What is the product of the reaction of nitrous acid with aliphatic primary amine in the cold?
(A) A diazonium salt
(B) An alcohol
(C) A nitrite
(D) A dye
Answer
266.7k+ views
Hint: Amines are the compounds that contain an $N{{H}_{2}}$functional group in their structure. These are basic in nature as they contain nitrogen with a lone pair of electrons. These compounds are sensitive to pyrolysis when the $N{{H}_{2}}$ group is present.
Complete Step by Step Solution:
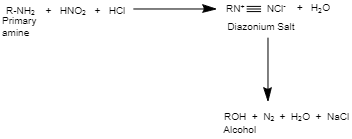
Reaction of $HN{{O}_{2}}$ with aliphatic amines, also known as the nitrous acid test, is an important test for distinguishing primary, secondary, and tertiary amines. When nitrous acid $HN{{O}_{2}}$ ) reacts with aliphatic primary amine in the cold, it first processes a diazonium salt, which is unstable. The diazonium salt then decomposes to alcohol along with the evolution of nitrogen gas (${{N}_{2}}$ ) and water.
Correct option: (B) An alcohol.
Additional information: At room temperature, primary amines are liquid, whereas secondary and tertiary amines are solid. Treatment of nitrous acid ($HN{{O}_{2}}$ ) with secondary amines gives an insoluble oil layer of nitrosamine; with tertiary amines, it gives a clear solution of quaternary ammonium salts.
Note: The nitrous acid test is used as a test for amines in organic chemistry to identify whether the compound has an amino functional group present or not. The primary amine can further be identified with the carbylamine test, in which amines are treated with chloroform in the presence of alkali. An alkyl isocyanide ($RNC$ ) is formed in this process. Both aliphatic and aromatic amines can give this test.
Complete Step by Step Solution:
Reaction of $HN{{O}_{2}}$ with aliphatic amines, also known as the nitrous acid test, is an important test for distinguishing primary, secondary, and tertiary amines. When nitrous acid $HN{{O}_{2}}$ ) reacts with aliphatic primary amine in the cold, it first processes a diazonium salt, which is unstable. The diazonium salt then decomposes to alcohol along with the evolution of nitrogen gas (${{N}_{2}}$ ) and water.

Correct option: (B) An alcohol.
Additional information: At room temperature, primary amines are liquid, whereas secondary and tertiary amines are solid. Treatment of nitrous acid ($HN{{O}_{2}}$ ) with secondary amines gives an insoluble oil layer of nitrosamine; with tertiary amines, it gives a clear solution of quaternary ammonium salts.
Note: The nitrous acid test is used as a test for amines in organic chemistry to identify whether the compound has an amino functional group present or not. The primary amine can further be identified with the carbylamine test, in which amines are treated with chloroform in the presence of alkali. An alkyl isocyanide ($RNC$ ) is formed in this process. Both aliphatic and aromatic amines can give this test.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




