Propionamide on Hofmann degradation gives-
A. Methyl amine
B. Ethyl amine
C. Propyl amine
D. Ethyl cyanide
Answer
268.8k+ views
Hint: Propionamide is an amide compound with general formula \[RCON{H_2}\] . The compound formed has one less carbon atom than the amide.
Complete Step by Step Solution:
Hoffmann degradation reaction is a type of reaction where primary amide is converted to a primary amine. The reaction takes place in presence of a strong base which attacks the amide. The reaction takes place by heating the primary amide using a halogen mixture of chlorine or bromine, a strong base and water.
In this reaction, halogen mainly reacts with sodium hydroxide which forms sodium hypobromite which changes the primary amide into an isocyanate intermediate. Water attacks the isocyanate intermediate causing the proton transfer and resulting in the formation of ammonium cation. Due to the thermal condition, the carbon dioxide explodes and after quenching ammonium cation, a primary amine product is formed.
When propionamide is heated with bromine in presence of potassium hydroxide or when propionamide undergoes Hofmann degradation, the product formed is ethyl amine.
The Hoffman degradation reaction of propionamide is shown below.
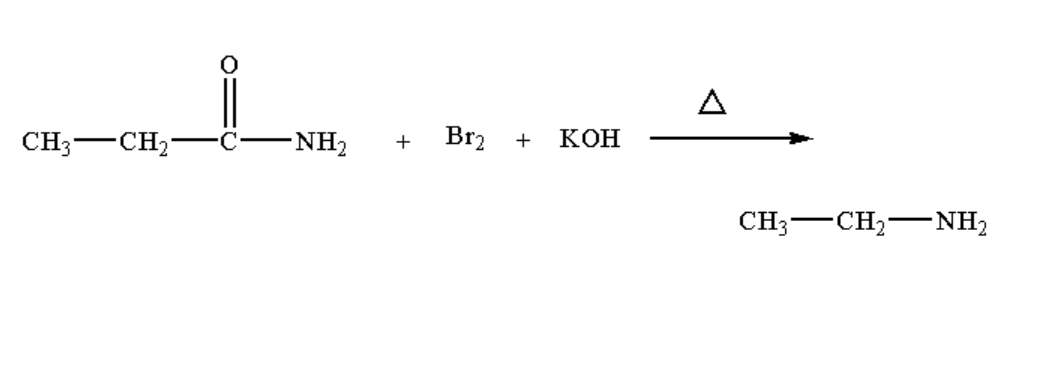
Image: Hofmann degradation reaction of propionamide
Therefore, the correct option is B.
Note: Hofmann degradation reaction is also termed as Hoffmann bromamide reaction. There is another term known as Hofmann elimination reaction in which tertiary amines and alkenes are formed as a product by reacting quaternary ammonium with methyl iodide and heating the compound formed with silver oxide and water.
The general reaction is shown below.
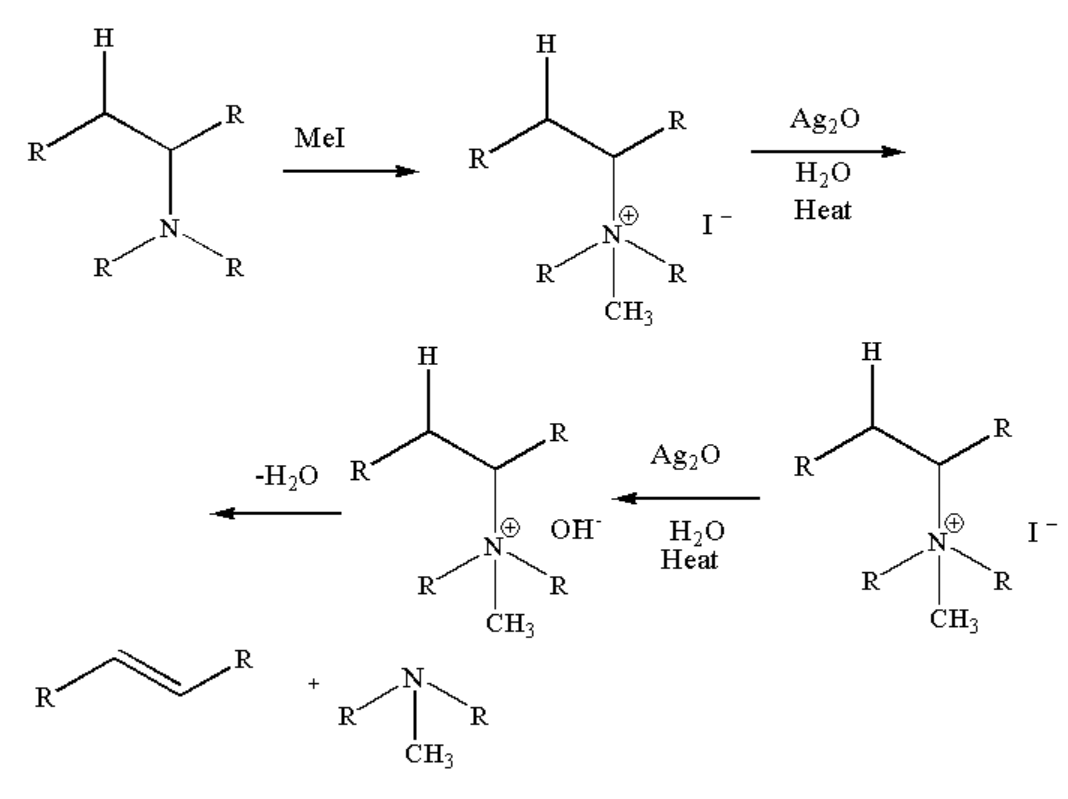
Image: Hofmann elimination reaction
Complete Step by Step Solution:
Hoffmann degradation reaction is a type of reaction where primary amide is converted to a primary amine. The reaction takes place in presence of a strong base which attacks the amide. The reaction takes place by heating the primary amide using a halogen mixture of chlorine or bromine, a strong base and water.
In this reaction, halogen mainly reacts with sodium hydroxide which forms sodium hypobromite which changes the primary amide into an isocyanate intermediate. Water attacks the isocyanate intermediate causing the proton transfer and resulting in the formation of ammonium cation. Due to the thermal condition, the carbon dioxide explodes and after quenching ammonium cation, a primary amine product is formed.
When propionamide is heated with bromine in presence of potassium hydroxide or when propionamide undergoes Hofmann degradation, the product formed is ethyl amine.
The Hoffman degradation reaction of propionamide is shown below.

Image: Hofmann degradation reaction of propionamide
Therefore, the correct option is B.
Note: Hofmann degradation reaction is also termed as Hoffmann bromamide reaction. There is another term known as Hofmann elimination reaction in which tertiary amines and alkenes are formed as a product by reacting quaternary ammonium with methyl iodide and heating the compound formed with silver oxide and water.
The general reaction is shown below.

Image: Hofmann elimination reaction
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




