The IUPAC name of the compound glycerine is:
A) 1,2,3-trihydroxypropane
B) 3-hydroxypentane-1,5-diol
C) 1,2,3-hydroxypropane
D) propane-1,2,3-triol
Answer
258.9k+ views
Hint: We know that IUPAC naming is the standard naming of organic compounds. There is a set of rules in naming of organic compounds. Identify the longest carbon chain and functional groups after drawing the structure.
Complete step-by-step solution:
Functional groups are made up of one or more atoms with distinctive chemical properties, independent of what is connected to them. The atoms of functional groups are bound by covalent bonds with each other and with the rest of the molecule.
The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is ‘prefix +parent chain +suffix’. The longest chain of carbon atoms is the parent chain of the compound. Suffix denotes the name and position of functional groups and prefix denote the substituent and its position in the compound.
The structure of glycerine is,
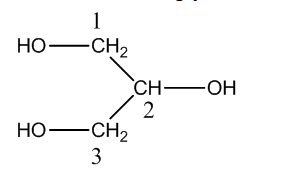
Now, we have to identify the parent chain in the glycerine. There are three carbon atoms in the parent chain and all bonds are single bonds. So, name of the parent chain is ‘propane’. The functional group –OH is present in all the three carbon atoms. So, suffix is ‘1,2,3-triol’ So, the IUPAC naming of glycerine is propan-1,2,3-triol.
Hence, correct option is D.
Note: The group with the formula OH is a hydroxyl group. This is made up of oxygen bound to hydrogen. Throughout organic chemistry, hydroxyl groups are found throughout alcohols and carboxylic acids. All the negatively charged ${\rm{ - O}}{{\rm{H}}^ - }$ anion, called hydroxide, and the stable radical ${\rm{ - OH}}$, known as hydroxyl radical, compose of an unbounded group of hydroxyl.
Complete step-by-step solution:
Functional groups are made up of one or more atoms with distinctive chemical properties, independent of what is connected to them. The atoms of functional groups are bound by covalent bonds with each other and with the rest of the molecule.
The name of an organic compound has three parts, prefix, parent chain and suffix. The format of naming is ‘prefix +parent chain +suffix’. The longest chain of carbon atoms is the parent chain of the compound. Suffix denotes the name and position of functional groups and prefix denote the substituent and its position in the compound.
The structure of glycerine is,

Now, we have to identify the parent chain in the glycerine. There are three carbon atoms in the parent chain and all bonds are single bonds. So, name of the parent chain is ‘propane’. The functional group –OH is present in all the three carbon atoms. So, suffix is ‘1,2,3-triol’ So, the IUPAC naming of glycerine is propan-1,2,3-triol.
Hence, correct option is D.
Note: The group with the formula OH is a hydroxyl group. This is made up of oxygen bound to hydrogen. Throughout organic chemistry, hydroxyl groups are found throughout alcohols and carboxylic acids. All the negatively charged ${\rm{ - O}}{{\rm{H}}^ - }$ anion, called hydroxide, and the stable radical ${\rm{ - OH}}$, known as hydroxyl radical, compose of an unbounded group of hydroxyl.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




