The reaction of an aromatic halogen compound with an alkyl halide in presence of sodium and ether is called:
A. Wurtz reaction
B. Sandmeyer’s reaction
C. Wurtz-Fittig reaction
D. Kolbe reaction
Answer
256.8k+ views
Hint: It is one of the important methods that can be employed for the laboratory preparation of substituted aromatic compounds. The product formed is an alkane.
Complete Step by Step Solution:
Many of the substitution reactions which do occur when organometallic reagents are used as nucleophiles proceed with only moderate yields. The coupling of haloalkanes in the presence of an active metal is known as the Wurtz reaction. The reaction is usually accomplished by adding sodium metal to an organohalogen compound in an inert solvent. The organosodium compound which forms combines with an unreacted haloalkane.
The general equation of Wurtz reaction is as shown below:
\[{\rm{R}} - {\rm{X}} + 2{\rm{Na}} + {\rm{X}} - {\rm{R}} \xrightarrow[ether]{dry} {\rm{R}} - {\rm{R}} + {\rm{2NaX}}\];
where \[{\rm{R}}\]= alkyl group
\[{\rm{X}}\]= halogen atom
Hence, option (A) is incorrect.
In Sandmeyer’s reaction, benzene diazonium chloride is treated with cuprous halide \[{\rm{(C}}{{\rm{u}}_{\rm{2}}}{\rm{C}}{{\rm{l}}_{\rm{2}}}\,\,{\rm{or}}\,\,{\rm{C}}{{\rm{u}}_{\rm{2}}}{\rm{B}}{{\rm{r}}_{\rm{2}}}{\rm{)}}\]. Iodobenzene is obtained by treating it with potassium iodide \[{\rm{(KI)}}\]. Hence, option (B) is incorrect.
In the Wurtz-Fittig reaction, an alkyl halide and a solution of an aryl halide (aromatic halogen) in ether is made to react with sodium.
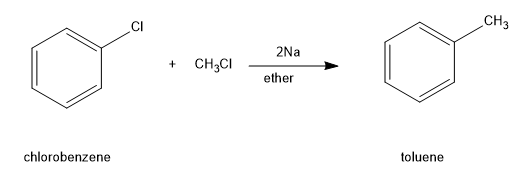
Image: reaction of an aromatic halogen (chlorobenzene) with alkyl halide (chloromethane) in presence of sodium and ether.
Hence, option (C) is correct.
In Kolbe’s reaction, sodium phenoxide on heating to \[{\rm{400}}\,\,{\rm{K}}\]with \[{\rm{C}}{{\rm{O}}_2}\]under pressure \[{\rm{(6 - 7}}\,\,{\rm{atm)}}\]yields sodium salicylate. This acidification gives salicylic acid. Hence, option (D) is incorrect.
Hence, the two alkyl halides are 2-chloropropane and chloromethane.
Therefore, option C is correct.
Note: Wurtz-Fittig reaction is named after Wilhelm Rudolph Fittig and Charles Adolphe Wurtz. Wurtz-Fittig reaction is used in the laboratory for the preparation of organosilicon compounds and many substituted aromatic compounds.
Complete Step by Step Solution:
Many of the substitution reactions which do occur when organometallic reagents are used as nucleophiles proceed with only moderate yields. The coupling of haloalkanes in the presence of an active metal is known as the Wurtz reaction. The reaction is usually accomplished by adding sodium metal to an organohalogen compound in an inert solvent. The organosodium compound which forms combines with an unreacted haloalkane.
The general equation of Wurtz reaction is as shown below:
\[{\rm{R}} - {\rm{X}} + 2{\rm{Na}} + {\rm{X}} - {\rm{R}} \xrightarrow[ether]{dry} {\rm{R}} - {\rm{R}} + {\rm{2NaX}}\];
where \[{\rm{R}}\]= alkyl group
\[{\rm{X}}\]= halogen atom
Hence, option (A) is incorrect.
In Sandmeyer’s reaction, benzene diazonium chloride is treated with cuprous halide \[{\rm{(C}}{{\rm{u}}_{\rm{2}}}{\rm{C}}{{\rm{l}}_{\rm{2}}}\,\,{\rm{or}}\,\,{\rm{C}}{{\rm{u}}_{\rm{2}}}{\rm{B}}{{\rm{r}}_{\rm{2}}}{\rm{)}}\]. Iodobenzene is obtained by treating it with potassium iodide \[{\rm{(KI)}}\]. Hence, option (B) is incorrect.
In the Wurtz-Fittig reaction, an alkyl halide and a solution of an aryl halide (aromatic halogen) in ether is made to react with sodium.

Image: reaction of an aromatic halogen (chlorobenzene) with alkyl halide (chloromethane) in presence of sodium and ether.
Hence, option (C) is correct.
In Kolbe’s reaction, sodium phenoxide on heating to \[{\rm{400}}\,\,{\rm{K}}\]with \[{\rm{C}}{{\rm{O}}_2}\]under pressure \[{\rm{(6 - 7}}\,\,{\rm{atm)}}\]yields sodium salicylate. This acidification gives salicylic acid. Hence, option (D) is incorrect.
Hence, the two alkyl halides are 2-chloropropane and chloromethane.
Therefore, option C is correct.
Note: Wurtz-Fittig reaction is named after Wilhelm Rudolph Fittig and Charles Adolphe Wurtz. Wurtz-Fittig reaction is used in the laboratory for the preparation of organosilicon compounds and many substituted aromatic compounds.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




