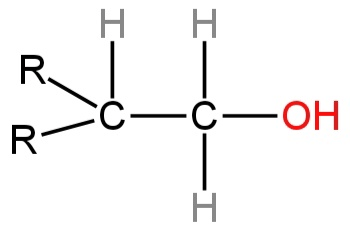
Product A in the following reaction is
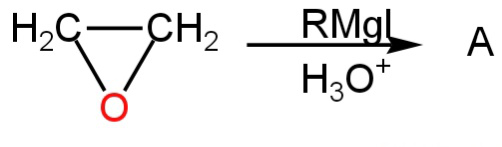
A.
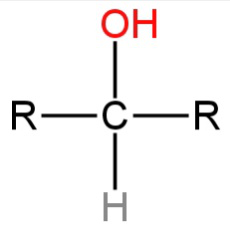
B.
C.
D.
Answer
253.8k+ views
Hint: C2H4O is ethylene oxide. It is an epoxide. Epoxides are ethers having the oxygen atom in a three-membered ring. Epoxides are also called oxiranes. RMgI i.e, alkyl magnesium iodide called Grignard's reagent.
Complete Step by Step Solution:
- Ethylene oxide is a strained ring, it readily undergoes several additional reactions which result in ring-opening.
- RMgI is an organometallic compound. An organometallic compound is defined as a compound that contains a direct carbon-metal bond.
- RMgX is Grignard's reagent. It is prepared by the action of alkyl halides on magnesium metal in the presence of dry ether.
- The C-Mg bond in Grignard reagents is polar. The carbon atom is additionally electronegative than magnesium.
- The electrons of the C-Mg bond are attracted to the carbon atom. So, the carbon atom is partially negatively charged, and the magnesium atom has a partial positive charge.
- The alkyl groups in Grignard reagents are electron-rich and can work as nucleophiles.
- These alkyl groups would strike polar molecules at the area of insufficient electron density. Thus, Grignard reagents typically undergo nucleophilic substitution and nucleophilic addition reactions.
- In ethylene oxide, on the other hand, oxygen is much more electronegative than carbon or hydrogen.
- The C-O and O-H bonds are all polar. Grignard reagents are powerful nucleophiles and can react with the carbon atom of epoxides. The reaction results in ring-opening and the formation of alcohol.
This reaction happens in two steps.
Step-1
The attack of the RMgI on ethylene oxide leads to the formation of an additional product that is unstable.
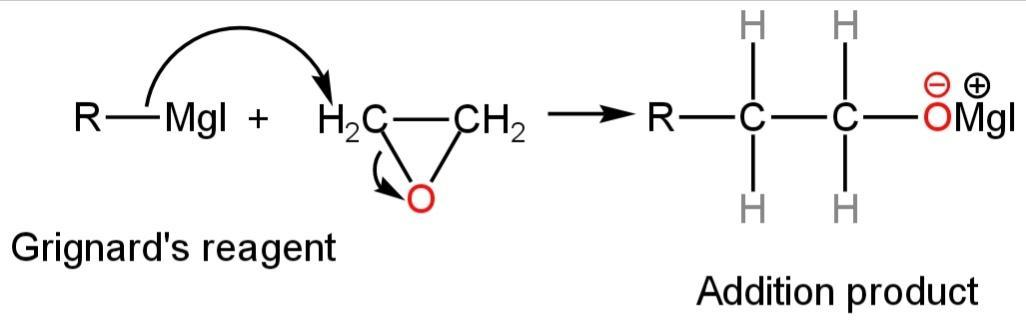
Image: Formation of additional products.
Step-2
The additional product undergoes hydrolysis to form alcohol.
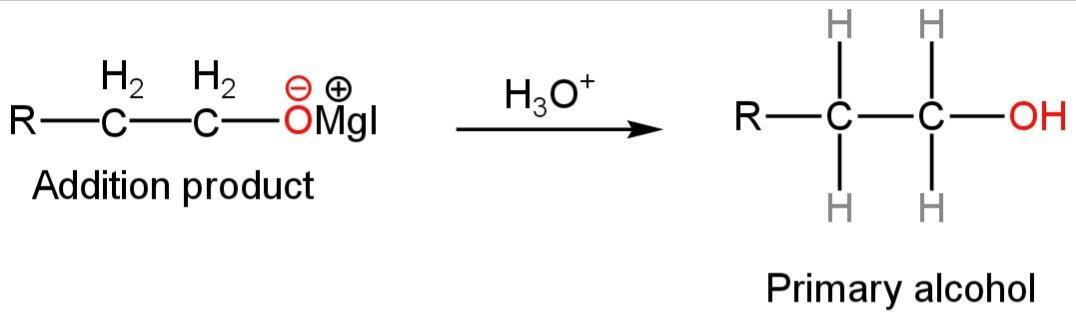
So, ethylene oxide in reaction with RMgI forms primary alcohol.
So, option A is correct.
Note: It must be noted that the ease of reaction of epoxide with RMgI increases with the increase in the ionic character of the C-Mg bond. In the preparation of Grignard reagents, a solution of the alkyl halide is dipped in dry ether into the reaction flask comprising magnesium ribbon suspended in dry ether. Dry ether is utilised as moisture may react with Grignard's reagent to form alkane. Thus, moisture or impurities discourage the yield of Grignard's reagent.
Complete Step by Step Solution:
- Ethylene oxide is a strained ring, it readily undergoes several additional reactions which result in ring-opening.
- RMgI is an organometallic compound. An organometallic compound is defined as a compound that contains a direct carbon-metal bond.
- RMgX is Grignard's reagent. It is prepared by the action of alkyl halides on magnesium metal in the presence of dry ether.
- The C-Mg bond in Grignard reagents is polar. The carbon atom is additionally electronegative than magnesium.
- The electrons of the C-Mg bond are attracted to the carbon atom. So, the carbon atom is partially negatively charged, and the magnesium atom has a partial positive charge.
- The alkyl groups in Grignard reagents are electron-rich and can work as nucleophiles.
- These alkyl groups would strike polar molecules at the area of insufficient electron density. Thus, Grignard reagents typically undergo nucleophilic substitution and nucleophilic addition reactions.
- In ethylene oxide, on the other hand, oxygen is much more electronegative than carbon or hydrogen.
- The C-O and O-H bonds are all polar. Grignard reagents are powerful nucleophiles and can react with the carbon atom of epoxides. The reaction results in ring-opening and the formation of alcohol.
This reaction happens in two steps.
Step-1
The attack of the RMgI on ethylene oxide leads to the formation of an additional product that is unstable.

Image: Formation of additional products.
Step-2
The additional product undergoes hydrolysis to form alcohol.

So, ethylene oxide in reaction with RMgI forms primary alcohol.
So, option A is correct.
Note: It must be noted that the ease of reaction of epoxide with RMgI increases with the increase in the ionic character of the C-Mg bond. In the preparation of Grignard reagents, a solution of the alkyl halide is dipped in dry ether into the reaction flask comprising magnesium ribbon suspended in dry ether. Dry ether is utilised as moisture may react with Grignard's reagent to form alkane. Thus, moisture or impurities discourage the yield of Grignard's reagent.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




