The product formed when benzene is nitrated by fuming nitric acid is
A. m-dinitrobenzene
B. Nitrobenzene
C. sym-trinitrobenzene
D. None of these
Answer
251.4k+ views
Hint: Benzene is an electron dense species due to presence of pi electrons. It can donate its electron pair. On reaction with nitric acid it undergoes nitration reaction and forms nitrobenzene as a product.
Complete Step by Step Answer:
Benzene is a nucleophile due to the presence of high electron density. On nitration the electrophile nitro group can easily attack any position of benzene and form a product. Benzene on fuming with nitric acid means the reaction takes place in presence of water.
Water is a polar solvent which activates the benzene ring so much that on treatment with nitric acid in water the electrophile or the nitro group attacks the ortho, meta and one para positions at a time and gives sym-trinitrobenzene as a major product along with three moles of water.
The nitration reaction of benzene is as follows:
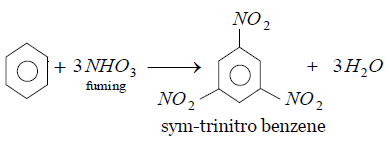
Thus the correct option is C.
Note: Nitration reaction is an aromatic electrophilic substitution reaction where an electrophile attacks a benzene ring. In the nitration of the benzene ring the nitro group acts as an electrophile due to low electron density and it easily attacks the more electron dense system that is the benzene ring to give product.
Complete Step by Step Answer:
Benzene is a nucleophile due to the presence of high electron density. On nitration the electrophile nitro group can easily attack any position of benzene and form a product. Benzene on fuming with nitric acid means the reaction takes place in presence of water.
Water is a polar solvent which activates the benzene ring so much that on treatment with nitric acid in water the electrophile or the nitro group attacks the ortho, meta and one para positions at a time and gives sym-trinitrobenzene as a major product along with three moles of water.
The nitration reaction of benzene is as follows:

Thus the correct option is C.
Note: Nitration reaction is an aromatic electrophilic substitution reaction where an electrophile attacks a benzene ring. In the nitration of the benzene ring the nitro group acts as an electrophile due to low electron density and it easily attacks the more electron dense system that is the benzene ring to give product.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




