The correct statement on the isomerism associated with the following complex ions is:
(a) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{5}}N{{H}_{3}} \right]}^{2+}}$
(b) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{4}}{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{2+}}$
(c) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{3}}{{\left( N{{H}_{3}} \right)}_{3}} \right]}^{2+}}$
(A) Both (a) and (b) show geometrical isomerism.
(B) (b) shows geometrical isomerism and (c) shows optical isomerism.
(C) (a) shows geometrical isomerism and (b) shows optical isomerism.
(D) Both (b) and (c) show geometrical isomerism.
Answer
260.4k+ views
Hint: Recollect the concept of coordination chemistry. Think about what is optical isomerism and what is geometrical isomerism. Draw the structures of the given three complexes and try to identify the isomerism present in them. Then choose the most suitable option as an answer.
Complete step by step solution:
- Stereoisomerism is the phenomena in which atoms have a different spatial arrangement. It is classified into two types: Optical isomerism and Geometrical isomerism.
- In optical isomerism, the two isomers are non-superimposable mirror images of each other. So, the point of contact between the metal and the ligands remains the same.
- In geometrical isomerism, the two isomers will have different arrangements of ligands around the central metal atom. This involves cis- and trans- isomers.
- Let’s draw the structure of the given coordination complexes.
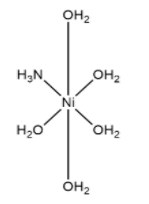
(a) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{5}}N{{H}_{3}} \right]}^{2+}}$
(b) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{4}}{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{2+}}$
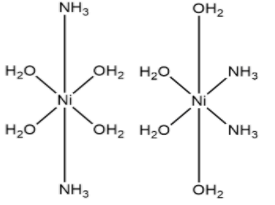
This complex shows geometrical isomerism and has trans and cis isomers which are shown above respectively.
(c) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{3}}{{\left( N{{H}_{3}} \right)}_{3}} \right]}^{2+}}$
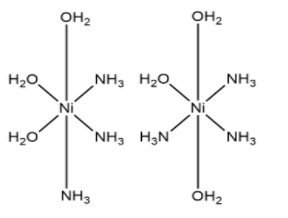
- This complex has facial and meridional, fac and mer isomers which are shown above. Therefore, this complex also shows geometrical isomerism.
- Therefore, the complexes (b) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{4}}{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{2+}}$ and (c) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{3}}{{\left( N{{H}_{3}} \right)}_{3}} \right]}^{2+}}$ show geometrical isomerism.
Therefore, the correct answer is option (D).
Note: Remember fac isomer is the one in which ligands two ligands are in plane with metal and one ligand is out of plane. mer-isomer is the one in which three ligands and metal are in the same plane. cis-isomer is the one in which same ligands are adjacent to each other and trans isomer is the one in which same ligands are exactly opposite to each other.
Complete step by step solution:
- Stereoisomerism is the phenomena in which atoms have a different spatial arrangement. It is classified into two types: Optical isomerism and Geometrical isomerism.
- In optical isomerism, the two isomers are non-superimposable mirror images of each other. So, the point of contact between the metal and the ligands remains the same.
- In geometrical isomerism, the two isomers will have different arrangements of ligands around the central metal atom. This involves cis- and trans- isomers.
- Let’s draw the structure of the given coordination complexes.
(a) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{5}}N{{H}_{3}} \right]}^{2+}}$

(b) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{4}}{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{2+}}$

This complex shows geometrical isomerism and has trans and cis isomers which are shown above respectively.
(c) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{3}}{{\left( N{{H}_{3}} \right)}_{3}} \right]}^{2+}}$

- This complex has facial and meridional, fac and mer isomers which are shown above. Therefore, this complex also shows geometrical isomerism.
- Therefore, the complexes (b) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{4}}{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{2+}}$ and (c) ${{\left[ Ni{{\left( {{H}_{2}}O \right)}_{3}}{{\left( N{{H}_{3}} \right)}_{3}} \right]}^{2+}}$ show geometrical isomerism.
Therefore, the correct answer is option (D).
Note: Remember fac isomer is the one in which ligands two ligands are in plane with metal and one ligand is out of plane. mer-isomer is the one in which three ligands and metal are in the same plane. cis-isomer is the one in which same ligands are adjacent to each other and trans isomer is the one in which same ligands are exactly opposite to each other.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




