The function(s) of salt bridge in a cell is/are
A. It maintains standard electrode potential of cell constant which depends on several factors.
B. It completes the electrical circuit.
C. It departs both the solutions from each other.
D. It maintains the electrical neutrality of both electrolytic solutions.
Answer
264.6k+ views
Hint: A salt bridge is a chemical junction that connects the anodic and cathodic compartments in a cell or electrolytic solution thereby completing the circuit. It prevents the cell from taking its reaction to equilibrium and balances the charges in both the solutions.
Complete step-by-step answer:
A salt bridge is a device that is used in a galvanic cell for connecting its oxidation and reduction half cells where a weak electrolyte is used. It usually consists of a strong electrolyte which is further made up of ions such as KCl. These are generally used in a galvanic cell such as a voltaic cell or Daniel cell.
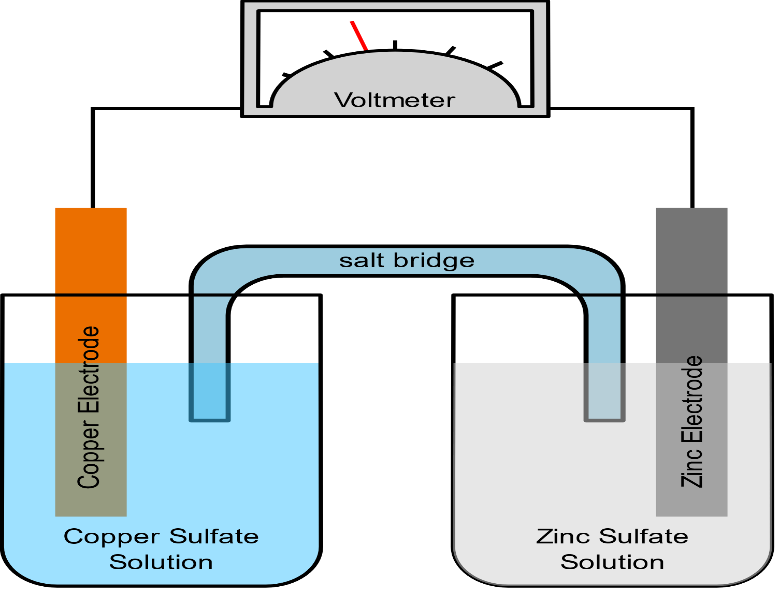
The main function of a salt bridge is to maintain the electrical neutrality of both electrolytic solutions within the internal circuit. It basically helps in preventing the accumulation of positive and negative charges around the respective electrolytic electrodes and further allowing a smooth reaction to occur. It also provides continual flow of electrons. However, the purpose of a salt bridge is not just to move electrons from the electrolyte rather to maintain charge balance because the electrons move from one-half cell to the other.
Salt bridge avoids the diffusion or mechanical flow of solution from one-half cell to another. It minimizes the liquid-liquid junction potential which arises between two solutions when they are in contact with each other. In short, it acts as an electrical contact between two half cells.
Hence, the correct options are (A), (B), (C) and (D).
Note: If there is no salt bridge in the cell, then the anodic and cathodic half cells will be joined only from one side and the electrical circuit will remain incomplete. There occurs a further accumulation of negative charge in one solution and positive in another.
Complete step-by-step answer:
A salt bridge is a device that is used in a galvanic cell for connecting its oxidation and reduction half cells where a weak electrolyte is used. It usually consists of a strong electrolyte which is further made up of ions such as KCl. These are generally used in a galvanic cell such as a voltaic cell or Daniel cell.

The main function of a salt bridge is to maintain the electrical neutrality of both electrolytic solutions within the internal circuit. It basically helps in preventing the accumulation of positive and negative charges around the respective electrolytic electrodes and further allowing a smooth reaction to occur. It also provides continual flow of electrons. However, the purpose of a salt bridge is not just to move electrons from the electrolyte rather to maintain charge balance because the electrons move from one-half cell to the other.
Salt bridge avoids the diffusion or mechanical flow of solution from one-half cell to another. It minimizes the liquid-liquid junction potential which arises between two solutions when they are in contact with each other. In short, it acts as an electrical contact between two half cells.
Hence, the correct options are (A), (B), (C) and (D).
Note: If there is no salt bridge in the cell, then the anodic and cathodic half cells will be joined only from one side and the electrical circuit will remain incomplete. There occurs a further accumulation of negative charge in one solution and positive in another.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




