Nylon-6,6 is a:
A. Natural polymer
B. Condensation polymer
C. Addition polymer
D. Substitution polymer
Answer
264k+ views
Hint: Nylon – 66 is formed with the help of two monomers, each of which contains six carbon atoms. The monomers are hexamethylenediamine and adipic acid. It is formed by the polycondensation of the two monomers.
Complete Step by Step Solution:
First we know about Nylon - 6,6
Nylon 6, 6 belongs to the family of synthetic polymers which have linear amide linkages which are known as linear polyamides or nylon. It is synthesised or prepared by the condensation of two similar monomers having 6 carbon atoms, hexamethylenediamine, and adipic acid.
Synthesis Of Nylon 6 6
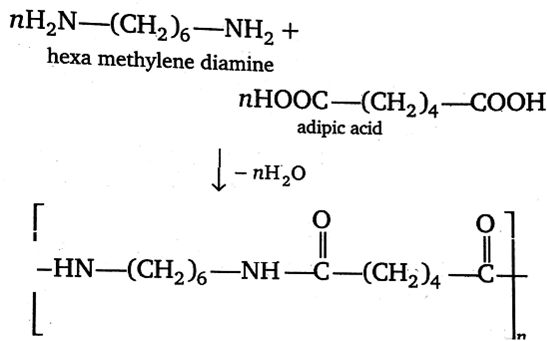
Synthesising or manufacturing the polymer Nylon 6,6 is a step-growth polymerization process that arise through the polycondensation technique, where two monomers hexamethylenediamine $({{H}_{2}}N-{{(C{{H}_{2}})}_{6}}-N{{H}_{2}})$ & adipic acid $(HOOC-{{(C{{H}_{2}})}_{4}}-COOH)$ are condensed to form a polymer called Nylon 6,6. This reaction doesn’t need the catalyst to enhance the rate of the reaction as one of the monomer ie. adipic acid itself acts as a catalyst. Nylon 6,6 is a fibre used in the textile and plastic industries and in daily activities of life.
Note: It is important to note that, cellulose cannot be synthesised by humans because humans take energy from carbohydrates present in food by breaking it into glucose with the help of suitable enzymes but it is very difficult to break cellulose bonding because of the lack of enzyme need to break it.
Complete Step by Step Solution:
First we know about Nylon - 6,6
Nylon 6, 6 belongs to the family of synthetic polymers which have linear amide linkages which are known as linear polyamides or nylon. It is synthesised or prepared by the condensation of two similar monomers having 6 carbon atoms, hexamethylenediamine, and adipic acid.
Synthesis Of Nylon 6 6

Synthesising or manufacturing the polymer Nylon 6,6 is a step-growth polymerization process that arise through the polycondensation technique, where two monomers hexamethylenediamine $({{H}_{2}}N-{{(C{{H}_{2}})}_{6}}-N{{H}_{2}})$ & adipic acid $(HOOC-{{(C{{H}_{2}})}_{4}}-COOH)$ are condensed to form a polymer called Nylon 6,6. This reaction doesn’t need the catalyst to enhance the rate of the reaction as one of the monomer ie. adipic acid itself acts as a catalyst. Nylon 6,6 is a fibre used in the textile and plastic industries and in daily activities of life.
Note: It is important to note that, cellulose cannot be synthesised by humans because humans take energy from carbohydrates present in food by breaking it into glucose with the help of suitable enzymes but it is very difficult to break cellulose bonding because of the lack of enzyme need to break it.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




