The addition of HBr to an alkene in the presence of peroxide is an example of:
A. Electrophilic addition reaction
B. Nucleophilic addition reaction
C. Free-radical addition reaction
D. Formation of carbocation as an intermediate
Answer
264k+ views
Hint: When hydrogen bromide is added to alkenes in presence of peroxide, it reacts with anti-Markovnikov’s addition mechanism. Peroxides form free radicals that initiate the reaction and add bromine radical at terminal carbon.
Complete step-by-step answer:
Peroxides have a weak oxygen-oxygen bond which on heating results in homolytic fragmentation of this bond i.e. the bond breaks in order to leave one unpaired electron on each atom involved in the reaction. Strong sources of light such as floodlight or other source of light radiation which reaches into the near UV might also serve to weaken this bond.
Only a catalytic amount of peroxide is needed to get the reaction started, although one molar equivalent of HBr is essentially required to result in complete addition of HBr to the alkene.
This results in a highly reactive alkoxy radical which then abstracts hydrogen from H-Br, releasing a bromine radical. The bromine radical is the one that is added to the alkene from the molecule hydrogen bromide.
Preferably addition to an alkene tends to occur in such a way that the most stable free radical is formed, tertiary radical here in HBr. That’s the reason that bromine ends up on the least substituted carbon of the alkene. This tertiary radical then eliminates hydrogen from H-Br, liberating a bromine radical, and this way the cycle continues.
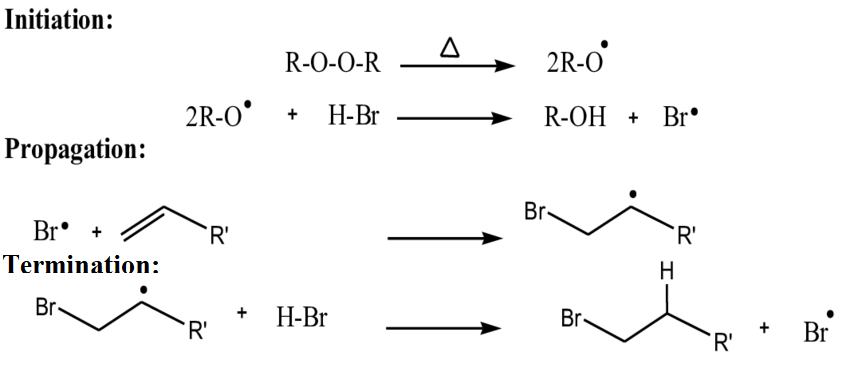
Hence, the correct option is (C).
Note: In absence of peroxide, if alkene reacts with hydrogen bromide, the attack of bromine is due to electrophilic addition reaction in which carbocation is formed as an intermediate. It is also called Markovnikov’s addition in which attack takes place on more substituted carbon.
Complete step-by-step answer:
Peroxides have a weak oxygen-oxygen bond which on heating results in homolytic fragmentation of this bond i.e. the bond breaks in order to leave one unpaired electron on each atom involved in the reaction. Strong sources of light such as floodlight or other source of light radiation which reaches into the near UV might also serve to weaken this bond.
Only a catalytic amount of peroxide is needed to get the reaction started, although one molar equivalent of HBr is essentially required to result in complete addition of HBr to the alkene.
This results in a highly reactive alkoxy radical which then abstracts hydrogen from H-Br, releasing a bromine radical. The bromine radical is the one that is added to the alkene from the molecule hydrogen bromide.
Preferably addition to an alkene tends to occur in such a way that the most stable free radical is formed, tertiary radical here in HBr. That’s the reason that bromine ends up on the least substituted carbon of the alkene. This tertiary radical then eliminates hydrogen from H-Br, liberating a bromine radical, and this way the cycle continues.

Hence, the correct option is (C).
Note: In absence of peroxide, if alkene reacts with hydrogen bromide, the attack of bromine is due to electrophilic addition reaction in which carbocation is formed as an intermediate. It is also called Markovnikov’s addition in which attack takes place on more substituted carbon.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




