The oxidation states of P atom in $POC{{l}_{3}},{{H}_{2}}P{{O}_{3}}$ and ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ respectively are:
Answer
249.3k+ views
Hint: Draw the structures of $POC{{l}_{3}},{{H}_{2}}P{{O}_{3}}$ and ${{H}_{4}}{{P}_{2}}{{O}_{6}}$ .Keep in mind the valency of the P atom to avoid any mistakes. Try calculating oxidation state of P by trying to balance the positive and
the negative charge in the compound.
Complete step by step solution:
We will draw the structure for the three compounds given above to find the oxidation state.
Structure of $POC{{l}_{3}}$:
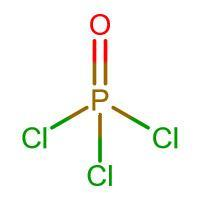
There are 5 bonds around the central P atom, hence the oxidation state of P is 5.
Structure of ${{H}_{2}}P{{O}_{3}}$:
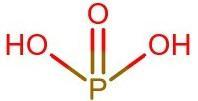
There are 4 bonds around the central P atom, hence the oxidation state of P is 4.
Structure of ${{H}_{4}}{{P}_{2}}{{O}_{6}}$:
In the above case, we consider the number of bonds around each P excluding the P-P bond as it is not counted when calculating the oxidation state. There are 4 bonds around each of the P atoms, hence the oxidation state is 4.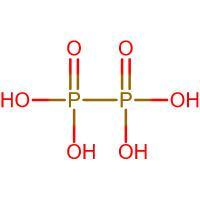
Therefore, the correct answer is option (A).
Additional information:
While calculating the oxidation state of an atom we count the number of bonds around the atom. In reality, we mean to count the number of heterogeneous bonds i.e. both the atoms are not identical. Homogenous bonds like P-P do not contribute to the oxidation state of the atom and hence it is not counted.
Note: While writing the structure of the compound, keep in mind the maximum valency the central atom can show and make the bonds accordingly. Do not count homogenous bonds while calculating the oxidation state of the central atom.
the negative charge in the compound.
Complete step by step solution:
We will draw the structure for the three compounds given above to find the oxidation state.
Structure of $POC{{l}_{3}}$:

There are 5 bonds around the central P atom, hence the oxidation state of P is 5.
Structure of ${{H}_{2}}P{{O}_{3}}$:

There are 4 bonds around the central P atom, hence the oxidation state of P is 4.
Structure of ${{H}_{4}}{{P}_{2}}{{O}_{6}}$:
In the above case, we consider the number of bonds around each P excluding the P-P bond as it is not counted when calculating the oxidation state. There are 4 bonds around each of the P atoms, hence the oxidation state is 4.

Therefore, the correct answer is option (A).
Additional information:
While calculating the oxidation state of an atom we count the number of bonds around the atom. In reality, we mean to count the number of heterogeneous bonds i.e. both the atoms are not identical. Homogenous bonds like P-P do not contribute to the oxidation state of the atom and hence it is not counted.
Note: While writing the structure of the compound, keep in mind the maximum valency the central atom can show and make the bonds accordingly. Do not count homogenous bonds while calculating the oxidation state of the central atom.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




