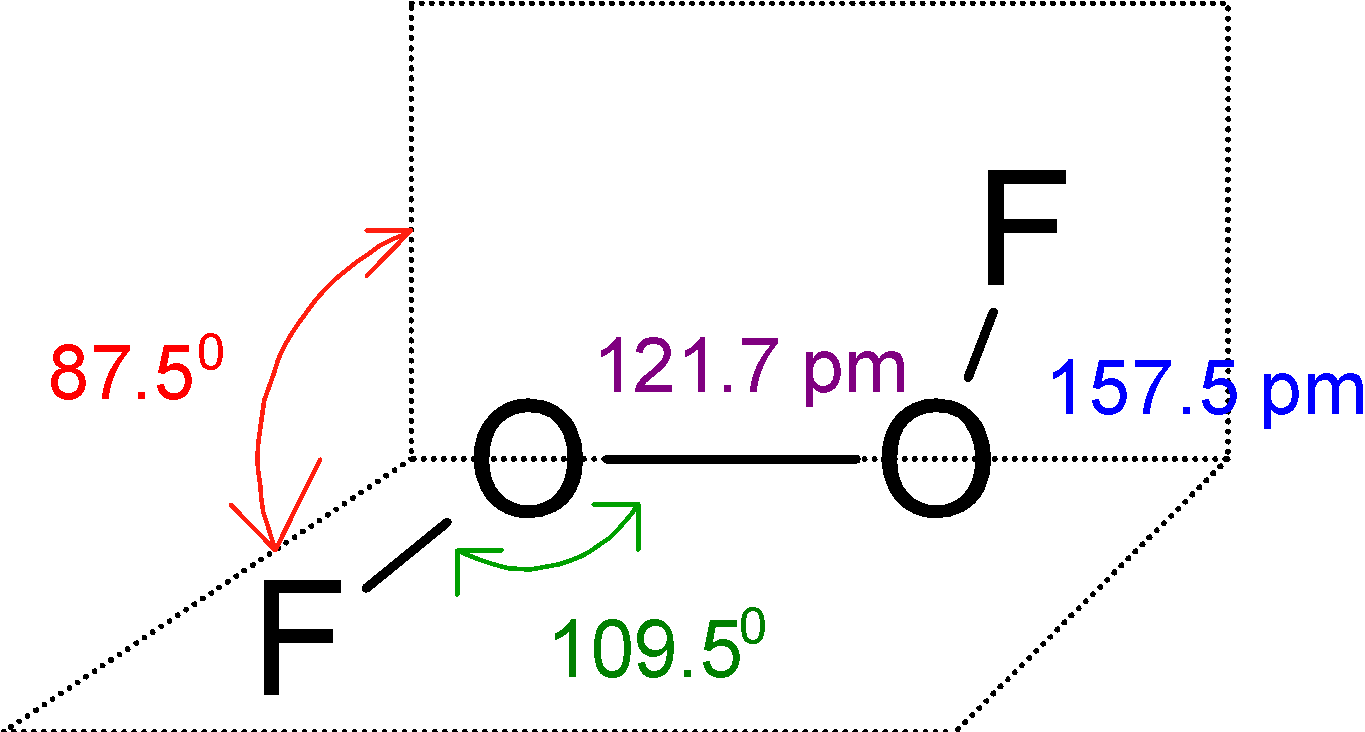
The ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$has a large dihedral angle that approaches $\text{9}{{\text{0}}^{\text{0}}}\text{C}$ and has ${{\text{C}}_{\text{2}}}$ an axis of symmetry.
The VSEPR theory is used to decide the geometry of${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$.
The two oxygen atoms are bonded by the peroxide linkage. Each oxygen atom is bonded to a fluorine atom. The Lewis dot structure for the ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$is as follows:
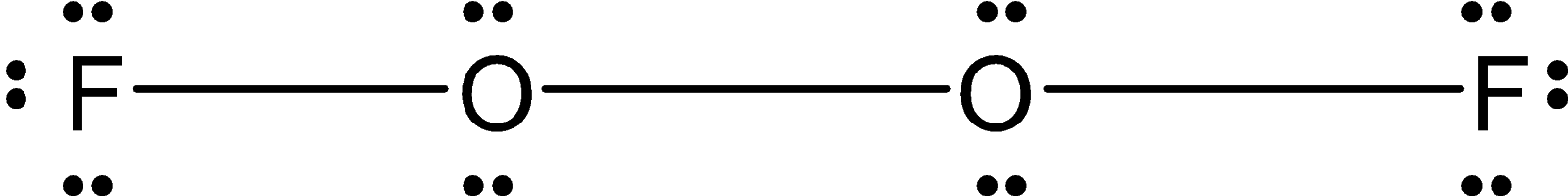
This ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is an open book type structure. Since the oxygen has the two-electron pair the structure cannot be linear but it is like the fluorine atom is in a different plane. The structure is as follows:
Let's have a look at the option.
In ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ , the two carbon atoms are forming a covalent bond and each carbon is bonded to the two fluorine atom. the structure of the ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is as shown below:
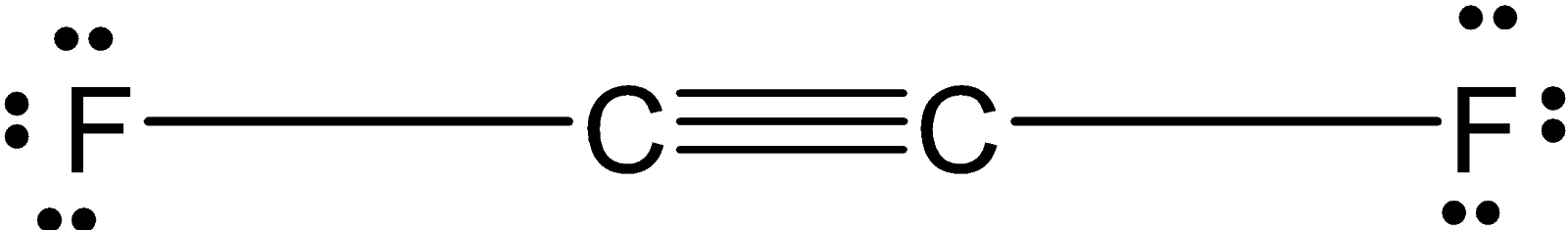
The two carbon atoms share the three bonds. Since the carbon is a $\text{sp}$ hybridized. The ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ have a linear structure.
Hydrogen peroxide ${{\text{O}}_{\text{2}}}{{\text{H}}_{\text{2}}}$ is a nonplanar molecule. Two oxygen are bonded with each other forming a peroxide bond. Each oxygen atom is bonded to the hydrogen atom. Since each oxygen has two lone pairs of electrons on it. The structure is not planar. Instead of that, it is non-planar when two hydrogen atoms are in a different plane. It has an open book structure as shown below.
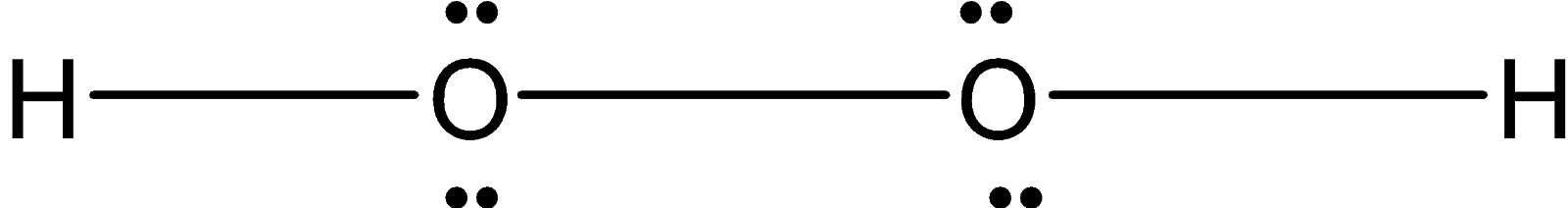
The ${{\text{H}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is a dimer of $\text{HF}$. Since the $\text{HF}$ is linear molecule the dimer ${{\text{H}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is also a linear molecule.

The ${{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}$ is a linear molecule. Here carbon is a $\text{sp}$ hybridized.
Therefore, hydrogen peroxide ${{\text{O}}_{\text{2}}}{{\text{H}}_{\text{2}}}$ has a similar shape ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$.
Hence, (B) is the correct option.
Note: In general cases, the oxygen exhibits the $-2$ oxidation state. The peroxides like hydrogen peroxide have an unusual oxidation state of oxygen. The compound is electrically neutral. Thus oxidation state of oxygen is:
$\begin{align}
& \text{2(+1) + 2(x) = 0} \\
& \text{+2 = }-\text{2 x} \\
& \therefore \text{O}\text{.S}\text{.of oxygen = }-\frac{2}{2}=\text{ }-1 \\
\end{align}$
Therefore, the oxidation state of oxygen in hydrogen peroxide is $\text{ }-1\text{ }$.
The lone pairs on oxygen restricts the linear structure of hydrogen peroxide or ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ and the structure is open book structure.
Complete step by step solution:
The ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$has a large dihedral angle that approaches $\text{9}{{\text{0}}^{\text{0}}}\text{C}$ and has ${{\text{C}}_{\text{2}}}$ an axis of symmetry.
The VSEPR theory is used to decide the geometry of${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$.
The two oxygen atoms are bonded by the peroxide linkage. Each oxygen atom is bonded to a fluorine atom. The Lewis dot structure for the ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$is as follows:
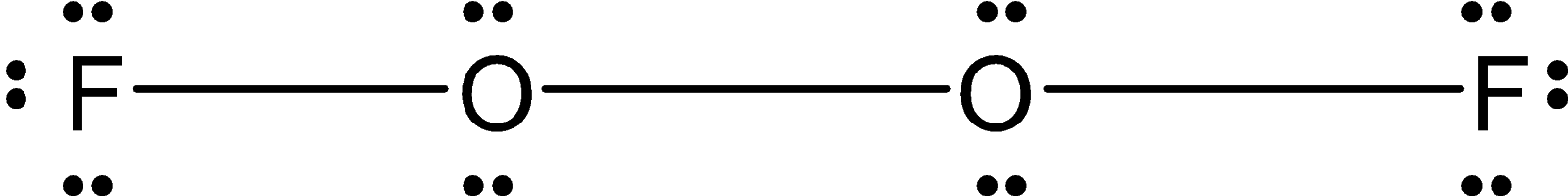
This ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is an open book type structure. Since the oxygen has the two-electron pair the structure cannot be linear but it is like the fluorine atom is in a different plane. The structure is as follows:

Let's have a look at the option.
In ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ , the two carbon atoms are forming a covalent bond and each carbon is bonded to the two fluorine atom. the structure of the ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is as shown below:
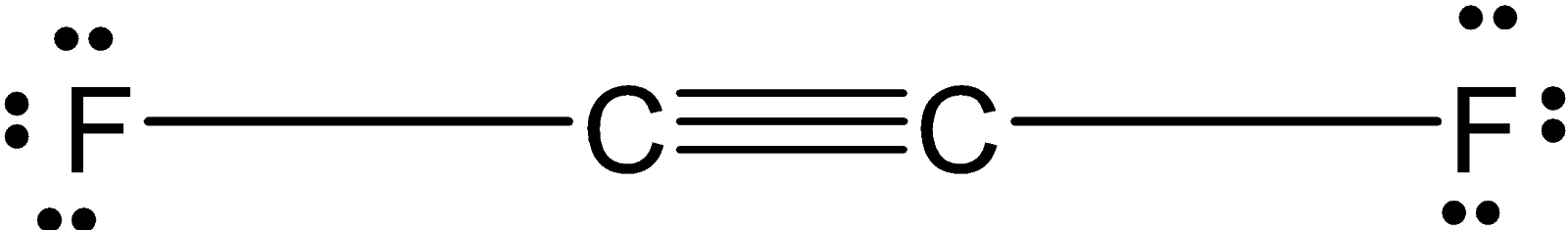
The two carbon atoms share the three bonds. Since the carbon is a $\text{sp}$ hybridized. The ${{\text{C}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ have a linear structure.
Hydrogen peroxide ${{\text{O}}_{\text{2}}}{{\text{H}}_{\text{2}}}$ is a nonplanar molecule. Two oxygen are bonded with each other forming a peroxide bond. Each oxygen atom is bonded to the hydrogen atom. Since each oxygen has two lone pairs of electrons on it. The structure is not planar. Instead of that, it is non-planar when two hydrogen atoms are in a different plane. It has an open book structure as shown below.
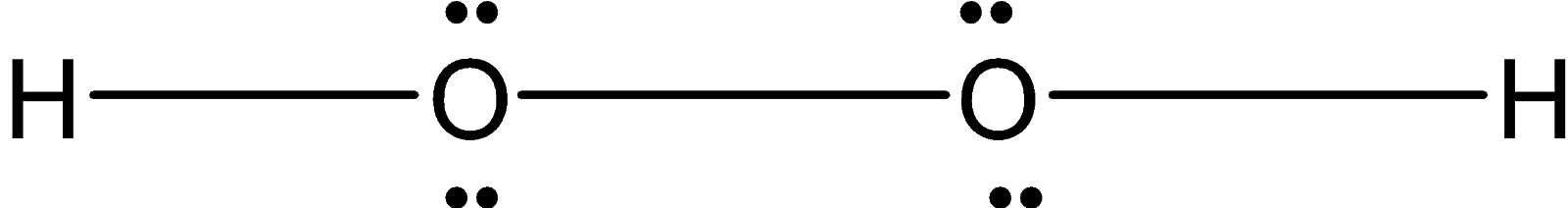
The ${{\text{H}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is a dimer of $\text{HF}$. Since the $\text{HF}$ is linear molecule the dimer ${{\text{H}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ is also a linear molecule.

The ${{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}$ is a linear molecule. Here carbon is a $\text{sp}$ hybridized.
Therefore, hydrogen peroxide ${{\text{O}}_{\text{2}}}{{\text{H}}_{\text{2}}}$ has a similar shape ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$.
Hence, (B) is the correct option.
Note: In general cases, the oxygen exhibits the $-2$ oxidation state. The peroxides like hydrogen peroxide have an unusual oxidation state of oxygen. The compound is electrically neutral. Thus oxidation state of oxygen is:
$\begin{align}
& \text{2(+1) + 2(x) = 0} \\
& \text{+2 = }-\text{2 x} \\
& \therefore \text{O}\text{.S}\text{.of oxygen = }-\frac{2}{2}=\text{ }-1 \\
\end{align}$
Therefore, the oxidation state of oxygen in hydrogen peroxide is $\text{ }-1\text{ }$.
The lone pairs on oxygen restricts the linear structure of hydrogen peroxide or ${{\text{O}}_{\text{2}}}{{\text{F}}_{\text{2}}}$ and the structure is open book structure.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




