The geometry of \[Ni{{(CO)}_{4}}\]and \[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]are :
(A) Both square planar
(B) Tetrahedral and square planar, respectively
(C) Both tetrahedral
(D) Square planar and tetrahedral, respectively
Answer
264.6k+ views
Hint: \[Ni{{(CO)}_{4}}\] and \[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\] are examples of organometallic complexes. Nickel belongs to d-block elements. Nickel can form metallic complexes easily with ligands. Generally strong field ligands form tetrahedral complexes with nickel and weak field complexes form square planar complexes with nickel.
Complete step by step by answer:
The IUPAC name of \[Ni{{(CO)}_{4}}\] is tetracarbonyl nickel (0).
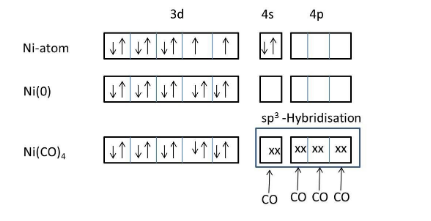
We know that carbonyl is a strong ligand, that’s why the two electrons in 4s orbital are shifted to 3d orbital of nickel.
Meanwhile no inner empty 3d orbital is available to form a bond with carbonyl ligand, 4s and 4p orbitals will take electrons from four carbonyl ligands and form four covalent bonds through the carbon end to the nickel atom.
The hybridization of nickel atoms in \[Ni{{(CO)}_{4}}\] is\[s{{p}^{3}}\].
The geometry of Nickel tetracarbonyl is tetrahedral.
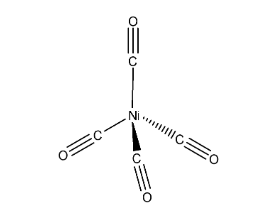
In the above structure it represents that the two carbonyls are in one plane (imagine x-axis), other two carbonyls are in different planes (y-axis and z-axis) to reduce the repulsions and then the structure will be tetrahedral.
IUPAC name of \[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]is Dichlorobis(triphenylphosphine)nickel(II).
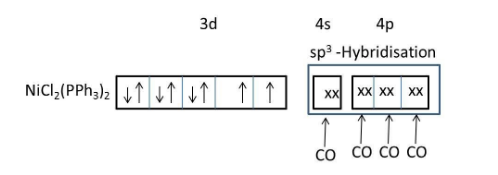
\[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]consists of both weak field ligand (\[Cl\]) and strong field ligand (\[PP{{h}_{3}}\]).
Presence of both weak field and strong field ligands \[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]should exist in two geometries (tetrahedral and square planar).
But, due to the presence of larger ligands favoring the less crowded tetrahedral geometry.
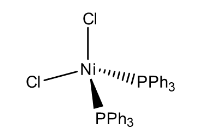
In the above structure it represents that the two chlorine atoms are in one plan (imagine x-axis), other two triphenylphosphine ligands are in different planes (y-axis and z-axis) to reduce the repulsions and then the structure will be tetrahedral.
Hence, the geometry of the Dichlorobis(triphenylphosphine)nickel(II) is tetrahedral.
So, the correct option is C.
Note: Don’t be confused with tetrahedral and square planar geometries. If the ligand forms bonds with 3d orbital of nickel then there is a chance of formation of square planar geometry.
Complete step by step by answer:
The IUPAC name of \[Ni{{(CO)}_{4}}\] is tetracarbonyl nickel (0).

We know that carbonyl is a strong ligand, that’s why the two electrons in 4s orbital are shifted to 3d orbital of nickel.
Meanwhile no inner empty 3d orbital is available to form a bond with carbonyl ligand, 4s and 4p orbitals will take electrons from four carbonyl ligands and form four covalent bonds through the carbon end to the nickel atom.
The hybridization of nickel atoms in \[Ni{{(CO)}_{4}}\] is\[s{{p}^{3}}\].
The geometry of Nickel tetracarbonyl is tetrahedral.

In the above structure it represents that the two carbonyls are in one plane (imagine x-axis), other two carbonyls are in different planes (y-axis and z-axis) to reduce the repulsions and then the structure will be tetrahedral.
IUPAC name of \[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]is Dichlorobis(triphenylphosphine)nickel(II).

\[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]consists of both weak field ligand (\[Cl\]) and strong field ligand (\[PP{{h}_{3}}\]).
Presence of both weak field and strong field ligands \[Ni{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}\]should exist in two geometries (tetrahedral and square planar).
But, due to the presence of larger ligands favoring the less crowded tetrahedral geometry.

In the above structure it represents that the two chlorine atoms are in one plan (imagine x-axis), other two triphenylphosphine ligands are in different planes (y-axis and z-axis) to reduce the repulsions and then the structure will be tetrahedral.
Hence, the geometry of the Dichlorobis(triphenylphosphine)nickel(II) is tetrahedral.
So, the correct option is C.
Note: Don’t be confused with tetrahedral and square planar geometries. If the ligand forms bonds with 3d orbital of nickel then there is a chance of formation of square planar geometry.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




