Oxirane is
A. Ethylene oxide
B. Diethyl ether
C. Ethyl glycolate
D. Glycolic ester
Answer
269.7k+ views
Hint: Oxirane is an organic compound having the chemical formula \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{4}}}{\rm{O}}\]. It is a three-membered cyclic compound containing the ether functional group. It is also the simplest epoxide.
Complete Step by Step Solution:
Oxirane is the IUPAC name of \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{4}}}{\rm{O}}\]. This compound is also known as ethylene oxide. This is a simple epoxide which is a three-membered ring having one oxygen atom and two carbon atoms. The atoms are bonded to each other through sigma bonds.
The structure of the compound is as comes after:
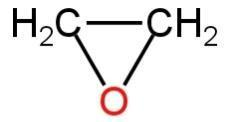
Image: Structure of oxirane.
As it is a strained ring, it readily takes part in several additional reactions which involve ring-opening.
Ethylene oxide has no colour and is an explosive gas with a slightly sweet aroma.
It is industrially yielded by oxidation of ethylene in the presence of a silver catalyst.
The reaction happens as comes next:

Image: Production of oxirane
It is utilised for producing numerous products like detergents, thickeners, solvents, plastics, and different organic chemicals like ethylene glycol, ethanolamines, polyglycol ethers, etc. Ethylene oxide is soluble in water, ethanol, diethyl ether, and many organic solvents. So, oxirane is ethylene oxide.
So, option A is correct.
Note: In epoxides, the ring atoms i.e., two carbon atoms and one oxygen atom are \[{\rm{s}}{{\rm{p}}^{\rm{3}}}\] hybridised. The two \[{\rm{s}}{{\rm{p}}^{\rm{3}}}\] hybrid bonds of oxygen are filled. The C-O-C bond angle of an epoxide must be \[{\rm{60^\circ }}\], a significant divergence from the tetrahedral bond angle. So, epoxides have an angle strain making them much more reactive than ethers as opening the ring relieves the strain.
Complete Step by Step Solution:
Oxirane is the IUPAC name of \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{4}}}{\rm{O}}\]. This compound is also known as ethylene oxide. This is a simple epoxide which is a three-membered ring having one oxygen atom and two carbon atoms. The atoms are bonded to each other through sigma bonds.
The structure of the compound is as comes after:

Image: Structure of oxirane.
As it is a strained ring, it readily takes part in several additional reactions which involve ring-opening.
Ethylene oxide has no colour and is an explosive gas with a slightly sweet aroma.
It is industrially yielded by oxidation of ethylene in the presence of a silver catalyst.
The reaction happens as comes next:

Image: Production of oxirane
It is utilised for producing numerous products like detergents, thickeners, solvents, plastics, and different organic chemicals like ethylene glycol, ethanolamines, polyglycol ethers, etc. Ethylene oxide is soluble in water, ethanol, diethyl ether, and many organic solvents. So, oxirane is ethylene oxide.
So, option A is correct.
Note: In epoxides, the ring atoms i.e., two carbon atoms and one oxygen atom are \[{\rm{s}}{{\rm{p}}^{\rm{3}}}\] hybridised. The two \[{\rm{s}}{{\rm{p}}^{\rm{3}}}\] hybrid bonds of oxygen are filled. The C-O-C bond angle of an epoxide must be \[{\rm{60^\circ }}\], a significant divergence from the tetrahedral bond angle. So, epoxides have an angle strain making them much more reactive than ethers as opening the ring relieves the strain.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




